Tang 07 carboxylic acids, amines, & thiols 2015
•
1 j'aime•2,818 vues
carboxylic acids
Signaler
Partager
Signaler
Partager
Recommandé
Contenu connexe
Tendances
Tendances (19)
IUPAC Nomenclature_Pharmaceutical Organic Chemistry

IUPAC Nomenclature_Pharmaceutical Organic Chemistry
IUPAC NOMENCLATURE_ORGANIC_for JEE(MAIN)-JEE(ADVANCED)-NEET

IUPAC NOMENCLATURE_ORGANIC_for JEE(MAIN)-JEE(ADVANCED)-NEET
Organic Chemistry: Classification of Organic Compounds: Seminar

Organic Chemistry: Classification of Organic Compounds: Seminar
NOMENCLATURE OF ORGANIC COMPOUNDS BY -- KHUSH AHUJA

NOMENCLATURE OF ORGANIC COMPOUNDS BY -- KHUSH AHUJA
En vedette
En vedette (20)
Tang 02 supplement organic rules and point of first difference

Tang 02 supplement organic rules and point of first difference
Tang 10 substitution & elimination reactions 2

Tang 10 substitution & elimination reactions 2
formulation and development of anti-acne cream using garlic cloves project pr...

formulation and development of anti-acne cream using garlic cloves project pr...
Similaire à Tang 07 carboxylic acids, amines, & thiols 2015
Similaire à Tang 07 carboxylic acids, amines, & thiols 2015 (20)
IUPAC Nomenclature of Organic Compounds Part-2.pptx

IUPAC Nomenclature of Organic Compounds Part-2.pptx
Classification, Nomenclature and structural isomerism of organic compound 

Classification, Nomenclature and structural isomerism of organic compound
Organic Introduction - Saturated Compounds and Naming

Organic Introduction - Saturated Compounds and Naming
Plus de mrtangextrahelp
Plus de mrtangextrahelp (20)
Dernier
Fertilization: Sperm and the egg—collectively called the gametes—fuse togethe...

Fertilization: Sperm and the egg—collectively called the gametes—fuse togethe...D. B. S. College Kanpur
THE ROLE OF PHARMACOGNOSY IN TRADITIONAL AND MODERN SYSTEM OF MEDICINE.pptx

THE ROLE OF PHARMACOGNOSY IN TRADITIONAL AND MODERN SYSTEM OF MEDICINE.pptxNandakishor Bhaurao Deshmukh
Dernier (20)
GenAI talk for Young at Wageningen University & Research (WUR) March 2024

GenAI talk for Young at Wageningen University & Research (WUR) March 2024
Fertilization: Sperm and the egg—collectively called the gametes—fuse togethe...

Fertilization: Sperm and the egg—collectively called the gametes—fuse togethe...
Vision and reflection on Mining Software Repositories research in 2024

Vision and reflection on Mining Software Repositories research in 2024
STOPPED FLOW METHOD & APPLICATION MURUGAVENI B.pptx

STOPPED FLOW METHOD & APPLICATION MURUGAVENI B.pptx
LIGHT-PHENOMENA-BY-CABUALDIONALDOPANOGANCADIENTE-CONDEZA (1).pptx

LIGHT-PHENOMENA-BY-CABUALDIONALDOPANOGANCADIENTE-CONDEZA (1).pptx
Pests of soyabean_Binomics_IdentificationDr.UPR.pdf

Pests of soyabean_Binomics_IdentificationDr.UPR.pdf
GenBio2 - Lesson 1 - Introduction to Genetics.pptx

GenBio2 - Lesson 1 - Introduction to Genetics.pptx
Observational constraints on mergers creating magnetism in massive stars

Observational constraints on mergers creating magnetism in massive stars
Thermodynamics ,types of system,formulae ,gibbs free energy .pptx

Thermodynamics ,types of system,formulae ,gibbs free energy .pptx
Best Call Girls In Sector 29 Gurgaon❤️8860477959 EscorTs Service In 24/7 Delh...

