SRAA y Ateroesclerosis: Inhibición del SRAA reduce estrés oxidativo y progresión de la ateroesclerosis
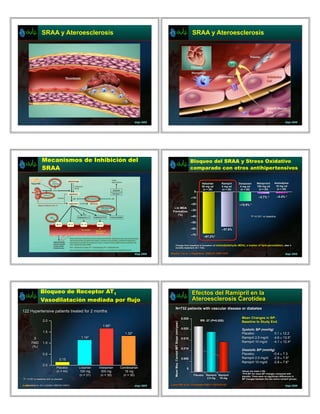
- 1. SRAA y Ateroesclerosis SRAA y Ateroesclerosis drpp 2009 drpp 2009 Mecanismos de Inhibición del Bloqueo del SRAA y Stress Oxidativo SRAA comparado con otros antihipertensivos Valsartan Ramipril Doxazosin Metoprolol Amlodipine 80 mg od 5 mg od 4 mg od 100 mg od 10 mg od (n = 20) (n = 20) (n = 20) (n = 22) (n = 20) 0 –10 –2.7% * –2.4% * –20 –15.9% * Aldosterone ! in MDA blockers Formation –30 (%) –40 *P <0.001 vs baseline. –50 –60 –57.6% –70 –67.2%* Change from baseline in formation of malondialdehyde (MDA), a marker of lipid peroxidation, after 3 months treatment (N = 102). drpp 2009 Baykal Y et al. J Hypertens. 2003;21:1207-1211. drpp 2009 Bloqueo de Receptor AT1 Efectos del Ramipril en la Vasodilatación mediada por flujo Ateroesclerosis Carotídea N=732 patients with vascular disease or diabetes 122 Hypertensive patients treated for 2 months 0.025 Mean Changes in BP: 2.0 RR -37 (P=0.028) Baseline to Study End Mean Max. Carotid IMT Slope (mm/year) 1.66* 0.020 Systolic BP (mmHg) 1.5 1.32* Placebo: 0.1 ± 12.2 ! 1.14* 0.015 Ramipril 2.5 mg/d: -4.6 ± 13.5* FMD 1.0 Ramipril 10 mg/d: -4.1 ± 12.4* (%) 0.010 Diastolic BP (mmHg) 0.5 Placebo: -0.4 ± 7.3 0.15 0.005 Ramipril 2.5 mg/d: -2.9 ± 7.9* Ramipril 10 mg/d: -2.8 ± 7.6* 0.0 Placebo Losartan Irbesartan Candesartan 0 (n = 44) 100 mg 300 mg 16 mg Values are mean ± SD. *P<0.001 for mean BP changes compared with (n = 31) (n = 30) (n = 30) Placebo Ramipril Ramipril placebo. There were no significant differences in *P < 0.05 vs baseline and vs placebo 2.5 mg 10 mg BP changes between the two active ramipril groups. Referencia al. Am J Cardiol. 2004;93:1432-5. Koh KK et drpp 2009 Lonn EM, et al. Circulation 2001; 103:919–25. drpp 2009
- 2. Val-PREST: Re-estenosis intra-Stent Mortalidad in SAVE, y Reintervención a 6 meses TRACE, AIRE, y VALIANT Valsartan 80 mg (n = 99) Hazard Ratio for Mortality Placebo (n = 101) 50 SAVE 50% reduction with valsartan vs placebo 40 58% reduction TRACE with valsartan vs placebo Restenosis (%) Patients with 30 AIRE * 20 * Combined 10 VALIANT (imputed placebo) 0 0.5 1 2 Restenosis Reintervention *P <0.005 vs placebo. Favors Favors Active Drug Placebo Adapted with permission from Peters S et al. J Invasive Cardiol. 2001;13:93-97. drpp 2009 Pfeffer, McMurray, Velazquez, et al. N Engl J Med 2003;349 drpp 2009 MICRO-HOPE: IECAs Mejoran Metas CV y Renales en Diabetes N=3577 patients with diabetes (SRAA) Sistema Overt CV nephro- Renina-Angiotensina- MI Stroke death pathy Mean albumin: 0 creatinine ratio 3.0 Placebo –10 2.5 Aldosterona: Risk 2.0 Reduction –20 P=0.02 (%) 1.5 Ramipril 10 mg 1.0 Efectos Renales –30 P=0.001 P=0.01 P=0.027 0.5 –40 0 1 2 3 4.5 P=0.007 Years P=0.0001 drpp 2009 HOPE Study Investigators. Lancet. 