Whitepaper cardiology-1
- 1. 1
Trendsmeme™ | White Paper
Cardiology
What’s Next for the Shrinking
Cardiovascular Drug Market?
The Multibillion-Dollar Cardiovascular Drug Market Takes a Hit
The World Health Organization (WHO) classifies cardiovascular disease as the leading cause
of death globally. The cardiovascular drug market is likewise among the top therapeutic
categories in terms of overall prescription drug sales. Leading contributors to this market are
several multibillion-dollar drugs for hyperlipidemia (high cholesterol), hypertension, and heart
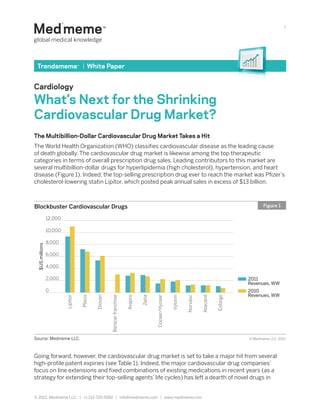
disease (Figure 1). Indeed, the top-selling prescription drug ever to reach the market was Pfizer’s
cholesterol-lowering statin Lipitor, which posted peak annual sales in excess of $13 billion.
Blockbuster Cardiovascular Drugs Figure 1
12,000
10,000
8,000
$US millions
6,000
4,000
2,000 2011
Revenues, WW
0 2010
Revenues, WW
Lipitor
Plavix
Diovan
Benicar franchise
Avapro
Zetia
Cozaar/Hyzaar
Vytorin
Norvasc
Atacand
Exforge
Source: Medmeme LLC. © Medmeme LLC. 2012
Going forward, however, the cardiovascular drug market is set to take a major hit from several
high-profile patent expiries (see Table 1). Indeed, the major cardiovascular drug companies’
focus on line extensions and fixed combinations of existing medications in recent years (as a
strategy for extending their top-selling agents’ life cycles) has left a dearth of novel drugs in
© 2012, Medmeme LLC. | +1 212-725-5992 | info@medmeme.com | www.medmeme.com
- 2. 2
Trendsmeme™ | White Paper
the pipeline. Table 2 shows some of the leading CV drug franchises; apart from these, few new
cardiovascular drugs have reached the market over the past decade.
Patent Expiries for Leading Cardiovascular Drugs Table 1
Product Patent Expiry
Diovan Sept, 2012
Atacand June, 2012
Plavix May, 2012
Avapro March, 2012
Lipitor Nov, 2011
Caduet Nov, 2012
Cozaar/Hyzaar Feb, 2010
Source: Medmeme LLC. © Medmeme LLC. 2012
Leading Cardiovascular Drug Franchises Table 2
Brand Name Generic Name Indications U.S. Launch
Year
Novartis Diovan valsartan Hypertension, HF, post-MI 1996
Diovan HCT valsartan-HCT Hypertension, HF, post-MI 1998
Exforge amlodipine- Hypertension 2007
valsartan
Exforge HCT amlodipine- Hypertension 2009
valsartan-HCT
Tekturna/Rasilez Aliskiren Hypertension 2007
Tekturna HCT/ aliskiren-HCT Hypertension 2008
Rasilez HCT
Valturna/Rasival* aliskiren-valsartan Hypertension 2009*
Tekamlo/Rasilamlo aliskiren-amlodipine Hypertension 2010
Amturnide/Rasitrio aliskiren- Hypertension 2010
amlodipine-HCT
(Continued)
© 2012, Medmeme LLC. | +1 212-725-5992 | info@medmeme.com | www.medmeme.com
- 3. 3
Trendsmeme™ | White Paper
Leading Cardiovascular Drug Franchises (Continued) Table 2
Brand Name Generic Name Indications U.S. Launch
Year
Pfizer Norvasc amlodipine Hypertension, angina 1992
Lipitor atorvastatin Hyperlipidemia, prevention of CV 1996
disease
Caduet amlodipine- Hypertension, CAD, 2004
atorvastatin hyperlipidemia, prevention of CV
disease, Fredrickson Types IIa, IIb,
III, and IV
Merck Zocor simvastatin Hyperlipidemia, reductions in risk 1991
of CHD mortality and CV events
Cozaar Losartan Hypertension, hypertension 1995
with LVH, nephropathy in Type 2
diabetics
Hyzaar losartan-HCT Hypertension, hypertension with 1995
LVH
Zetia ezetimibe Hyperlipidemia (monotherapy 2002
and combination therapy), HoFH,
homozygous sitosterolemia
Vytorin ezetimibe- Hyperlipidemia, HoFH 2004
simvastatin
Daiichi-Sankyo Benicar Olmesartan Hypertension 2002
medoxomil
Benicar HCT Olmesartan Hypertension 2003
medoxomil
Azor Amlodipine- Hypertension 2007
olmesartan
medoxomil
Tribenzor Amlodipine- Hypertension 2010
olmesartan
medoxomil-HCT
*Novartis has agreed to voluntarily withdraw Valturna/Rasival from the U.S. and Swiss markets for safety reasons (Rasival never reached
the broader EU markets).
