Approach to sle
- 1. Approach to SLE By: Tasneem Bashir IM resident
- 2. Case presentation • Nahid is a 29 years old woman, married with 1 kid. She works in Banking Assistant in Oman. She completed Business degree at university. • In April 2015 she presented to a Suliman Habib hospital in Dubai with the following: Gradual joint pain affecting small joints of hands (MCPs, PIPs, wrists, knees, feet) Associated swelling in some of these joints No AM stiffness, no nocturnal pain • Based on this history of polyarthritis (MCPs, PIPs, wrists, knees, feet) since April 2015, a/w swelling in some of these joints, ANA 1:5120 (homogenous pattern), dsDNA +ve 1:160, RNP +ve, anti-SM +ve, +ve anti-histone, SS-A +ve, low C3 & C4. • She was diagnosed as SLE & kept on one of the DMARDs until September 2015
- 3. September 2015 = went to India • - +ve ANA, +ve dsDNA (ELISA), anti-Sm equivocal, anti-SSA +ve, anti-Scl 70. • - WBC 3.7, Lymphocytes 43% (NR 20-40%), PMNs 54% (NR 45 - 85), • - Negative tests = anti-CCP, RF, syphilis • - Normal tests = iron, TIBC, TSH, FBG, creatinine (0.6 mg/dL), lipid profile, LFT, ESR 18 • - CRP 7 mg/L (NR < 6) • - Blood film = mild microcytic anemia, target cells, no polychromasia. normal platelets & WBC. • - Normal urinalysis excpet albumin +, normal microscope. • - Imaging: - CXR = normal. • - 99m Tc MDP whole body bone scan = increased tracer uptake in bilateral shoulders, bilateral SI joints, dorso-lumbar vertebrae, hips, knees • - US abdomen = normal. • - Trans-thoracic Echo = normal esp. no pulm HTN. • Dx = undifferentiated CTD. • Given xx drugs.
- 4. Jan 2016 --> went to 2 hospitals in Thailand: 15-16 Jan 2016 = hospital # 1. • History: polyarthritis (hands, shoulders, knees, feet) of 10 months duration, back pain, ? uveitis 10 months ago (she did not provide the doctor with a report about uveitis), hair loss, oral ulcers, rash over dorsum of left hand • Physical exam: synovitis of left 4th MCP, bilateral MTPs, ankles, small knees effusion, Schober's tests = 10 to 14 cm. Red skin patch with thick white scale on left hand, hyper-pigmented patches on both ear pinna, diffuse thin hair, ? pitting nails of some fingernails. • - Xrays: • - SI joints = normal except subchondral sclerosis on iliac sides of both SI joints • - lumbo-sacral spine = decreased height at anterior aspect of T12 & L1. Subchondral sclerosis at both SI joints • - hands = normal. • - Feet = bilateral symmetric narrowing of 2-5 PIPs. • - CXR = normal. • - rheumatologist diagnosed her with psoriatic arthritis + possible SLE • - Rheum explained to her that she needed to follow up with a local rheumatologist because this is a chronic disease.
- 5. 18-19 Jan 2016 = hospital # 2 • History: polyarthritis, AM stiffness, hair loss, oral ulcers, rash over dorsum of left hand, left leg edema • Investigations: ANA 1:5120 (homogenous pattern), dsDNA +ve 1:160, RNP +ve, anti-SM +ve, +ve anti-histone, Negative anti-ribosoma P protein, negative CEN B • - Hb 85, low WBC 2.48, normal neutrophils %, normal lymphocytes %, +ve direct Coomb's test • - High triglycerides 248 mg/dL, high CRP 19.9, ESR 54 • - Normal LFT, uric acid, TSH • - Negative Hep C. Immune against Hep B • - normal Creatinine 0.7 mg/dL & BUN 9 mg/dL, • - urinalysis = trace protein, WBC 5-10, RBC 2-3, urine protein / Cr 0.44 • - low C3 = 31.9 mg/dL (NR 90 - 180), low C4 = 7.34 mg/dL (NR 10 - 40), low CH50 < 10 U/mL (NR 20-40). • - Dx: SLE with hemolytic anemia + lupus nephritis class 2 --> induction Rx = Cellcept 1g bid + Pred 30 mg PO OD + Omeprazole 20mg OD + Plaquenil 200mg OD + Cholecalciferol 20,000 units weekly + Ca+ 600mg OD
- 6. With our rheumatologist: • Seen first on 28 Feb 2016, when she was diagnosed as SLE. • Received plaquenil & Cellcept as prescribed before. • kept on Cellcept (MMF) upto 13 March 2016 when she felt unsure if this is the right drug for her now, given no severe end-organ damage. • Started on Imuran, on 28 April 2016.