2000;355:253–259. drpp 2009 (SRAA) Sistema Renina-Angiotensina- Aldosterona: Diabetes N Engl J Med. www.mechanismsincardiology.com 2001;345:851-860. drpp 2009 drpp 2009
- 3. Bloqueo del SRAA y metabolismo de Bloqueo del SRAA: Mejoría del glucosa: Insulina y mediadores Metabolismo de glucosa BK NO Angiotensin I Bradykinin BK2 + Receptor "NO Glucose ACE/Kininase II Transport Akt1 Degradation + Angiotensin II Insulin products Receptor + + + GLUT-4 Insulin IRS-1 PI3-K + Trans- location SRAA Blockage - GLUT-4 Biosynthesis $Angiotensin II "Bradykinin - GLUT-4 AT1 Receptor !Skeletal muscle "Nitric oxide blood flow Ang II !Glucose metabolism Adapted from Henriksen EJ, Jacob S. J Cell Physiol. 2003;196:171–9. drpp 2009 Henriksen EJ, Jacob S. J Cell Physiol. 2003;196:171–9. drpp 2009 Efecto de los IECA’s en Diabetes Metas de PA diastólica: Estudio HOPE Estudio HOT Reducción relativa del riesgo con Ramipril vs Placebo en pacientes con Diabetes 30 -2.1 mmHg = -2.1 mmHg = CV Events /1,000 Patient-Years in Patients with Diabetes at Baseline - 5.8 events/1000 py - 6.7 events/1000 py 25 22% de Infarto de Miocardio p = 0.01 20 33% de EVC p = 0.0074 15 37% de muerte cardiovascular p = 0.0001 24% de nefropatía p = 0.027 10 17% de revascularización p = 0.031 5 20% de Insuficiencia Cardíaca p = 0.019 0 Target: # 90 mmHg DBP # 85 mmHg DBP #80 mmHg DBP (Achieved): (mean 85.2 mmHg) (mean 83.2 mmHg) (mean 81.1 mmHg) p for trend = 0.005 HOPE investigators. Lancet 2000;355:253-259. drpp 2009 Referencia Adapted from Hansson et al. Lancet 1998;351(9118):1755-1762. drpp 2009 Antihipertensivos: Prevención de Nuevos Casos de Sensibilidad a la Insulina Diabetes tipo 2 Mejor Nuevo Mejor convencional Estudio Seguimiento RR (IC 95%) 18 20 CAPPP (IECA vs Diu-BB) 6,1 16 0,86 (0,74-0,99); p= 0,03 12 HOPE (IECA vs Placebo)* 5,0 0,66 (0,51-0,85); p= 0,001 Cambio (%) 10 NORDIL (CA vs Diu-BB) 4,5 0,87 (0,73-1,04); p= 0,14 0 INVEST (CA vs BB) 2,7 0,85 (0,77-0,95); p= 0,01 -4 ALLHAT (IECA vs Diu) 4,6 -10 0,77 (0,66-0,89); p= 0,001 -15 LIFE (ARA II vs BB) 4,8 -22 -18 0,75 (0,63-0,88); p= 0,001 -30 -27 -20 CHARM (ARA II vs Placebo) 3,1 0,78 (0,64-0,96); p= 0,01 PRO MET -30 VALUE (ARA II vs CA) 4,2 0,77 (0,69-0,86); p= 0,0001 ATEN HCTZ NIF VER DOXA -40 0,50 0,75 1 1,25 IECA ARA II drpp 2009 drpp 2009
- 4. ARA II en Nefropatía Diabética Referencia drpp 2009 Referencia drpp 2009