Source: Medmeme LLC. © Medmeme LLC. 2012
© 2012, Medmeme LLC. | +1 212-725-5992 | info@medmeme.com | www.medmeme.com
- 4. 4
Trendsmeme™ | White Paper
What Pipeline Drugs Could Sustain the Market?
There are several drugs with novel mechanisms in late-stage trials for cardiovascular disease,
including renin inhibitors for hypertension, CETP inhibitors for hyperlipidemia, and a variety of
approaches for acute heart failure (AHF). Below is a snapshot of the broader discussion provided
in the Medmeme report titled Trendsmeme™ Report: Cardiology.
Renin Inhibitors. Based on results from the ALTITUDE trial—which was halted by Novartis in
late 2011 based on findings of increased adverse events (risk of stroke and renal complications)
and no apparent benefits among type 2 diabetes patients taking the company’s renin inhibitor
aliskiren (Tekturna/Rasilez) on top of an ACE inhibitor or ARB—Novartis withdrew its follow-
on Valturna (aliskiren-valsartan) from the market and added a warning to aliskiren’s label
advising against use with ACE inhibitors or ARBs in patients with diabetes and/or moderate
renal impairment. It is not yet clear how these findings will affect sales of aliskiren-containing
products (Tekturna, Tekturna-HCT, Tekamlo, Amturnide), but Novartis could potentially take a
big hit, especially since the company was banking on the aliskiren product franchise to soften
the blow from Diovan’s patent expiry in September of this year (Diovan sales exceeded $5.6
billion in 2011). Moreover, renin inhibitors as a class are now under a cloud of doubt, leaving the
hypertension market again relying on fixed combinations of older agents (e.g., CCBs, ARBs).
CETP Inhibitors. One new class of cholesterol-lowering drugs—the cholesteryl ester transfer
protein (CETP) inhibitors—has shown some promise as add-on therapy to statins. Their future,
however, remains up in the air after some high-profile Phase III failures including Pfizer’s
torcetrapib and Roche’s more recent discontinuation of dalcetrapib. At present, both Merck and
Lilly are investigating their own CETP inhibitors in late-stage trials for dyslipidemia (anacetrapib
and evacetrapib, respectively).
New Treatments for Acute Heart Failure. There are several investigational therapies in mid-to-late
stage clinical development for AHF that represent novel approaches to treatment, making this an
area to watch over the next few years.
• Novartis is investigating RLX 030, a drug that mimics human relaxin-2, in Phase III trials for
AHF.
• Cytogenetics and Amgen have an IV formulation of the cardiac-specific myosin activator
omecamtiv mecarbil in Phase IIb trials for AHF.
• Cardiorentis is investigating its synthesized form of urodilatin called ularitide in Phase III trials
for AHF.
© 2012, Medmeme LLC. | +1 212-725-5992 | info@medmeme.com | www.medmeme.com
- 5. 5
About Medmeme
Medmeme, LLC, is a global database company that offers comprehensive knowledge about
the people, institutions, and research that drive medical practice in all major therapeutic areas.
Founded in 2006, Medmeme serves pharmaceutical and biotechnology clients worldwide,
including 16 of the leading 20 pharmaceutical companies.
Our proprietary database of thought leaders, medical meetings, clinical trials, and Centers-of-
Excellence provides intelligence that allows clients to reach the most important channels through
which medical information travels. Detailed profiles of medical science experts, including their
medical contributions, levels of influence, and professional networks, allow clients to strengthen
product messaging by enlisting thought leaders whose opinions increasingly shape medical
practice. In addition, by identifying effective research practices, investigators, and sites, our
products help clients improve clinical trial effectiveness while reducing costs.