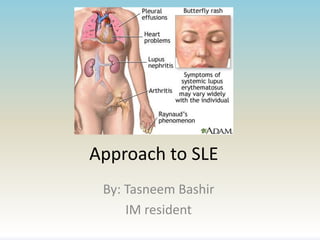
- 7. Introduction to SLE • Systemic lupus erythematosus is a chronic inflammatory disease of unknown cause that can affect virtually every organ. Immunologic abnormalities, particularly the production of a number of antinuclear antibodies, are another prominent feature of the disease. • More than 90% of cases of SLE occur in women, frequently starting at childbearing age. The annual incidence of SLE averages 5 cases per 100,000 population. • The most common pattern is a mixture of constitutional complaints with skin, musculoskeletal, mild hematologic, and serologic involvement.
- 8. • The clinical course of SLE is highly variable among patients and is marked by remissions and relapses and may vary from mild to severe. • Women are affected more frequently than men.
- 10. Constitutional symptoms • Fatigue & fever are present in most lupus patients at some point during the course of the disease. • Myalgia is also common among patients with SLE • Weight changes are frequent in patients with SLE and may be related to the disease or to its treatment. Weight loss often occurs prior to the diagnosis of SLE. - Unintentional weight loss may be due to decreased appetite, side effects of medications (particularly diuretics), and gastrointestinal disease (eg, GERD, abdominal pain, peptic ulcer disease) - Weight gain in SLE may be due to salt and water retention associated with hypoalbuminemia (nephrotic syndrome).
- 11. Arthritis and arthralgias • Occur in over 90 percent of patients with SLE and are often one of the earliest manifestations. Arthritis, with inflammation, occurs in 65-70% of patients and tends to be migratory, polyarticular, and symmetrical. The arthritis is moderately painful, usually does not cause erosion, and rarely deforming.
- 12. Skin and mucous membrane involvement • Acute cutaneous lupus erythema (ACLE) AKA “the butterfly rash” presents as erythema in a malar distribution over the cheeks and nose (but sparing the nasolabial folds), which appears after sun exposure. • Some patients may develop discoid lesions, which are more inflammatory and which have a tendency to scar. Associaed with scarring alopecia. • Many patients develop oral and/or nasal ulcers, which are usually painless.
- 13. Vascular disease • Raynaud phenomenon: occurs in 50% of SLE pts. • Vasculitis: The most frequent type is cutaneous small vessel vasculitis which can manifest as palpable purpura, petechiae, papulonodular lesions, livedo reticularis, panniculitis, splinter hemorrhages, and superficial ulcerations. In one study 11% of patients with vasculitis had visceral involvement (eg, peripheral nerves, lung, pancreas, and kidney) the remainder had cutaneous manifestaitions.
- 14. Thromboembolic disease • It can complicate SLE, particularly in the context of antiphospholipid antibodies. Although the precise mechanism is unknown, thromboembolic disease can affect both the venous and arterial circulations
- 15. Renal involvement • Occur in ~50% of SLE patients, causes significant morbidity & mortality. • Periodic screening for lupus nephritis: [urinalyses, quantitation of proteinuria, and eGFR] is an important component of the ongoing management of SLE patients. Several forms of glomerulonephritis can occur, and renal biopsy is useful to define the type and extent of renal involvement. • Lupus nephritis can present as asymptomatic hematuria and/or proteinuria to nephrotic syndrome and rapidly progressive GN with loss of renal function. Some patients with lupus nephritis also have hypertension.
