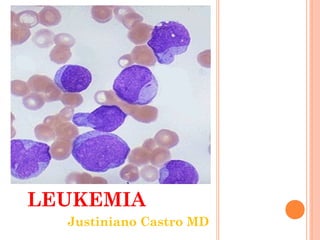
Diagnosing and Treating Acute Leukemia
- 2. BLAST CELL: DEFINITON Blast cells are immature cells found in the bone marrow. They are not fully developed, and therefore, do not carry out any normal function in the blood. More than 5% of these cells in the bone marrow is abnormal and need to be evaluated for the possibility of a malignant disorder of the hematopoietic system.
- 3. LEUKEMIA: DEFINITION Leukemia is a malignant clonal condition of the bone marrow and blood. It is characterized by the acumulation of blast cells in the marrow, usually more than 20%. These blast cells are frequently seen in the peripheral blood.
- 5. LYMPHOID VS. MYELOID CELLS Immature lymphoid cells have minimal differentiation Myeloid blast has some differentiation Cytoplasmic granules in myeloid cells (auer rod) Cytochemical stains (mpo and pas) Immunological markers, Ig and TCR
- 6. Figure 1. Arrow marks an Auer rod in this myeloid blast Maslak, P. ASH Image Bank 2004;2004:101101 Copyright ©2004 American Society of Hematology. Copyright restrictions may apply.
- 7. Figure 1. Blasts may appear "rounded" or "regular" with scant cytoplasm Maslak, P. ASH Image Bank 2005;2005:101390 Copyright ©2005 American Society of Hematology. Copyright restrictions may apply.
- 8. PATHOPHYSIOLOGY OF AL Leukemias typically fill up the marrow with abnormal cells, displacing normal hematopoiesis. The marrow here is essentially 100% cellular, but composed almost exclusively of leukemic cells. Normal hematopoiesis is reduced via replacement (a "myelophthisic" process) or by suppressed stem cell division. Thus, leukemic patients are prone to anemia, thrombocytopenia, and granulocytopenia and all of the complications that ensue, particularly complications of bleeding and infection. Bone marrow failure secondary to leukemic infiltration producing anemia, neutropenia and thrombocytopenia Maturation arrest and lineage infidelity Chromosomal abnormalities involving oncogenes and tumor suppressor genes
- 9. TECHNIQUE FOR ACUTE LEUKEMIA DIAGNOSIS Morphologic features Cytochemical stains Flow cytometry and markers Chromosomal analysis Fluorescence In Situ hybridization (FISH) PCR (polymerase chain reaction)
- 10. ACUTE LYMPHOBLASTIC LEUKEMIA This a clonal disorder of hematopietic cells There is accumulation of immature cells and tissue infiltration. Risk factors (chemicals, family history, radiation and chemotherapy)
- 11. Figure 2. A high power view reveals coarse chromatin with an irregular nuclear contour Maslak, P. ASH Image Bank 2007;2007:6-00053 Copyright ©2007 American Society of Hematology. Copyright restrictions may apply.
- 12. Figure 11. Blasts have scant cytoplasm Maslak, P. ASH Image Bank 2004;2004:101018 Copyright ©2004 American Society of Hematology. Copyright restrictions may apply.