Best Call Girls In Sector 29 Gurgaon❤️8860477959 EscorTs Service In 24/7 Delh...
Pests of castor_Binomics_Identification_Dr.UPR.pdf

Pests of castor_Binomics_Identification_Dr.UPR.pdf
GENERAL PHYSICS 2 REFRACTION OF LIGHT SENIOR HIGH SCHOOL GENPHYS2.pptx

GENERAL PHYSICS 2 REFRACTION OF LIGHT SENIOR HIGH SCHOOL GENPHYS2.pptx
THE ROLE OF PHARMACOGNOSY IN TRADITIONAL AND MODERN SYSTEM OF MEDICINE.pptx

THE ROLE OF PHARMACOGNOSY IN TRADITIONAL AND MODERN SYSTEM OF MEDICINE.pptx
Base editing, prime editing, Cas13 & RNA editing and organelle base editing

Base editing, prime editing, Cas13 & RNA editing and organelle base editing
Topic 9- General Principles of International Law.pptx

Topic 9- General Principles of International Law.pptx
Tang 07 carboxylic acids, amines, & thiols 2015
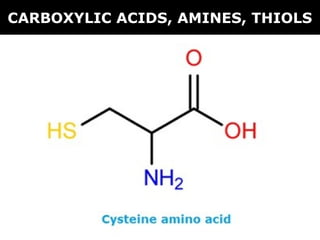
- 1. CARBOXYLIC ACIDS, AMINES, THIOLS
- 3. IUPAC naming system A carboxylic acid contains a –COOH functional group (carboxyl). This group must be included in the main C- chain that is named as carbon #1. CARBOXYLIC ACIDS
- 4. The “e” of –ane, –ene and –yne endings is replaced with –oic acid. HCOOH CH3COOH CH3CH2COOH methanoic acid ethanoic acid propanoic acid CARBOXYLIC ACIDS
- 5. Common naming system The prefix used with aldehydes may also be used with carboxylic acids. In this case an –ic acid is the suffix. HCOOH CH3COOH CH3CH2COOH formic acid acetic acid proprionic acid CARBOXYLIC ACIDS
- 6. Example #1Example #1 a) What is the structural formula for propenoic acid? b) What is the IUPAC name for the following acid? CH2-CH-COOH CH3 CH3 OH O 2-methylbutanoic acid CARBOXYLIC ACIDS
- 7. Properties of Carboxylic AcidsProperties of Carboxylic Acids Carboxylic acids have higher boiling points than their corresponding hydrocarbon. Carboxylic acids have similar solubility properties as their corresponding alcohol. All carboxylic acids have acidic properties. CARBOXYLIC ACIDS
- 8. AMINES
- 9. All amines are essentially derived from NH3. Depending on the number of carbon side- chains off of the N, we can form different types of amines. primary amine secondary amine tertiary amine AMINES
- 10. IUPAC naming system This system adds the suffix “amine” to the end of the name. butan-1-amine propan-2-amine H2N H2N AMINES
- 11. IUPAC naming system When there is more than one amine group present, keep the “e” ending of the C-chain root. butane-1,2-diamine hexane-2,3,4-triamine AMINES H2N NH2 NH2 NH2 NH2
- 12. IUPAC naming system If there are carbon side-chains off of the N- group, it is denoted by an “N-” prefix. N-methylbutan-1-amine N,N-dimethylmethananime N H NH3C CH3 CH3 AMINES
- 13. IUPAC naming system The N-group is treated as a side-chain and is added as a prefix on the C-chain root if a group with a higher priority is present. AMINES NH2 OH 3-aminobutan-2-ol 5-amino-3-hydroxy-2-oxohexanal O OH O NH2
- 14. Example #2Example #2 Write the IUPAC name for the following molecule. CH3 – N – CH3 CH3CHCH3 N,N-dimethylpropan-2-amine AMINES
- 15. Example #3Example #3 Draw a 1°, 2° and 3° amine which each contain a total of three carbons. NH3C CH3 CH3 NCH3CH2 H CH3 NCH3CH2CH2 H H primary secondary tertiary AMINES
- 16. Properties of AminesProperties of Amines Amines have higher boiling points than their hydrocarbon counterparts. Smaller amines are more soluble in water than larger amines. For mp and bp of amines of the same size, 3° < 2° < 1° In general, amines are basic in nature. AMINES
- 17. THIOLS
- 18. THIOLS Properties of thiolsProperties of thiols Contains the sulfhydryl group (-SH) Strong odours
- 19. IUPAC naming system Suffix ‘thiol’ is added to the end of the hydrocarbon group Example: THIOLS methanethiol