Calculating Solution Concentrations: Grams/Liter, Molarity, Mass Percent, PPM
•Télécharger en tant que PPSX, PDF•
3 j'aime•883 vues
Signaler
Partager
Signaler
Partager
Contenu connexe
Tendances
Tendances (20)
solutions and their concentrations in Analytical chemistry by Azad Alshatteri

solutions and their concentrations in Analytical chemistry by Azad Alshatteri
Similaire à Calculating Solution Concentrations: Grams/Liter, Molarity, Mass Percent, PPM
Similaire à Calculating Solution Concentrations: Grams/Liter, Molarity, Mass Percent, PPM (20)
Concentration Expression of Solution| L 07|PhysicalPharmacyLectureSeries

Concentration Expression of Solution| L 07|PhysicalPharmacyLectureSeries
Plus de Mahbub Alwathoni
Plus de Mahbub Alwathoni (20)
Mata Kuliah Komputer & Media Pembelajaran S1 PGSD UT

Mata Kuliah Komputer & Media Pembelajaran S1 PGSD UT
Dernier
Dernier (20)
Time Series Foundation Models - current state and future directions

Time Series Foundation Models - current state and future directions
Ensuring Technical Readiness For Copilot in Microsoft 365

Ensuring Technical Readiness For Copilot in Microsoft 365
"Subclassing and Composition – A Pythonic Tour of Trade-Offs", Hynek Schlawack

"Subclassing and Composition – A Pythonic Tour of Trade-Offs", Hynek Schlawack
Tampa BSides - Chef's Tour of Microsoft Security Adoption Framework (SAF)

Tampa BSides - Chef's Tour of Microsoft Security Adoption Framework (SAF)
Unraveling Multimodality with Large Language Models.pdf

Unraveling Multimodality with Large Language Models.pdf
Use of FIDO in the Payments and Identity Landscape: FIDO Paris Seminar.pptx

Use of FIDO in the Payments and Identity Landscape: FIDO Paris Seminar.pptx
Developer Data Modeling Mistakes: From Postgres to NoSQL

Developer Data Modeling Mistakes: From Postgres to NoSQL
Digital Identity is Under Attack: FIDO Paris Seminar.pptx

Digital Identity is Under Attack: FIDO Paris Seminar.pptx
The Role of FIDO in a Cyber Secure Netherlands: FIDO Paris Seminar.pptx

The Role of FIDO in a Cyber Secure Netherlands: FIDO Paris Seminar.pptx
Dev Dives: Streamline document processing with UiPath Studio Web

Dev Dives: Streamline document processing with UiPath Studio Web
The Fit for Passkeys for Employee and Consumer Sign-ins: FIDO Paris Seminar.pptx

The Fit for Passkeys for Employee and Consumer Sign-ins: FIDO Paris Seminar.pptx
New from BookNet Canada for 2024: BNC CataList - Tech Forum 2024

New from BookNet Canada for 2024: BNC CataList - Tech Forum 2024
Merck Moving Beyond Passwords: FIDO Paris Seminar.pptx

Merck Moving Beyond Passwords: FIDO Paris Seminar.pptx
Calculating Solution Concentrations: Grams/Liter, Molarity, Mass Percent, PPM
- 2. CA Standards Students know the definitions of solute and solvent. Students know how to calculate the concentration of a solute in terms of grams per liter, molarity, parts per million, and percent composition.
- 3. Calculations of Solution Concentration: Grams per Liter Grams per liter is the ratio of mass units of solute to volume (liters) of solution mass of solute Grams per liter volume of solution
- 4. Calculations of Solution Concentration: Molarity Molarity is the ratio of moles of solute to liters of solution moles of solute Molarity M Liter of solution
- 5. Calculations of Solution Concentration: Mass Percent Mass percent is the ratio of mass units of solute to mass units of solution, expressed as a percent mass of solute Mass percent x100 mass of solution
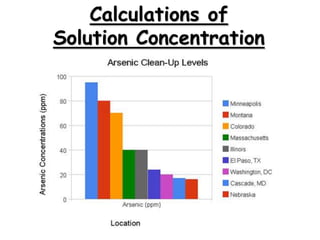
- 6. Calculations of Solution Concentration: Parts per Million Parts per million is the ratio of mass units of solute to mass units of solution, multiplied by one million (106) mass of solute 6 Mass percent x10 mass of solution
- 7. A Simplifying Assumption • 1 ml of water = 1 gram of water • 1000 ml of water = 1 liter = 1000 grams • Assume that solutions with water as the solvent have the density of pure water (1 mL = 1 gram) – It’s not true, but it’s close enough
