Thalidomide: A History of the Drug and Its Victims
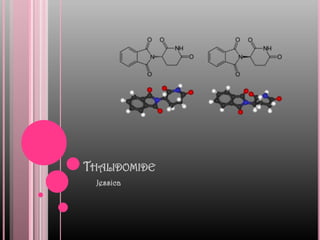
- 2. What is Thalidomide? Thalidomide is a sedative-hypnotic and multiple myeloma medication. The drug is a potent teratogen in rabbits and primates including humans: severe birth defects may result if the drug is taken during pregnancy. Formula for thalidomide is C13H10N2O4.
- 3. History of thalidomide They say that Thalidomide was developed by Germans in the pharmaceutical company Grünenthal in Stolberg but some people say that this claim has been canceled. Dr. Martin W Johnson, who is the director of Thalidomide Trust in the United Kingdom, showed evidences that Thalidomide was developed as an antidote to nerve gases. Some nerve gases are sarin. Others say the drug was synthesized by British scientists. They found that Thalidomide was found to be an effective tranquilizer and painkiller and was proclaimed a "wonder drug" for insomnia, coughs, colds and headaches. It was also found to be an effective antiemetic which had an inhibitory effect on morning sickness, and so thousands of pregnant women took the drug to relieve their symptoms.
- 4. Birth defects In Europe and Canada during the early 1960s, thalidomide was prescribed to treat morning sickness in pregnant women. At that time, it was not approved in the United States because Dr. Frances Kelsey from the Food and Drug Administration (FDA) stated there was insufficient proof of the drug’s safety in humans. Thalidomide was soon banned worldwide when it was discovered that it caused tragic birth defects. According to the March of Dimes (MOD, November 1998), "More than 10,000 children around the world were born with major malformations, many missing arms and legs, because their mothers had taken the drug during early pregnancy. Mothers who had taken the drug when arms and legs were beginning to form had babies with a widely varying but recognizable pattern of limb deformities. The most well-known pattern, absence of most of the arm with the hands extending flipper-like from the shoulders, is called phocomelia. Another frequent arm malformation called radial aplasia was absence of the thumb and the adjoining bone in the lower arm. Similar limb malformations occurred in the lower extremities. The affected babies almost always had both sides affected and often had both the arms and the legs malformed. In addition to the limbs, the drug caused malformations of the eyes and ears, heart, genitals, kidneys, digestive tract (including the lips and mouth), and nervous system. Thalidomide was recognized as a powerful human teratogen (a drug or other agent that causes abnormal development in the embryo or fetus). Taking even a single dose of thalidomide during early pregnancy may cause major birth defects." 1962 photo of a baby born to a mother who had taken Thalidomide while pregnant; note the extra appendage connected to the foot and the malformation of the right arm.
- 5. UNITED STATES PRESCRIBING THALIDOMIDE On July 16, 1998, the FDA approved the use of thalidomide for the treatment of lesions associated with Erythema Nodosum Leprosum (ENL). Because of thalidomide’s potential for causing birth defects, the distribution of the drug was permitted only under tightly controlled conditions. The FDA required that Celgene Corporation, which planned to market thalidomide under the brand name Thalomid, establish a System for Thalidomide Education and Prescribing Safety (S.T.E.P.S.) oversight program. For safety the pregnancy teat women who were prescribed thalidomide.
- 6. United Kingdom`s view on prescribing thalidomide Thalidomide is available to only a small number of patients in the UK, generally in specialist cancer treatment centers, where research trials are taking place and specialist doctors have experience in its use.
