liquid metal battery
- 1. LIQUID METAL BATTERIES BY – ABHISHEK SINGH 1401440004 ME 14
- 2. CONTENTS • Introduction • Liquid metal batteries • Principle involved • Working • Charging and discharging • Choice of materials • Benefits of going liquid • Commercialization • Conclusion
- 3. • Donald Sadoway (right) of the Department of Material science and engineering MIT, David Bradwell MEng ’06, PhD ’11, and their collaborators have developed a novel molten-metal battery that is low-cost, high- capacity, efficient, long- lasting, and easy to manufacture • Characteristic that make it ideal for storing electricity on power grids today and in the future.
- 4. LIQUID METAL BATTERIES • Molten salt batteries ( liquid metal batteries) are a class of battery that uses molten salts as an electrolyte • offers both a high energy density and a high power density. • Traditional thermal batteries can store in their solid state at room temperature for long periods • Rechargeable liquid metal batteries are used for electric vehicles and potentially for grid energy storage,
- 6. PRINCIPLE INVOLVED • Sadoway turned to a process he knew well: aluminum smelting. • Aluminum smelting is a huge scale, inexpensive process conducted inside electrochemical cells that operate reliably over long periods and produce metal at very low cost while consuming large amounts of electrical energy. • Sadoway thought: "Could we run the smelter in reverse so it gives back its electricity?” i.e applying heat and getting electricity .
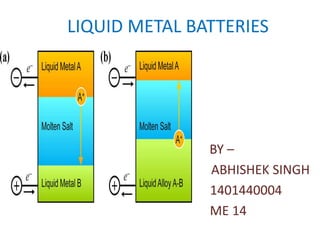
- 7. • WORKING – the negative electrode (top) is a low- density metal called here Metal A; – the positive electrode (bottom) is a higher-density metal called Metal B; – The electrolyte between them is a molten salt. – During discharge (shown here), Metal A loses electrons (e-), becoming ions (A+) that travel through the electrolyte to the bottom electrode – The electrons pass through an external circuit, powering an electric load on the way. – At the bottom electrode, the Metal A ions and electrons rejoin and then alloy with the Metals electrode.
- 8. CHARGING AND DISCHARGING • Discharging Charging
- 9. CHOICE OF MATERIALS • The challenge was to choose the best materials for the new battery, particularly for its electrodes. • To keep costs down, Sadoway and Bradwell needed to use electrode materials that were earth abundant, inexpensive, and long lived. • To achieve high voltage, they had to pair a strong electron donor with a strong electron acceptor. • And finally, all the materials had to be liquid at practical temperatures. • Sadoway and Bradwell chose magnesium for the top electrode, antimony for the bottom electrode, and a salt mixture containing magnesium chloride for the electrolyte
- 10. • To keep the components melted, the battery had to operate at 700 degrees Celsius (1,292 degrees Fahrenheit), which would corrode and degrade the cell wall . • In place of the antimony, they check lead, tin, bismuth, and alloys of similar metals . • New cell chemistries began to show significant reductions in operating temperature. • Cells of sodium and bismuth operated at 560 degrees Celsius. • Lithium and bismuth cells operated at 550 C.
- 11. • And a battery with a negative electrode of lithium and a positive electrode of an antimony - lead alloy operated at 450 C. • They recently assembled a proof of concept cell using a positive electrode of a lead - bismuth alloy, a negative electrode of sodium metal, and a novel electrolyte of a mixed Hydroxide halide. • Cell operated at just 270 C—more than 400 C lower than the initial magnesium antimony battery while maintaining the same novel cell design of three naturally separating liquid layers
- 12. BENEFITS OF GOING LIQUID • Because the components are liquid, the transfer of electrical charges and chemical constituents within each component and from one to another is ultrafast, permitting the rapid flow of large currents into and out of the battery. • While solid electrodes are prone to cracking and other forms of mechanical failure over time, liquid electrodes do not degrade with use. • The self segregating nature of the liquid components could facilitate simpler, less expensive manufacturing compared to conventional batteries.
- 13. COMMERCIALIZATION • In 2010, the Liquid Metal Battery Corporation (LMBC) was formed to commercialize the Liquid Metal Battery technology invented at MIT. • LMBC was renamed Ambri in 2012; the name "Ambri" is derived from "cAMBRIdge” , where the company is headquartered • In 2012, Ambri received $15 million in funding from Bill Gates, Khosla Ventures and Total S.A. • In 2014, Ambri raised $35 million in funding from Bill Gates, Khosla Ventures, Total S.A., KLP Enterprises and GVB.
- 14. CONCLUSION • Cells made of low cost , Earth abundant materials confirm that the liquid battery operates efficiently without losing significant capacity or mechanically degrading—common problems in today's batteries with solid electrodes. • Large scale storage would also make today's power grid more resilient and efficient, allowing operators to deliver quick supplies during outages and to meet temporary demand .
- 15. THANK YOU