- 16. GI involvement • GI symptoms occurs in up to 40% of patients. The majority of gastrointestinal symptoms are caused by adverse medication reactions and viral or bacterial infections. SLE-related gastrointestinal abnormalities can involve almost any organ along the gastrointestinal tract and include esophagitis, intestinal pseudo-obstruction, protein-losing enteropathy, lupus hepatitis, acute pancreatitis, mesenteric vasculitis or ischemia, and peritonitis
- 17. Pulmonary involvement • Pulmonary manifestations of SLE include pleuritis (with or without effusion), pneumonitis, interstitial lung disease, pulmonary hypertension, shrinking lung syndrome, and alveolar hemorrhage. Respiratory symptoms must also be distinguished from infection, particularly in patients on immunosuppressive therapy. The risk of thromboembolic involvement is increased in those with antiphospholipid antibodies or with lupus anticoagulant.
- 18. Cardiac disease • Can involve the pericardium, myocardium, valves, conduction system, and coronary arteries. Pericarditis, with or without an effusion, is the most common cardiac manifestation of SLE occurring in approximately 25% of patents at some point during their disease course. Verrucous (Libman-Sacks) endocarditis is usually clinically silent, but it can produce valvular insufficiency and can serve as a source of emboli. Myocarditis is uncommon but may be severe. Patients with SLE also have an increased risk of coronary artery disease.
- 19. • Ophthalmologic involvement: KCS, vasculitis, retinopathy. • Neuropsychiatric involvement: cognitive dysfunction, organic brain syndromes, delirium, psychosis, seizures, headache, and/or peripheral neuropathies. • Hematologic abnormalities: Anemia (Hemolytic anemia), leukopenia in 50%, mild thrombocytopenia. • Hepatosplenomegally / lymphadenopathy.
- 20. Clinical Approach • The initial evaluation requires a careful history and physical exam, along with selected laboratory testing to identify features that are characteristic of SLE or that suggest an alternative diagnosis.
- 21. Lab investigations: • CBC w differential may reveal leukopenia, mild anemia, and/or thrombocytopenia • Elevated serum creatinine may be suggestive of renal dysfunction • Urinalysis with urine sediment may reveal hematuria, pyuria, proteinuria, and/or cellular casts In addition to the routine laboratories described above, we perform the following laboratory tests which support the diagnosis of SLE if abnormal: • ANA (+ve in virtually all SLE patients) If the ANA is positive, one should test for other specific antibodies such as dsDNA, anti-Sm, Ro/SSA, La/SSB, and U1 ribonucleoprotein (RNP). • Antiphospholipid antibodies (lupus anticoagulant [LA], IgG and IgM anticardiolipin [aCL] antibodies; and IgG and IgM anti-beta2-glycoprotein [GP] I) • C3 and C4 or CH50 complement levels • Erythrocyte sedimentation rate (ESR) and/or C-reactive protein (CRP) levels • Urine protein-to-creatinine ratio
- 22. Diagnostic imaging not routinely done • Plain radiographs of swollen joints. Unlike affected joints in RA, erosions are observed infrequently in SLE. Depending on the stage of disease, deformities may be present on radiograph. • Renal ultrasonography to assess kidney size and to rule out urinary tract obstruction when there is evidence of renal impairment • Chest radiography (eg, for suspected pleural effusion, interstitial lung disease, cardiomegaly). • Echocardiography (eg, for suspected pericardial involvement, to assess for a source of emboli, or noninvasive estimation of pulmonary artery pressure; and for evaluation of suspected valvular lesions, such as verrucae). • CT: (eg, for abdominal pain, suspected pancreatitis, interstitial lung disease). • MRI: (eg, for focal neurologic deficits or cognitive dysfunction).
- 23. SLICC criteria for classification of SLE (4 of 17 criteria, at least 1 clinical & 1 immunological, or biopsy-proven LN.
