10.2 - First law of Thermodynamics and PV graphs
•Télécharger en tant que PPTX, PDF•
1 j'aime•10,381 vues
Signaler
Partager
Signaler
Partager
Contenu connexe
Tendances
Tendances (20)
En vedette
En vedette (20)
Thermodynamic Chapter 3 First Law Of Thermodynamics

Thermodynamic Chapter 3 First Law Of Thermodynamics
Application of first law thermodynamics (yoga n zian)

Application of first law thermodynamics (yoga n zian)
5.2 - Internal Resistance, Power & Combining Resistors

5.2 - Internal Resistance, Power & Combining Resistors
Similaire à 10.2 - First law of Thermodynamics and PV graphs
Similaire à 10.2 - First law of Thermodynamics and PV graphs (20)
Heat and thermodynamics - Preliminary / Dr. Mathivanan Velumani

Heat and thermodynamics - Preliminary / Dr. Mathivanan Velumani
Plus de simonandisa
Plus de simonandisa (18)
Dernier
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️9953056974 Low Rate Call Girls In Saket, Delhi NCR
Model Call Girl in Tilak Nagar Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Tilak Nagar Delhi reach out to us at 🔝9953056974🔝9953056974 Low Rate Call Girls In Saket, Delhi NCR
HỌC TỐT TIẾNG ANH 11 THEO CHƯƠNG TRÌNH GLOBAL SUCCESS ĐÁP ÁN CHI TIẾT - CẢ NĂ...

HỌC TỐT TIẾNG ANH 11 THEO CHƯƠNG TRÌNH GLOBAL SUCCESS ĐÁP ÁN CHI TIẾT - CẢ NĂ...Nguyen Thanh Tu Collection
YOUVE GOT EMAIL_FINALS_EL_DORADO_2024.pptx

YOUVE GOT EMAIL_FINALS_EL_DORADO_2024.pptxConquiztadors- the Quiz Society of Sri Venkateswara College
Dernier (20)
ISYU TUNGKOL SA SEKSWLADIDA (ISSUE ABOUT SEXUALITY

ISYU TUNGKOL SA SEKSWLADIDA (ISSUE ABOUT SEXUALITY
Influencing policy (training slides from Fast Track Impact)

Influencing policy (training slides from Fast Track Impact)
ENGLISH 7_Q4_LESSON 2_ Employing a Variety of Strategies for Effective Interp...

ENGLISH 7_Q4_LESSON 2_ Employing a Variety of Strategies for Effective Interp...
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️
Model Call Girl in Tilak Nagar Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Tilak Nagar Delhi reach out to us at 🔝9953056974🔝
Incoming and Outgoing Shipments in 3 STEPS Using Odoo 17

Incoming and Outgoing Shipments in 3 STEPS Using Odoo 17
HỌC TỐT TIẾNG ANH 11 THEO CHƯƠNG TRÌNH GLOBAL SUCCESS ĐÁP ÁN CHI TIẾT - CẢ NĂ...

HỌC TỐT TIẾNG ANH 11 THEO CHƯƠNG TRÌNH GLOBAL SUCCESS ĐÁP ÁN CHI TIẾT - CẢ NĂ...
10.2 - First law of Thermodynamics and PV graphs
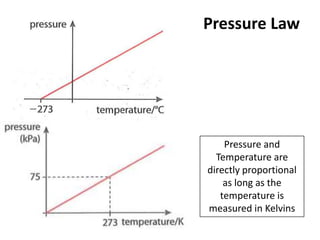
- 1. Pressure Law Pressure and Temperature are directly proportional as long as the temperature is measured in Kelvins
- 2. Boyle’s Law Each line represents an experiment done at a different temperature
- 4. Pistons Wd = P.A.Δd Wd = P. ΔV
- 5. Wd = P. ΔV As long as there is an AREA under the line then you can see that work is done (and calculate it)
- 6. First Law of Thermodynamics It just says that if you add Heat to a gas it must be equal to the Work Done by the gas + any change in Internal Energy This is mainly difficult because: 1. Heat can be gained or lost 2. Internal Energy can rise or fall 3. Work can be done ‘on the gas’ or ‘by the gas’
- 7. 1. Isobaric Constant Pressure 1. The gas is expanding – doing Work (Work is +ve) 2. The temperature is increasing so ΔU is increasing (ΔU is +ve) 3. ΔQ = ΔU + W so Q must be +ve 4. Heat must have been added
- 8. 2. Isovolumetric / Isochoric Constant Volume 1. The gas isn’t expanding or contracting. No Work is done. ΔW=0 2. Because the Pressure is increasing the temperature must be rising so ΔU is +ve 3. Q = ΔU + W so Q = ΔU so heat is +ve 4. Heat must have been added
- 9. 3. Isothermic Constant Temperature 1. The gas is expanding – doing Work (Work is +ve) 2. The temperature is constant so ΔU is 0 3. Q = ΔU + W so Q =W 4. Heat must have been added to enable the gas to do work
- 10. 4. Adiabatic No Heat is being exchanged – perfectly insulated. 1. No heat is exchanged so Q = 0 2. Volume is reducing so Work is being done ON THE GAS (W= -ve) 3. Q = ΔU + W so 0 = ΔU - W 4. ΔU = W so work done on the gas increases the Internal energy of the gas Adiabatic lines are always steeper than Isotherms
- 11. A Heat Cycle A-B Isochoric / Isovolumetric Temperature rise B-C Isobaric Expansion C-D Isochoric / Isovolumetric Temperature drop D-A Isobaric Contraction
- 13. The Carnot Cycle
- 15. Reverse Carnot Cycle D