- 13. ACUTE LYMPHOBLASTIC LEUKEMIA Accumulation of lymphoblast Most common in children and better prognosis than adults (3-7years) Disease in children is different than the adults Rise incidence after 40 years with a very poor prognosis
- 14. CLASSIFICATION: ALL B VS. T • Precursor B cell lymphoblastic leukemia/lymphoma, also called precursor B cell acute lymphoblastic leukemia (precursor B cell ALL) • Precursor T cell lymphoblastic leukemia/lymphoma (precursor T-LBL), also called precursor T cell acute lymphoblastic leukemia (precursor T cell ALL)
- 15. CLASSIFICATION OF ALL FAB (L1, L2 and L3) Immunological (B vs. T lineage) B cell 80% of all ALL. Tcell 20% of all ALL. B cell precursor ALL (80% ALL) CD10,CD19,CD20 and CD22) markers Early pre-B, CD10 neg (infant). Usually CD34+ Pre-B, CD10+ (common ALL) B-ALL (Burkitt’s type or L3, large vacuole and surface immunoglobulin). Its now consider a lymphoma T-cell ALL (TdT, CD3, CD5 and CD7)
- 16. DIAGNOSIS OF ALL Peripheral smear with blast cells Bone marrow with >20% of blasts Immunophenotype B vs. T cell, markers Help to differentiate from myeloid Chromosomal analysis (Hyper vs.hypodiploidy, t(9;22) or Ph+, t(4;11), t(v;11q23),t(12;21) and t(1;19))
- 17. SPECIAL TEST IN ALL Due to high incidence of CNS involvement a lumbar puncture is usually required and intrathecal treatment CT’s Scan for T cell and Burkitt Testicular us Tumor lysis syndrome
- 18. CYTOGENETICS IN ALL Philadelphia Chromosome present in 30% of adult ALL with a very poor prognosis. T(4;11) association with secondary leukemia T(v;11q23),t(12;21) and t(1;19) TEL and AML1 genes are present in 30% of childhood ALL and is associated with a good proognosis. Tyrosine Kinase inhibitors are included in treatment strategies
- 19. TREATMENT OF ALL Cytotoxic chemotherapy include induction, consolidation, intensification and maintenance Complete response after induction is 80% Cure 35-40% in the adult population T cell and L3 ALL require special consideration Response and cure in children could be as high as 90%
- 20. PROGNOSIS IN ALL Cure rate of 75-90% in children’s Cure rate in adults of 30% CD10 negative ALL has worse prognosis A presentation with a WBC > 30,000/m3 is associated with a poor prognosis Cytogenetics Slow response to induction is considered a poor prognosis feature
- 21. SUPPORTIVE CARE IN AL Blood products Hydration and treatment with allopurinol Prevention of N/V Aggressive antibiotic cover Central line catheters
- 22. SALVAGE THERAPY IN AL Bone marrow transplant Cord Blood Transplant Experimental treatment
- 24. CLINICAL OVERVIEW Acute myeloid leukemia (AML) is the most common acute leukemia in the adult AML is cancer of the blood-forming tissue (bone marrow). Normal bone marrow produces red cells, white cells, and platelets. AML causes bone marrow to produce too many immature white blood cells (blast cells) . Suppresses normal blood cell production. Anemia, leucopenia, thrombocytopenia
- 25. RISK FACTORS Age Older adults are more likely to develop AML Smoking 20% of AML cases are linked to smoking Doubles the risk of disease in people > 60 y/o Genetic disorders High doses of radiation Long-term survivors of atomic bombs Previous chemotherapy treatment Down syndrome, Fanconi’s anemia Breast cancer, ovarian cancer, lymphoma Exposure to industrial chemicals Benzene long term exposure
- 26. STATISTICS Incidence: 3-5 new cases expected per 100,000 population Mortality: 8,900 estimated deaths/year M:54%, F:46% Prognosis: 5-year survival rate in adults under 65 is 33% Prognosis: 5-year survival rate in adults over 65 is 4-5% Prognosis: 20-30% experience remission or are cured Sources: American Cancer Society; The Leukemia & Lymphoma Society,
- 27. CLINICAL FEATURES OF AML Bone marrow failure Coagulopathy (DIC) specially in APL Tumor infiltration of monocytic leukemia Tumor lysis syndrome specially with the initial chemotherapy
- 29. CLASSIFICATION BY FAB Mo Undifferentiated(5%) M1 no maturation(20%) M2 maturation(30%) M3 Promyelocytic (7%) M4 myelomonocytic M5 monocytic M6 Erythroleukemia (very rare) M7 Megakarioblast (very rare)
- 30. AML BLAST WITH AUER ROD
- 31. THE WHO CLASSIFICATION Reduce the percentage of blast to 20% Emphasis in the Cytogenetic characteristics Recognize therapy related leukemia's Recognize AML from previous Myelodyplasia
- 32. CYTOGENETIC IN AML Good prognostic group T(15;17) T(8;21) Inv (16) (p13q22) with eosinophilia Poor prognostic group Deletion of chromosome 5/7 t(11;q23) t(6;9) +8 Complex rearrangement
- 33. CYTOCHEMICAL STUDIES AND MARKERS MPO or Sudan black Nonspecific esterase Periodic Acid Shift (PAS) Markers CD13, CD33 an CD15 are only helpful for diagnosis of AML
- 34. AML – PEROXIDASE STAIN Blast with + MPD
- 35. Acute Myelogenous Leukemia M2 T(8;21) (q22;q22) RUNX1-RUNX1T1 (TEL and AML1 genes) Constitute 7% of AML Auer rods are easily identified and cytoplasm is generally basophilic Favorable prognosis in adults but not in children
- 36. Myeloid maturational arrest is demonstrated in this image, AML M2
- 38. ACUTE MYELOGENOUS LEUKEMIA M4 Approximately 20% of AML but 5% of them are AML M4 with eosinophilia and inv(16) Mixture of blast with myeloid and monocytic features Better prognosis with the inv(16) expression.