- 25. DDx • Rheumatoid arthritis (RA) • Overlap of SLE and RA • Mixed connective tissue disease (MCTD): characterized by overlapping features of SLE, systemic sclerosis (SSc), and polymyositis (PM) • Undifferentiated connective tissue disease (UCTD) • Systemic sclerosis (SSc) • Sjögren’s syndrome • Vasculitis • Behçet’s syndrome • Kikuchi’s disease • Fibromyalgia
- 26. ASSESSMENT OF DISEASE ACTIVITY AND SEVERITY Clinical evaluation • History & ROS. • The physical examination should be extensive (complete) and include examination of the skin (including scalp and mucous membranes) and lymph nodes, as well as respiratory, cardiovascular, abdominal, musculoskeletal, and neurologic systems. Laboratory evaluation • Leukopenia is common and may reflect active disease & can be result of drugs. • CRP & ESR • Urinalysis/urinary sediment • Spot (untimed) urine protein and creatinine • Serum creatinine and estimated glomerular filtration rate (eGFR) • Anti-double-stranded deoxyribonucleic acid (dsDNA) • C3 and C4 – Low complement levels often indicate active lupus (LN).
- 27. Approach to drug therapy • In general, all patients with SLE with any degree and type of disease activity should be treated with hydroxychloroquine or chloroquine, unless CI. The benefits of hydroxychloroquine or chloroquine in SLE are broad and include relief of constitutional symptoms, musculoskeletal manifestations, and mucocutaneous manifestations
- 28. • Patients with mild lupus manifestations (eg, skin, joint, and mucosal involvement) may be treated with hydroxychloroquine or chloroquine, with and without nonsteroidal antiinflammatory drugs (NSAIDs), and/or short-term use of low-dose glucocorticoids (eg, ≤ 7.5 mg prednisone equivalent per day). • Patients with moderate lupus involvement are defined as having significant but non-organ-threatening disease (eg, constitutional, cutaneous, musculoskeletal, or hematologic). Patients usually respond to hydroxychloroquine or chloroquine plus short-term therapy with 5 to 15 mg of prednisone (or equivalent) daily. Prednisone is usually tapered once hydroxychloroquine or chloroquine has taken effect. A steroid-sparing immunosuppressive agent (eg, azathioprine or methotrexate) is often required to control symptoms.
- 29. • Severe/life-threatening manifestations 2ndry to major organ involvement (eg, renal & CNS) generally require an initial period of intensive immunosuppressive therapy (induction therapy) to control the disease and halt tissue injury. Patients are usually treated for a short period of time with high doses of systemic glucocorticoids (eg, intravenous “pulses” of methylprednisolone, 0.5 to 1 g/day for three days in acutely ill patients, or 1 to 2 mg/kg/day in more stable patients) used alone or in combination with other immunosuppressive agents. • Glucocorticoid therapy: rapidly reduces inflammation, helps to achieve disease control. However, long-term adverse effects. • Examples of other immunosuppressive agents that may be used include mycophenolate, cyclophosphamide, or rituximab. This initial therapy is subsequently followed by a longer period of less intensive, and ideally less toxic, maintenance therapy to consolidate remission and prevent flares. During this phase of treatment, the dose of prednisone or equivalent is reduced while monitoring clinical and laboratory measures of disease activity.
- 30. Summary of the medications: Medications used to treat SLE manifestations includes: • Biologic DMARDs (disease-modifying antirheumatic drugs): Belimumab, rituximab, IV immune globulin • Nonbiologic DMARDS: Cyclophosphamide, methotrexate, azathioprine, mycophenolate, cyclosporine • Nonsteroidal anti-inflammatory drugs (NSAIDS; eg, ibuprofen, naproxen, diclofenac) • Corticosteroids (eg, methylprednisolone, prednisone) • Antimalarials (eg, hydroxychloroquine)
- 31. • Genetic factors predisposing to ↑ frequency & severity include African-American race where the incidence among those in US is 34-51%. Also younger age, male gender. • Glomerular injury in SLE is primarily mediated by immune complex formation • Some antibodies bind directly to GBM & mesangium & this can cause activation of classical pathway >> chemo attractants to neutrophils/mononuclear cells causing: mesangial/focal/diffuse proliferative GN. Lupus Nephritis
- 32. • Most patients will have renal involvement at some point, usually abnormal urinalysis. Most abnormality emerge soon after SLE diagnosis (6-36 months). Eventually ↑creatinine in 30% of pts, but uncommon within first few years. • Renal biopsy should be performed in most patients to confirm the diagnosis & classify Lupus nephritis. X (proteinuria that is less than 500 mg/day and a bland urine sediment)
- 33. The ISN classification system • Developed in 2004 (the Renal Pathology Society/International Society of Nephrology, or RPS/ISN, classification). • Classifies Lupus Nephritis into 6 different classes based upon kidney biopsy histopathology.
