Symbols and Masses of Common Elements and Ions
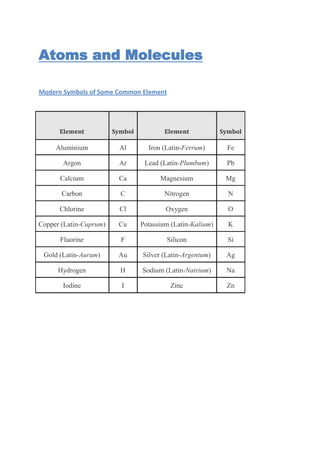
- 1. Atoms and Molecules Modern Symbols of Some Common Element Element Symbol Element Symbol Aluminium Al Iron (Latin-Ferrum) Fe Argon Ar Lead (Latin-Plumbum) Pb Calcium Ca Magnesium Mg Carbon C Nitrogen N Chlorine Cl Oxygen O Copper (Latin-Cuprum) Cu Potassium (Latin-Kalium) K Fluorine F Silicon Si Gold (Latin-Aurum) Au Silver (Latin-Argentum) Ag Hydrogen H Sodium (Latin-Natrium) Na Iodine I Zinc Zn
- 2. Atomic Masses of Some Common Elements u – Unified atomic mass unit Atomic mass unit mass of a C-12 atom 1u mass of a C-12 atom Element Atomic mass Element Atomic mass (u) (u) Hydrogen 1 Potassium 39 Helium 4 Calcium 40 Carbon 12 Argon 40 Nitrogen 14 Iron 56 Oxygen 16 Copper 63.5 Fluorine 19 Zinc 65 Neon 20 Bromine 80 Sodium 23 Silver 108 Magnesium 24 Gold 197 Chlorine 35.5 Sulphur 32
- 3. The name of some common ions with their symbols is given below. Cation Symbol Atomicity Anion Symbol Atomicity Aluminium Al3+ Monoatomic Bromide Br– Monoatomic Ammonium Polyatomic Carbonate Tetra-atomic Calcium Ca2+ Monoatomic Chloride Cl– Monoatomic Cuprous ion Cu+ Monoatomic Fluoride F– Monoatomic Cupric ion Cu2+ Monoatomic Hydride H– Monoatomic Hydrogen H+ Monoatomic Hydroxide OH– Diatomic Ferric ion Fe3+ Monoatomic Iodide I– Monoatomic Magnesium Mg2+ Monoatomic Nitrate Tetra-atomic Nickel Ni2+ Monoatomic Nitride N3– Monoatomic Potassium K+ Monoatomic Nitrite Tetra-atomic Silver Ag+ Monoatomic Oxide O2– Monoatomic Sodium Na+ Monoatomic Phosphate Polyatomic Zinc Zn2+ Monoatomic Sulphate Polyatomic Hydrogen carbonate Polyatomic Sulphite Tetra-atomic The table given below illustrates some examples of ionic compounds. Ionic Compound Combining Ratio by Ratio by number of Elements mass ions Calcium oxide Calcium, oxygen 5:2 1:1
- 4. Magnesium Magnesium, chlorine 24:71 1:2 chloride Aluminium, sulphur 27:32 2:3 Aluminium sulphide The valencies of some common ions are given in the following table. Name of ion Symbol Valency Name of ion Symbol Valency Aluminium Al3+ 3 Sulphite 2 Ammonium 1 Bromide Br− 1 Calcium Ca2+ 2 Carbonate 2 Copper(II) Cu2+ 2 Chloride Cl− 1 Hydrogen H+ 1 Hydride H− Iron(II) Fe2+ 2 Hydrogen 1 carbonate Iron(III) Fe3+ 3 Hydroxide OH− 1 Magnesium Mg2+ 2 Nitrate 1 Nickel Ni2+ 2 Nitrite 1 Potassium K+ 1 Oxide O2− 2 Silver Ag+ 1 Phosphate 3 Sodium Na+ 1 Sulphate 2 Zinc Zn2+ 2 Sulphide S2− 2
