Membrane transport1
- 1. The Cell as the Fundamental Unit of Life: Membrane Transport Processes
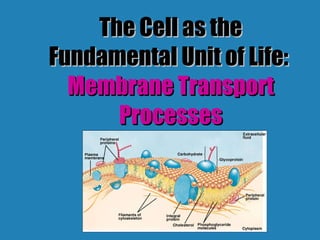
- 2. Selectively permeable- allows certain substances to pass through By 2 ways: active or passive transport Passive - downhill Active - uphill (needs energy) Plasma Membrane Function:
- 4. No Barrier: Substances “spread out” High concentration to low concentration e.g.: Red dye placed in glass of water Passive Diffusion
- 5. Substances diffuse High concentration to low concentration Pores in membrane must be large “ Down the concentration gradient” Dynamic equilibrium, equal rates in both directions Passive Diffusion Biological membrane:
- 8. Passive Diffusion: Terms & Osmosis More concentrated to less concentrated Until concentration same on both sides: isotonic
- 9. Passive Diffusion: Osmosis &Terms Concentration of solute less: solution is hypotonic . Concentration of solute greater: solution is hypertonic .
- 10. Passive Diffusion: Osmosis: Outcomes to Living Cells (Animal) Animal cells No cell walls Isotonic environment: Influx of water equals the efflux of water No change in cell shape
- 11. Passive Diffusion: Osmosis: Outcomes to Living Cells (Animal) Hypertonic solution: Water leaves cell Shriveled, or crenate Hypotonic solution: Water enters cell Bursts, or lyses
- 13. Passive Transport and Facilitated Diffusion Passive transport & facilitated diffusion do NOT require ATP
- 14. Active Transport DOES require the input of Transport proteins AGAINST concentration gradient outside cell inside cell ATP
- 15. ATP
- 16. Role of ATP in Energy Metabolism ATP ADP + P i + Energy
- 17. Endocytosis
- 18. Exocytosis
- 19. Secretion
- 21. Membrane Permeability Cell membrane: selectively permeable 4 factors that determine permeability lipid solubility molecular size polarity charge 1 2 3 4
- 22. Lipid solubility Most important factor Hydrophobic molecules Passively diffuse Hydrocarbons, carbon dioxide, & oxygen
- 23. Molecular Size and Polarity Larger molecules, less permeable Lower kinetic energy Small pore sizes in the membrane Polar molecules hydrophilic, less permeable Very small, polar uncharged (water) molecules can diffuse Molecular Size Polarity - +
- 24. Charge Charged molecules hydrophilic, less permeable Surrounded by coat of water (hydration shell), increases the size