Intro Basic Chemistry
•Télécharger en tant que PPTX, PDF•
1 j'aime•1,159 vues
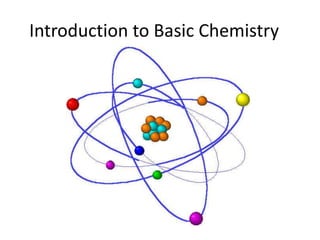
Matter is made up of atoms, which contain a nucleus of protons and neutrons surrounded by an electron cloud. Atoms are electrically neutral and bond with other atoms by sharing, losing, or gaining electrons in their outer valence shell. There are two main types of bonds: covalent bonds form when atoms share electrons, and ionic bonds form when one atom transfers an electron to another. Understanding the basic building blocks of matter and how atoms bond is fundamental to chemistry.
Signaler
Partager
Signaler
Partager
Recommandé
Contenu connexe
Tendances (20)
Similaire à Intro Basic Chemistry
Similaire à Intro Basic Chemistry (20)
chapter2-Atoms-Molecules-Ions.pp chemistry for pharmacyt

chapter2-Atoms-Molecules-Ions.pp chemistry for pharmacyt
Plus de Rachael Hubbard
Plus de Rachael Hubbard (20)
Dernier
USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...

USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...Postal Advocate Inc.
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️9953056974 Low Rate Call Girls In Saket, Delhi NCR
Dernier (20)
ENGLISH 7_Q4_LESSON 2_ Employing a Variety of Strategies for Effective Interp...

ENGLISH 7_Q4_LESSON 2_ Employing a Variety of Strategies for Effective Interp...
USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...

USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...
Global Lehigh Strategic Initiatives (without descriptions)

Global Lehigh Strategic Initiatives (without descriptions)
AUDIENCE THEORY -CULTIVATION THEORY - GERBNER.pptx

AUDIENCE THEORY -CULTIVATION THEORY - GERBNER.pptx
Procuring digital preservation CAN be quick and painless with our new dynamic...

Procuring digital preservation CAN be quick and painless with our new dynamic...
Inclusivity Essentials_ Creating Accessible Websites for Nonprofits .pdf

Inclusivity Essentials_ Creating Accessible Websites for Nonprofits .pdf
Barangay Council for the Protection of Children (BCPC) Orientation.pptx

Barangay Council for the Protection of Children (BCPC) Orientation.pptx
Virtual-Orientation-on-the-Administration-of-NATG12-NATG6-and-ELLNA.pdf

Virtual-Orientation-on-the-Administration-of-NATG12-NATG6-and-ELLNA.pdf
4.18.24 Movement Legacies, Reflection, and Review.pptx

4.18.24 Movement Legacies, Reflection, and Review.pptx
MULTIDISCIPLINRY NATURE OF THE ENVIRONMENTAL STUDIES.pptx

MULTIDISCIPLINRY NATURE OF THE ENVIRONMENTAL STUDIES.pptx
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️
Incoming and Outgoing Shipments in 3 STEPS Using Odoo 17

Incoming and Outgoing Shipments in 3 STEPS Using Odoo 17
Visit to a blind student's school🧑🦯🧑🦯(community medicine)

Visit to a blind student's school🧑🦯🧑🦯(community medicine)
How to do quick user assign in kanban in Odoo 17 ERP

How to do quick user assign in kanban in Odoo 17 ERP
Influencing policy (training slides from Fast Track Impact)

Influencing policy (training slides from Fast Track Impact)
Intro Basic Chemistry
- 1. Introduction to Basic Chemistry
- 2. I. Building Blocks of Matter: MATTER is made up of the smallest BASIC particle called an ATOM (matter is anything that has mass…pretty much everything around you)
- 4. ENERGY LEVEL or ELECTRON CLOUD: area outside the nucleus that contains the electrons
- 6. II. Properties of Atoms Atoms are electrically NEUTRAL Number of ELECTRONS is equal to the number of PROTONS
- 7. 2. In nature, atoms will BOND to other atoms to become stable ELECTRONS are part of why atoms bond together
- 8. There are 2 types of electrons CORE and VALENCE electrons: ***Valence Electrons: OUTSIDE electrons that are involved in bonding.
- 9. 3. BONDING: an attraction within atoms to form a chemical compound All atoms are electrically neutral but are UNSTABLE until they form an attraction with another atom The outermost energy level may SHARE, LOSE, or GAIN electrons to form a BOND and become stable
- 10. Two types of bonds found within atoms: 1. Covalent: Atoms share electrons (can be single, double, or triple)
- 11. Two types of bonds found within atoms: 2. Ionic: When one or more electrons is lost from one atom and transferred to another