- 7. BRAZIL PRESCRIBING THALIDOMIDE Brazil has the second highest prevalence rate of leprosy in the world and thalidomide has been used by Brazilian physicians as the drug of choice for the treatment of severe ENL since 1965. A study published in 1994 found 61 people born after 1965 whose limb defects and exposure history were compatible with thalidomide embryopathy. In 63.6% of these cases, thalidomide had been prescribed without the physician informing the patient about the drug's teratogenicity. Since then production, dispensing and prescription of thalidomide have been strictly controlled and cases of thalidomide embryopathy are thought to have occurred until today
- 8. Full list of indications currently being investigated in clinical trials Amyotrophic lateral sclerosis Aphthous ulcer Behcet's syndrome Brain cancer Breast cancer Cachexia Colorectal cancer Congestive heart failure Crohn's disease Diarrhea Fibrodysplasiaossificansprogressiva Graft-versus-host disease Haematological malignancy HIV infection Hodgkin's Diseases Kaposi's sarcoma Leukaemia Macular degeneration Malignant melanoma Mycobacterium avium complex infections Myelodysplastic syndrome Myelofibrosis Myeloid leukaemia Non-Hodgkin's lymphoma Non-small cell lung cancer Ovarian cancer Pain Prostate cancer Prurigonodularis Renal cancer Rheumatoid arthritis Small cell lung cancer Systemic lupus erythematosus Thyroid cancer Tuberculosis Thalidomide is still used today. Doctor`s are trying to see if the drug will help cure cancer and other illness. Thalidomide stopped a certain development in the womb, so they think that it might help stop cancer and other Illness in a certain stage of developement
- 9. Side affects of thalidomide Sleepiness Constipation skin rash severe headaches stomach aches peripheral neuropathy (numbness and pain in your arms, hands, legs and feet) Dizziness Nausea giddiness or nervousness at higher doses shivering and buzzing in the ears depression or mood-swings severe birth defects
- 10. Louise MedusMansell Louise MedusMansell is 48 years old and was born in 1962. Lousie fathers name is David Mason. Her mother was prescribed thalidomide during pregnancy. She was born with shorten arms and legs. Not long after birth Louise was placed in a home for disabled children in Kent. Her parents were told that Louise was only going to live for a few weeks. Louise first married in 1987 to John. They had two children together and they separated in 1994. She then remarried in August 2008 to Darren Mansell who is 49 years old. They dated as children and he is also a thalidomide victim.
- 11. Thomas Quasthoff Thomas Quasthoff was born November 9, 1959 in Hildesheim, Germany. He was born with serious birth defects due to his mother`s exposure to the drug thalidomide during pregnancy. She was prescribed thalidomide as an antiemetic to combat her morning sickness. Thomas is short due to shortening to the long bones in the legs and he had phocomelia of the upper extremities with very short or absent long bones and flipper-like appearance of his hands. He is four foot tall. Thomas is a musician and has been denied admission to the music conservatory.
- 12. David Lega David Lega was born in 1973 in Gothenburg, Sweden. He was born with AMC, a serious disability which means he was paralyzed in both arms and lacked muscles in his legs.
- 13. Mat Fraser Mat was born in 1962 in England, UK. Mat Fraser was born with phocomelia of both arms, due to his mother being prescribed thalidomide during her pregnancy
- 14. Terry Wiles Terrence 'Terry' Wiles was born in Peterborough, Cambridgeshire, England on January 12, 1962. Andrew (as he was originally named) was one of the most disabled thalidomide babies born in the UK. His mother had morning sickness so the Germany prescribed her thalidomide. This caused Terry to have birth defects. Terry as a child.
- 15. Niko von Glasow Niko von Glasow was born in 1960 in Cologne Germany. He is the son of Ernst Brücher and Majella Neven DuMont. He`s mother also took thalidomide During her pregnancy and it caused her son to have birth defects.
- 16. Dr. Kelsey Frances Kathleen Oldham Kelsey, Ph.D., M.D., (born 24 July 1914) is a Pharmacologist. most famous as the reviewer for the U.S. Food and Drug Administration (FDA) who refused to authorize thalidomide for market because she had concerns about the drug's safety. Her concerns proved to be justified when it was proven that thalidomide caused serious birth defects. Kelsey's career intersected with the passage of laws strengthening the FDA's oversight of Pharmaceuticals. In the 1960s, Kelsey prevented the marketing of thalidomide in the U.S. through her insistence on credible proof of its safety.
- 17. Resources http://www.aegis.com/factshts/network/simple/thalid.html http://www.aegis.com/factshts/network/simple/thalid.html http://cerhr.niehs.nih.gov/common/thalidomide.html http://www.dailymail.co.uk/news/article-561360/A-truly-special-love-story-Two-married-thalidomide-survivors-living-happily-50-years-drugs-launch.html http://www.davidlega.com/en/page.asp?pageid=2 http://en.wikipedia.org/wiki/Thalidomide http://en.wikipedia.org/wiki/Thomas_Quasthoff http://en.wikipedia.org/wiki/Terry_Wiles
