More Related Content
Similar to Liang_Shiochee_ArestyPoster_FINAL
Similar to Liang_Shiochee_ArestyPoster_FINAL (20)
Liang_Shiochee_ArestyPoster_FINAL
- 1. RESEARCH POSTER PRESENTATION DESIGN © 2012
www.PosterPresentations.com
The proper development of the neocortex is dependent upon the precise specification of
neuronal subtypes. This specification relies on the integration of both intrinsic and extrinsic
signals to output functionally different cell types. However, molecular and cellular mechanisms
behind this specification are still poorly understood. Here I show that the timed arrival of
trophic factor, Neurotrophin-3 (NT3), determines neocortical dendritic development. In
particular, I show that the arrival of thalamic NT3 into the developing neocortex is critical for
dendritic outgrowth. These data suggest that timed thalamocortical axon ingrowth determines
neocortical circuit formation.
ABSTRACT
INTRODUCTION
Figure 1. Axons extending from the thalamus into the neocortex
The six layers are formed in an inside-out matter in which the subcortically projecting neurons of
the lower layers are formed first and the intracoritcally projecting neurons are formed later. In
mice, the radial glia start producing the neurons of the lower layers (Layers V and VI) around
embryonic day 11 (E11). Around E15, the radial glia stop producing lower layer neurons are start
producing upper layer neurons (Layers II – IV). By E17, the final upper layer neurons are made
and the radial glia then produce glia. While the neocortex is developing, axons from the
thalamus can influence the development of the neocortex by secreting proteins called trophic
factors. NT-3 is one of those trophic factors secreted by the Crh+ nucleus of the thalamus. The
thalamocortical axons approach the neocortex roughly the point where the radial glia switches
from lower layer neurogenic state to a upper layer neurogenic state as seen in Figure 2. Prior
data from this laboratory suggests these axons extrinsically influence this neurogenesis.
RESULTS
CONCLUSIONS
1Divison of Life Sciences, Rutgers, the State University of New Jersey, 604 Allison Road Piscataway, NJ 08854
2Department of Neuroscience and Cell Biology, Rutgers-Robert Wood Johnson Medical School, 675 Hoes Lane, Piscataway, NJ 08854
Shiochee Liang1,2, Erik DeBoer2, Matthew L. Kraushar2, HR Sagara Wijeratne2, Mladen-Roko Rasin2
The Decreased Expression of NT-3 Results in Decreased Dendritic Outgrowth
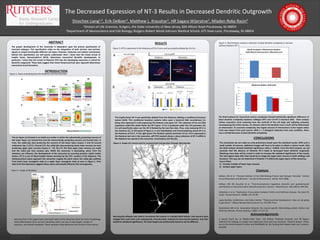
Figure 3. NT3 is expressed in the thalamus of E14.5 mice and successfully ablated by Crh-Cre.
The trophic factor NT-3 was specifically ablated from the thalamus utilizing a conditional knockout
system (A/B). This conditional knockout systems relies upon a bacterial DNA recombinase Cre
being only expressed in cells expressing the thalamic-only gene Crh. The substrate of Cre are DNA
sequences called flox which flanks the NT-3 gene. If Cre is produced under the Crh promoter, the
Cre will specifically splice out the NT-3 flanked by the two flox sites. Thus, only removing NT-3 in
the thalamus (C). In left panel of Figure 3, in situ hybridzation and immunostaining shows NT-3 in
the thalamus at E14.5. In the right panel, the thalamic specific promoter of Cre, Crh is expressed in
the thalamus but not in the neocortex. qRT-PCR analysis shows a sharp reduction of NT-3 mRNA in
the thalamus but spared in the neocortex, chroid plexus (Ch Plx) (D).
Figure 4. Image of a neuron and its reconstructed tracing done through Neurolucida.
Neurolucida software was used to reconstruct the neuron in a double blind fashion. Five neurons were
imaged from each brain and subsequently reconstructed, analyzed by Neurolucida Explorer, and then
tested for statistical significance. The total length was preliminarily found to not be different.
Figure 5. Sholl Analysis reveals a reduction in basal dendrite complexity in neurons
without thalamic NT-3
The Sholl analysis for neocortical neuron complexity showed statistically significant difference of
basal dendrite complexity between wildtype (WT) and Crh-NT-3 knockout (KO). Sholl analysis
utilizes concentric circle centering from the centroid of the cell body and radiating outwards
encompassing the entire neuron. An instance where the dendrite cross a circle will be determined
an intersection. To ascertain complexity, the largest amount of intersections at the largest radius
circle was chosen from each neuron. With n = 2 biological replicates from each condition, there
was a marked decrease in basal dendrite complexity.
The conclusions we can make are only preliminary based upon the statistical results. With such a
small number of neurons, additional images will have to be taken to obtain a clearer result. Only
the Sholl analysis showed statistical significance with p = 0.0018. From the Sholl analysis, we can
conclude that the absence of thalamic NT-3 leads to decreased basal dendritic outgrowth.
Obtaining more data will be crucial in order to increase the statistical significance of the results.
The next logical steps after that would be to image the upper layer neurons in both wildtype and
knockout. This way, we can determine if thalamic T-3 affects the upper layers of the neocortex.
Future Plans:
1) Increase number of lower layer neurons
2) Analyze upper layers
Citations
DeBoer, EM et al. "Prenatal Deletion of the RNA-Binding Protein HuD Disrupts Postnatal Cortical
Circuit Maturation and Behavior." Journal of Neuroscience. (2014): 3674-3686.
DeBoer, EM, ML Kraushar et al. "Post-transcriptional regulatory elements and spatiotemporal
specification of neocortical stem cells and projection neurons." Neuroscience. 248. (2013): 499-528.
DeStefano A et al. “Replication of association between ELAVL4 and Parkinson disease: the Gene PD
study.” Human Genetics. (2008): 124: 95-99.
Lopez-Bendito, Guillermina, and Zoltan Molnar. "Thalamocortical Development: How are we going
to get there?." Nature Reviews Neuroscience 4 (): 276-289. Print.
Noureddine MA et al. Association between the neuron-specific RNA-binding protein ELAVL4 and
Parkinson disease. Human Genetics. (2005): 117:27-33.
A special thank you to Mladen-Roko Rasin, Erik DeBoer, Matthew Krausher, and HR Sagara
Wijeratne for helping me design and implement these past two semester’ research project. Thank
you to the Aresty Research Center and NIH/NINDS for the funding that helped make such research
possible.
Acknowledgements
Neurons from in the upper layers and lower layers of the neocortex share the same morphology.
What differentiates them are their dendrites, which can vary in total length, number of
branches, and overall complexity. These variations help determine the function of the neuron.
Figure 2. Image of dendrites
A
B
C
D