Combined sci c8 syllabus
•Télécharger en tant que PPT, PDF•
0 j'aime•424 vues
Combined Sci
Signaler
Partager
Signaler
Partager
Recommandé
Recommandé
Contenu connexe
Tendances
Tendances (20)
Chemistry prediction of products and workshop homework

Chemistry prediction of products and workshop homework
RATE OF REACTION (FAST & SLOW REACTION, AVERAGE RATE OF REACTION AND INSTATAN...

RATE OF REACTION (FAST & SLOW REACTION, AVERAGE RATE OF REACTION AND INSTATAN...
QUALITATIVE ANALYSIS (CONFIRMATORY TEST FOR CATIONS)

QUALITATIVE ANALYSIS (CONFIRMATORY TEST FOR CATIONS)
En vedette
En vedette (9)
Similaire à Combined sci c8 syllabus
Similaire à Combined sci c8 syllabus (20)
6.1 Role of water in showing acidic and alkaline properties (PPT)

6.1 Role of water in showing acidic and alkaline properties (PPT)
Routine analysis of wastewaters quality parameters

Routine analysis of wastewaters quality parameters
Plus de cartlidge
Plus de cartlidge (20)
Dernier
Mehran University Newsletter is a Quarterly Publication from Public Relations OfficeMehran University Newsletter Vol-X, Issue-I, 2024

Mehran University Newsletter Vol-X, Issue-I, 2024Mehran University of Engineering & Technology, Jamshoro
https://app.box.com/s/7hlvjxjalkrik7fb082xx3jk7xd7liz3TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...Nguyen Thanh Tu Collection
Dernier (20)
On National Teacher Day, meet the 2024-25 Kenan Fellows

On National Teacher Day, meet the 2024-25 Kenan Fellows
Asian American Pacific Islander Month DDSD 2024.pptx

Asian American Pacific Islander Month DDSD 2024.pptx
This PowerPoint helps students to consider the concept of infinity.

This PowerPoint helps students to consider the concept of infinity.
Jual Obat Aborsi Hongkong ( Asli No.1 ) 085657271886 Obat Penggugur Kandungan...

Jual Obat Aborsi Hongkong ( Asli No.1 ) 085657271886 Obat Penggugur Kandungan...
TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...
ICT role in 21st century education and it's challenges.

ICT role in 21st century education and it's challenges.
Seal of Good Local Governance (SGLG) 2024Final.pptx

Seal of Good Local Governance (SGLG) 2024Final.pptx
Combined sci c8 syllabus
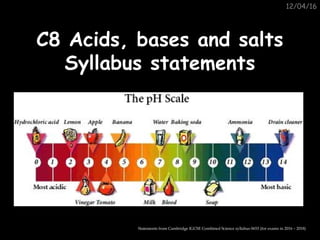
- 1. 12/04/16 C8 Acids, bases and saltsC8 Acids, bases and salts Syllabus statementsSyllabus statements Statements from Cambridge IGCSE Combined Science syllabus 0653 (for exams in 2016 – 2018)
- 2. 8.1 The characteristic8.1 The characteristic properties of acids & basesproperties of acids & bases 1 Describe neutrality and relative acidity and alkalinity in terms of pH (whole numbers only) measured using full-range indicator and litmus. 2 Describe the characteristic reactions between acids and metals, bases (including alkalis) and carbonates. 3 Describe & explain the importance of controlling acidity in the environment (air, water and soil).
- 3. 8.2 Preparation of salts8.2 Preparation of salts 1 Describe the preparation, separation and purification of salts using techniques selected from section C2.1 and the reactions specified in section C8.1. 2 Suggest a method of making a given salt from suitable starting materials, given appropriate information.
- 4. 8.3 Identification of ions8.3 Identification of ions and gasesand gases 1 Use the following tests to identify: Aqueous cations: ammonium, copper(II), iron(II), iron(III) and zinc by means of aqueous sodium hydroxide and aqueous ammonia as appropriate. (Formulae of complex ions are not required.)
- 5. 8.4 Identification of ions8.4 Identification of ions and gasesand gases 1 Use the following tests to identify: Anions: •carbonate by means of dilute acid and then limewater, •chloride by means of aqueous silver nitrate under acidic conditions, •nitrate by reduction with aluminium, •sulfate by means of aqueous barium ions under acidic conditions,
- 6. 8.4 Identification of ions8.4 Identification of ions and gasesand gases 1 Use the following tests to identify: Gases: •ammonia by means of damp red litmus paper; •carbon dioxide by means of limewater; •chlorine by means of damp litmus paper; •hydrogen by means of a lighted splint; •oxygen by means of a glowing splint.
- 7. 8.4 Identification of ions8.4 Identification of ions and gasesand gases 1 Use the following tests to identify: Gases: •ammonia by means of damp red litmus paper; •carbon dioxide by means of limewater; •chlorine by means of damp litmus paper; •hydrogen by means of a lighted splint; •oxygen by means of a glowing splint.