- 39. The dysphasic eosinophil precursors , AML M4 inv(16) eos
- 40. ACUTE PROMYELOCYTIC LEUKEMIA DIC very frequent t(15;17) is present in almost every case RAR gene present in chromosome 17 juxtaposition with PML gene in chrom 15 producing a maturation arrest All transretinoic acid (ATRA) induce maturation
- 41. AML M3 FAGGOT
- 42. Cells with multiple Auer rods (arrow) may be appreciated AML M3
- 43. ACUTE PROMYELOCYTIC LEUKEMIA Retinoid acid syndrome is associated with an increase in WBC counts with respiratory problems, fever and hypotension Arsenic trioxide is active in this leukemia Antracyclines are essential in the treatment
- 44. Proliferation: FLT3, Normal Cytogenetics A receptor tyrosine kinase expressed in 70 – 100% of AML cases. Activating mutations in FLT3 are seen in ~30% of AML cases. Tandem duplication of the juxtamembrane region. Point mutation within the activation loop of the kinase domain. Activation of FLT3 leads to deregulated proliferation of AML cells.
- 45. MANAGEMENT OF AML Supportive treatment similar to all AL AML M3 need special attention to DIC Monocytic leukemia's can infiltrate CNS, gingival and skin Induction chemotherapy produce 70% CR Usually 3 consolidation treatments Cure between 35 to 40% of the patients MUGA scan is necessary as one of the initial test for evaluation of cardiovascular function
- 46. SALVAGE TREATMENT IN AML Allogeneic BMT can cure high risk and relapse AML Monoclonal antibodies Experimental therapy
- 47. MYELODYSPLASTIC SYNDROME A heterogeneous group of hematopoietic disorders characterized by peripheral blood cytopenias and hypercellular marrow Clonal malignant disorder of the hematopoietic cells Transformation to AML is frequent
- 48. FAB CLASSIFICATION IN MDS Refractory Chronic anemia RA with ring sideroblast RA excess of blast myelomonocytic leukemia RA with excess of blast in transformation (RAEBT)
- 50. Figure 7. Refractory anemia with ringed sideroblasts (RARS) Vardiman, J. W ASH Image Bank 2001;2001:100189 Copyright ©2001 American Society of Hematology. Copyright restrictions may apply.
- 51. WHO CLASSIFICATION OF MDS RA RARS Refractory anemia with multilineage dysplasia (RCMD) RAEB 5q- syndrome Unclassifiable
- 52. Figure 9. Refractory cytopenia with multilineage dysplasia (RCMD) Vardiman, J. W ASH Image Bank 2001;2001:100188 Copyright ©2001 American Society of Hematology. Copyright restrictions may apply.
- 53. PROGNOSIS OF MDS Presence of blast indicate poor prognosis Uniformly fatal disorder due to infection and bleeding Chromosomal abnormalities are frequent including partial loss of chromosome 5, 7 and trisomy 8 5q- syndrome usually involve band q13 to q33 with anemia but no thrombocytopenia The International Prognostic Index ( % blasts cells, cytogenetic and number of cytopenias)
- 54. PATHOGENESIS OF MDS Toxic exposure and genetic predisposition Immune response Hypermethylation and angiogenesis RAS mutation as a late effect Transformation
- 55. TREATMENT OF MDS BY RISK STRATIFICATION Cytogenetics Blast % No. cytopenias Antiangiogenic Factor( Lenalidomi de and thalidomide) Hypomethylating agents Azacytidine and dezacitidine) Bone Marrow Transplantation
Notes de l'éditeur
- While EXACT Sciences’ technologies may someday be used to detect other common cancers, our first target is colorectal cancer. Colorectal cancer is prevalent, deadly and expensive. Importantly, though, colorectal cancer is treatable IF detected early. This is a CRITICAL part of our story. Genomics is essentially the operating system for the human body. Because the genomics underlying colorectal carcinogenesis is well-understood – as you will see- EXACT Sciences is able to use this knowledge to detect cancers early in their development.
