The Parts of an Atom: Electrons, Nucleus, Protons, Neutrons, Energy Levels
•
1 j'aime•1,181 vues
An atom consists of a dense nucleus containing protons and neutrons surrounded by a cloud of electrons. The nucleus contains protons which have a positive charge and neutrons which have a neutral charge. Electrons orbit the nucleus and have a negative charge. The atomic number represents the number of protons in an atom's nucleus, while the mass number is the total number of protons and neutrons.
Signaler
Partager
Signaler
Partager
Télécharger pour lire hors ligne
Recommandé
Recommandé
Contenu connexe
Tendances
Tendances (20)
Similaire à The Parts of an Atom: Electrons, Nucleus, Protons, Neutrons, Energy Levels
Similaire à The Parts of an Atom: Electrons, Nucleus, Protons, Neutrons, Energy Levels (20)
The atom is a basic unit of matter consisting of .pdf

The atom is a basic unit of matter consisting of .pdf
Plus de MrsKendall
Plus de MrsKendall (20)
Dernier
BÀI TẬP BỔ TRỢ TIẾNG ANH 11 THEO ĐƠN VỊ BÀI HỌC - CẢ NĂM - CÓ FILE NGHE (GLOB...

BÀI TẬP BỔ TRỢ TIẾNG ANH 11 THEO ĐƠN VỊ BÀI HỌC - CẢ NĂM - CÓ FILE NGHE (GLOB...Nguyen Thanh Tu Collection
BÀI TẬP BỔ TRỢ TIẾNG ANH 8 - I-LEARN SMART WORLD - CẢ NĂM - CÓ FILE NGHE (BẢN...

BÀI TẬP BỔ TRỢ TIẾNG ANH 8 - I-LEARN SMART WORLD - CẢ NĂM - CÓ FILE NGHE (BẢN...Nguyen Thanh Tu Collection
Mattingly "AI & Prompt Design" - Introduction to Machine Learning"

Mattingly "AI & Prompt Design" - Introduction to Machine Learning"National Information Standards Organization (NISO)
Mattingly "AI & Prompt Design: Large Language Models"

Mattingly "AI & Prompt Design: Large Language Models"National Information Standards Organization (NISO)
Plagiarism,forms,understand about plagiarism,avoid plagiarism,key significanc...

Plagiarism,forms,understand about plagiarism,avoid plagiarism,key significanc...Nigar Kadar Mujawar,Womens College of Pharmacy,Peth Vadgaon,Kolhapur,416112
Dernier (20)
BÀI TẬP BỔ TRỢ TIẾNG ANH 11 THEO ĐƠN VỊ BÀI HỌC - CẢ NĂM - CÓ FILE NGHE (GLOB...

BÀI TẬP BỔ TRỢ TIẾNG ANH 11 THEO ĐƠN VỊ BÀI HỌC - CẢ NĂM - CÓ FILE NGHE (GLOB...
Employablity presentation and Future Career Plan.pptx

Employablity presentation and Future Career Plan.pptx
Oppenheimer Film Discussion for Philosophy and Film

Oppenheimer Film Discussion for Philosophy and Film
Decoding the Tweet _ Practical Criticism in the Age of Hashtag.pptx

Decoding the Tweet _ Practical Criticism in the Age of Hashtag.pptx
BÀI TẬP BỔ TRỢ TIẾNG ANH 8 - I-LEARN SMART WORLD - CẢ NĂM - CÓ FILE NGHE (BẢN...

BÀI TẬP BỔ TRỢ TIẾNG ANH 8 - I-LEARN SMART WORLD - CẢ NĂM - CÓ FILE NGHE (BẢN...
INCLUSIVE EDUCATION PRACTICES FOR TEACHERS AND TRAINERS.pptx

INCLUSIVE EDUCATION PRACTICES FOR TEACHERS AND TRAINERS.pptx
Mattingly "AI & Prompt Design" - Introduction to Machine Learning"

Mattingly "AI & Prompt Design" - Introduction to Machine Learning"
Satirical Depths - A Study of Gabriel Okara's Poem - 'You Laughed and Laughed...

Satirical Depths - A Study of Gabriel Okara's Poem - 'You Laughed and Laughed...
prashanth updated resume 2024 for Teaching Profession

prashanth updated resume 2024 for Teaching Profession
Mattingly "AI & Prompt Design: Large Language Models"

Mattingly "AI & Prompt Design: Large Language Models"
Q-Factor General Quiz-7th April 2024, Quiz Club NITW

Q-Factor General Quiz-7th April 2024, Quiz Club NITW
Plagiarism,forms,understand about plagiarism,avoid plagiarism,key significanc...

Plagiarism,forms,understand about plagiarism,avoid plagiarism,key significanc...
The Parts of an Atom: Electrons, Nucleus, Protons, Neutrons, Energy Levels
- 1. The parts of an Atom
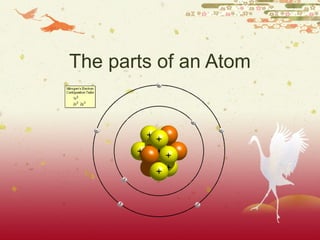
- 2. The atom An Atom: The most basic unit of matter. An atom has a dense nucleus and a cloud of negatively charged electrons around it. If matter is what everything is made of, and matter is made of atoms… Then, everything is really made of atoms!
- 3. An Electron Electron: A subatomic particle with a negative electrical charge. An electron’s mass is so small that it is generally not even considered.
- 4. A Nucleus Nucleus: The dense center of an atom containing protons and neutrons. Nucleus
- 5. A Proton A Proton: A subatomic particle within the nucleus of an atom that has a positive charge.
- 6. Neutron A Neutron: A subatomic particle within the nucleus of an atom that has a neutral charge.
- 7. Energy Level The energy level of an atom is as follows: 2,8,8 The first shell has two electrons The second shell has 8 electrons The third shell has 8 electrons MEMORIZE: 2,8,8!!!!!!!!!!
- 8. Atomic Number The atomic number is the number mainly associated with the element and represents the number of protons present in the atom.
- 9. Mass Number The total number of protons and neutrons. The mass of an atom comes from the protons and neutrons. Therefore, the mass includes the masses of each added together. This number can be found Mass Number at the bottom, typically.
- 10. Atomic Mass Sub atomic particles are measured in AMU’s. AMU stands for Atomic Mass Unit. Every proton has a mass of 1 AMU Every Neutron has a mass of 1 AMU Remember, electrons don’t have a measureable mass.
- 11. Element A pure chemical substance consisting of ONE type of atom.