IB Chemistry Electrolytic Cells Guide
•Télécharger en tant que PPTX, PDF•
0 j'aime•1,712 vues
This document discusses electrolytic cells, which use an electric current to drive non-spontaneous redox reactions called electrolysis. In electrolytic cells, the cathode is the negative electrode where reduction occurs and the anode is the positive electrode where oxidation occurs, unlike in voltaic cells. Electrolytic cells are used for processes like metal extraction and refining, electroplating, and producing industrial gases. They involve immersing inert electrodes in a common electrolyte, with the cathode being the negative terminal of the power supply where positive ions are reduced.
Signaler
Partager
Signaler
Partager
Contenu connexe
Tendances
Tendances (20)
04(t) thermal and chemical effects of electric current

04(t) thermal and chemical effects of electric current
En vedette
En vedette (6)
2016 topic 0 - oxidation and reduction (INTRODUCTION)

2016 topic 0 - oxidation and reduction (INTRODUCTION)
Similaire à IB Chemistry Electrolytic Cells Guide
Similaire à IB Chemistry Electrolytic Cells Guide (20)
Plus de David Young
Plus de David Young (20)
Dernier
Dernier (20)
SQL Database Design For Developers at php[tek] 2024![SQL Database Design For Developers at php[tek] 2024](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![SQL Database Design For Developers at php[tek] 2024](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
SQL Database Design For Developers at php[tek] 2024
Kalyanpur ) Call Girls in Lucknow Finest Escorts Service 🍸 8923113531 🎰 Avail...

Kalyanpur ) Call Girls in Lucknow Finest Escorts Service 🍸 8923113531 🎰 Avail...
Handwritten Text Recognition for manuscripts and early printed texts

Handwritten Text Recognition for manuscripts and early printed texts
IAC 2024 - IA Fast Track to Search Focused AI Solutions

IAC 2024 - IA Fast Track to Search Focused AI Solutions
Presentation on how to chat with PDF using ChatGPT code interpreter

Presentation on how to chat with PDF using ChatGPT code interpreter
Injustice - Developers Among Us (SciFiDevCon 2024)

Injustice - Developers Among Us (SciFiDevCon 2024)
The 7 Things I Know About Cyber Security After 25 Years | April 2024

The 7 Things I Know About Cyber Security After 25 Years | April 2024
The Role of Taxonomy and Ontology in Semantic Layers - Heather Hedden.pdf

The Role of Taxonomy and Ontology in Semantic Layers - Heather Hedden.pdf
08448380779 Call Girls In Diplomatic Enclave Women Seeking Men

08448380779 Call Girls In Diplomatic Enclave Women Seeking Men
Enhancing Worker Digital Experience: A Hands-on Workshop for Partners

Enhancing Worker Digital Experience: A Hands-on Workshop for Partners
WhatsApp 9892124323 ✓Call Girls In Kalyan ( Mumbai ) secure service

WhatsApp 9892124323 ✓Call Girls In Kalyan ( Mumbai ) secure service
Unblocking The Main Thread Solving ANRs and Frozen Frames

Unblocking The Main Thread Solving ANRs and Frozen Frames
08448380779 Call Girls In Greater Kailash - I Women Seeking Men

08448380779 Call Girls In Greater Kailash - I Women Seeking Men
IB Chemistry Electrolytic Cells Guide
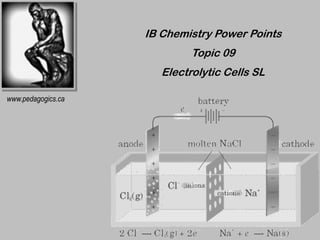
- 1. IB Chemistry Power Points Topic 09 Electrolytic Cells SL www.pedagogics.ca
- 2. Electrolytic Cells an electric current is used to drive a non-spontaneous redox reaction This process is called electrolysis
- 3. Electric current enters and exits the cell by electrodes Electrolytic cells often use inert electrodes (electrodes that don’t react). Like voltaic cells: 1. Cathode: - electrode at which reduction occurs 2. Anode: - electrode at which oxidation occurs Unlike voltaic cells: cathode is (-) & anode is (+) Electrodes are immersed in a common electrolyte.
- 4. Cathode is the negative terminal of the power supply This is the site of reduction of +ive ions in electrolyte Negative ions are oxidized at the anode. Electrolytic cells are used for extraction, refining, electroplating, commercial manufacture of gases, etc.
- 5. Predicting electrolysis products Molten Salt Electrolytes REQUIRES – molten salt and inert electrodes ALWAYS – metal cation reduced at cathode, nonmetal anion oxidized at anode. Write the cathode, anode and overall rxns for this example Pb2+ (l) + 2e- Pb (l) 2Br- (l) Br2 (g) + 2e- Pb2+ (l) + 2Br- (l) Pb (l) + Br2 (g)