Our Approach
Medmeme’s approach is designed to help clients meet the multifaceted challenges facing the
healthcare industry, including demand for specialty medical treatments with better outcomes,
the high costs of developing these treatments, and pressure from patients and payers to keep
treatment costs under control.
Finding a better balance among these challenges requires gathering more focused and detailed medical
knowledge. To achieve this goal, Medmeme has developed a unique system of finding, validating, and
analyzing data using proprietary algorithms and platforms that deliver in-depth information across
drugs and disease areas in 50 countries. The quality, breadth, and detail of our data set us apart:
• Credible - We use only the most reliable sources, such as governments, medical societies, and
accredited medical institutions
• Accurate - Medmeme processes data using proprietary algorithms, first to normalize it to standardize
fields for easy comparison, followed by a disambiguation process that prevents duplication. Data is
then integrated across database platforms to provide a comprehensive view of each therapeutic area.
• Comprehensive - We aggregate 50 million data points from more than 60,000 sources
worldwide. Ten-year historical records encompass clinical trials, meetings, publications, grants,
patents, treatment guidelines, and more.
• Forward-looking - Updated daily, our database includes information on emerging as well as
developed markets, and on promising as well as established thought leaders.
Flexible Solutions
Medmeme can offer database solutions based on client-specified needs, including segmentation
by disease, therapeutic area, compound, drug class, development phase, country/region, keyword,
and more. Client-specified search parameters provide targeted information to support functions
across the organization.
Medmeme offers annual subscriptions to our proprietary database platforms. We also provide syndicated
reports for those who want data compiled by therapeutic area. Discounts for longer subscriptions
and multiple-product purchases make it economical to acquire greater quantities of information.
© 2012, Medmeme LLC. | +1 212-725-5992 | info@medmeme.com | www.medmeme.com
- 6. 6
About Medmeme
About Medmeme Syndicated Reports
Medmeme reports slice the data by therapeutic categories from more than 60,000 URLs yielding
50 million data points, the professional bodies of work of over 1.5 million key opinion leaders, more
than 6.3 million medical conference presentations, over 600,000 clinical trials in 50 countries, and
information from governments, societies, and institutions to map, measure, and project the pathways
of future market movement. Medmeme’s data-intensive reports compile and analyze trends over
time to show the top conferences, the top journals, the top contributors, the top institutions, and
more, to illustrate—using easily navigated text and extensive tables and graphs—the most successful
approaches to creating awareness, interest, and demand in any therapeutic category.
• Trendsmeme™ Reports – These reports cover key indications, companies, and drugs to reveal
dominant player positioning as well as relevant issues and developments in treatment and the
marketplace. They explain the status of a therapeutic category and the major trends that are
driving change and influencing R&D success.
• Intellmeme™ Reports – Reports that identify the disease areas and drugs that receive the most
attention—that is, the largest share-of-voice—from peer-reviewed medical journals, societies,
meetings and congresses, and key opinion leaders. They illustrate trends over time and highlight
strategic options.
• C-O-E/meme™ Reports – These reports pinpoint the top 50 medical centers-of-excellence by
specific therapeutic categories, giving guidance for selection of partners to conduct clinical
research, recruit KOLs for specific disease expertise and affiliations, provide educational
initiatives, and more. The reports are a direct derivative of Medmeme’s on-line, continuously
updated, searchable database product platform C-O-E/meme™.
• Trialmeme™ Reports – The reports identify, by location, leading clinical trial sites worldwide,
including principal and site investigators, trial status, entity conducting the trial, drug, and where the
results have been presented. This knowledge can help reduce both trial timelines and costs of future
trials by providing historic perspective to sponsors and CROs. The reports are a direct derivative of
Medmeme’s on-line, continuously updated, searchable database product platform Trialmeme™.
These syndicated reports can be purchased separately, based on therapeutic category of interest,
or by series at discounted pricing. To learn more about the reports, see www.medmeme.com/
products/syndicated-reports.
To purchase Reports or for more information, contact
Ray Wright, Vice President, Sales Yan Barshay, Executive Vice President Mahesh Naithani, CEO
+1 508-278-4595 +1 212-725-5992 +1 212-725-5990
rwright@medmeme.com yan@medmeme.com mahesh@medmeme.com
© 2012, Medmeme LLC. | +1 212-725-5992 | info@medmeme.com | www.medmeme.com