- 34. • Overlap of lupus and ANCA-associated glomerulonephritis
- 35. Other renal manifestations • Renal-limited lupus-like nephritis • Tubulointerstitial nephritis: (interstitial infiltrate, tubular injury) with or without immune deposits along the tubular basement membrane is a common finding in lupus nephritis and is almost always associated with concurrent glomerular disease. Should be suspected when there’s rise in creatinine, with normal urinalysis or few rbcs. It is associated with higher risk to develop ESRD.
- 36. • Vascular disease (such as thrombotic microangiopathy) is common and can adversely affect the prognosis of the renal disease. Patients may present with glomerular and vascular thrombi, often in association with antiphospholipid antibodies such as the lupus anticoagulant (LA) and anticardiolipin antibodies.
- 37. • Drug-induced lupus: A variety of drugs can induce a lupus-like syndrome, such as minocycline, hydralazine, isoniazid and, historically, procainamide. Renal involvement is uncommon, but a proliferative glomerulonephritis or the nephrotic syndrome can occur.
- 38. • Silent lupus nephritis is defined as the presence of mesangial, focal, or diffuse proliferative glomerulonephritis in patients without clinical evidence of renal disease. Silent lupus nephritis is associated with a benign renal outcome.
- 39. Treatment in Lupus Nephritis • Delayed therapy: The likelihood of a successful initial outcome is > if therapy for LN is initiated relatively early in the course of the disease.
- 40. • Delaying therapy because of presumed mild disease can be associated with ↑ glomerular injury, progressive tubulointerstitial fibrosis, glomerulosclerosis, and therefore a ↓ response to immunosuppressive drugs. Also relapses and lack of response to therapy are associated with an increased risk of progressive CKD.
- 41. NONIMMUNOSUPPRESSIVE THERAPY • Aggressive antihypertensive and, in patients with proteinuria, antiproteinuric therapy with blockade of the renin-angiotensin system (eg, angiotensin-converting enzyme [ACE] inhibitor or angiotensin II receptor blocker [ARB]). • Lipid lowering with statin therapy (CKD > cardiovascular mobidity & mortality).
- 42. Goal of immunosuppressive therapy • The goal is resolution of inflammatory and immunologic activity, with achievement of a complete response: - A substantial reduction in urine protein excretion - Improvement or stabilization of the serum creatinine - Improvement of the urinary sediment
- 43. Complete response Vs. complete remession • Complete response: the three above mentioned • Complete remession: Renal biopsy showing resolution of inflammation
- 44. PRINCIPLES OF IMMUNOSUPPRESSIVE THERAPY • Initial (induction) therapy involves the administration of potent immunosuppressive drugs to achieve a renal response. The duration of the initial therapy period varies; it can be 3 months - one year • (maintenance) with less aggressive extended immunosuppressive therapy is given once a renal response is achieved for a prolonged period to prevent relapse.
- 45. • Initial therapy should consist of glucocorticoids combined with either cyclophosphamide or mycophenolate mofetil.
- 46. • EXTENDED (MAINTENANCE) THERAPY: Up to 50% of patients with proliferative LN relapse following ↓ or cessation of immunosuppressive therapy. • The relapse rates with an average of about 8%. Relapse is more common when partial rather than complete response is obtained with induction therapy.
- 47. • We prefer mycophenolate mofetil as a maintenance agent (azathriprine can be used). The optimal duration of maintenance therapy is not well defined, although durations of 12 to 24 months have been best studied
- 48. References: • UpToDate • http://www.rheumatologie.at/pdf/sledai.pdf • www.istockphoto.com
Notes de l'éditeur
- In 2012, the Systemic Lupus International Collaborating Clinics (SLICC) proposed revised classification criteria that were developed to address inherent weaknesses of the 1997 American College of Rheumatology (ACR) classification criteria.
- Undifferentiated connective tissue disease — Other patients who have even fewer features suggestive of SLE may be classified as having undifferentiated connective tissue disease (UCTD). Possible SLE — We consider SLE a possible diagnosis in individuals who have only one of the ACR/SLICC criteria, in addition to at least one or two of the other features listed above.