Asd ppt
- 1. ASD SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 1
- 2. ASD • Small ASDs with diameter <5 mm and no evidence of RV volume overload may not require closure as these do not usually impact the natural history. • Unrepaired ASDs with significant shunting can result in right-sided volume overload, with progressive heart failure, arrhythmias, hemodynamically significant tricuspid regurgitation, pulmonary hypertension, and reduced survival. • Current guidelines recommend ASD closure in the presence of right- sided volume overload, that is, right ventricular or right atrial dilatation in a symptomatic or asymptomatic patient. SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 2
- 3. ASD An ASD other than secundum ASD should be repaired surgically. Closure of ASD may be considered in some patients regardless of evidence of right-sided enlargement due to risk of paradoxical embolism: in professional divers patients undergoing pacemaker implantation prior to pregnancy SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 3
- 4. ASD • Closure in presence of symptoms or right-sided heart enlargement prevents further deterioration and helps normalize the right-sided dilatation. • Natural history studies of ASD closure show reduced survival after closure in patients older than 24 years of age or with pulmonary hypertension (systolic PAP ≥ 40 mm Hg) • Also closure in patients over 40 years of age, while improving symptoms and mortality compared with a medically managed group, did not reduce the risk of atrial arrhythmias. SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 4
- 5. ASD closure indications Class I Transcatheter secundum ASD closure is indicated in patients with hemodynamically significant ASD with suitable anatomic features Class IIa It is reasonable to perform transcatheter secundum ASD closure in patients with transient right-to- left shunting at the atrial level who have experienced sequelae of paradoxical emboli such as stroke or recurrent transient ischemic attack It is reasonable to perform transcatheter secundum ASD closure in patients with transient right-to- left shunting at the atrial level who are symptomatic because of cyanosis and who do not require such a communication to maintain adequate cardiac output Class IIb Transcatheter closure may be considered in patients with a small secundum ASD who are believed to be at risk of thromboembolic events (eg, patients with a transvenous pacing system or chronically indwelling intravenous catheters, patients with hypercoagulable states) SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 5
- 6. ASD Class III Transcatheter secundum ASD closure is not indicated in patients with a small secundum ASD of no hemodynamic significance and with no other risk factors Transcatheter ASD closure should not be performed with currently available devices in patients with ASDs other than those of the secundum variety. This would include defects of septum primum, sinus venosus defects, and unroofed coronary sinus defects (Level of Evidence: C). Transcatheter ASD closure is contraindicated in the management of patients with a secundum ASD and advanced pulmonary vascular obstructive disease SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 6
- 7. ASD closure:Procedural Details A complete right heart catheterization is first performed to measure shunt fraction, pulmonary artery pressures, and pulmonary capillary wedge pressure. In patients older than 40 years, a coronary angiogram is also performed,also perform pulmonary angiogram with levo phase imaging to assess drainage of all four pulmonary veins into the left atrium. Some operators perform right upper pulmonary vein angiogram in 35-degree LAO cranial projection, which provides an angiographic roadmap of the interatrial septum to facilitate closure. The ICE catheter is advanced into the right atrium and the interatrial septum adequately interrogated for assessment of various rims, measuring defect size and confirming pulmonary venous drainage. A rim is considered to be deficient if its length is <5 mm, and absent if it is ≤1 mm. The rims should not be deficient (except anterior rim, as many patients lack the anterior rim and it is not a contraindication). The directions include a “warning” that a deficient aortic rim may incur increased risk of erosion, but data are insufficient as discussed later.
- 8. Management and Indications for Atrial Septal Defect Closure In patients with PAH, pulmonary vasodilator testing to assess for reversibility and test occlusion of ASD should be performed. Inhaled nitric oxide is used commonly as a pulmonary vasodilator. A positive vasoreactivity response is defined as a reduction of mean PAP of >10 mm Hg with resultant mean PAP of 40 mm Hg or less, without fall in cardiac output. Closure in such patients may be performed if there is net left-to-right shunting, PA pressure <2/3 systemic levels, PVR <2/3 SVR, or when responsive to either pulmonary vasodilator testing or test occlusion. A favorable response is indicated by a fall in mean pulmonary artery pressure with test occlusion with no decrease in cardiac output and no rise in right atrial pressure.
- 9. Management and Indications for Atrial Septal Defect Closure In patients with PAH, when pulmonary vasodilator testing has unfavorable response, pulmonary vasodilator therapy should be initiated and hemodynamics reassessed a few months later. ASD closure is also indicated in presence of paradoxical embolism and documented platypnea orthodeoxia. An absolute contraindication (Class III) for closure is irreversible PAH and no evidence of left-to-right shunt.
- 10. ASD hemodynamic features influence difficulties of transcatheter ASD closure. Candidates for ASD closure have a hemodynamically significant atrial shunt or the presence of right ventricular volume overload, and/or clinical symptoms of dyspnea, reduced exercise capacity, or paradoxical embolism. Pulmonary vascular resistance <5 Wood units/m2 and the peak pulmonary artery pressure ≤70% of the systemic blood pressure are also important conditions for ASD closure. Although most pediatric patients with ASD fulfill the hemodynamic criteria, the incidence of pulmonary artery hypertension is significantly increased in adult populations. It is well known that the natural course in patients with ASD and pulmonary hypertension is significantly worse than in patients without pulmonary hypertension transcatheter closure without cardiopulmonary bypass can be performed even in such high- risk patients, and Importantly, the more significant reduction of pulmonary artery pressure can be achieved even in severe pulmonary hypertension. Even if the initial hemodynamic parameter appears to be untreatable (or contraindicated) for ASD closure, catheter closure of ASD may be performed if such pulmonary vascular resistance can be considered as responder for pulmonary artery specific vasodilators .Long- term follow-up is mandatory especially in these high-risk populations SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 10
- 11. ASD in he elderly patients In the elderly patients with ASD, hemodynamic features are significantly different from those in children and young adults. Elderly patients with ASD frequently present with hemodynamic abnormalities such as pulmonary hypertension, atrial arrhythmias, and valvular regurgitation, which causes congestive heart failure As the incidence of pulmonary hypertension significantly increases with age in ASD patients, the decision of ASD closure is sometimes difficult especially in patients with severe pulmonary hypertension. Moreover, various comorbidities, such as systemic hypertension, chronic obstructive pulmonary disease, coronary artery disease, chronic kidney disease, and left ventricular diastolic dysfunction often complicate the clinical features in this population. Left ventricular diastolic dysfunction, which is also seen as part of normal aging and frequently occurs in elderly individuals with hypertension or increased arterial stiffness, may cause acute congestive heart failure after ASD closure Previous studies have suggested that development of acute congestive heart failure is due to abrupt elevation in left ventricular preload following transcatheter ASD closure, especially in elderly patients with impaired left ventricular systolic or diastolic function SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 11
- 12. ASD in he elderly patients impaired left ventricular diastolic function estimated by decreased e′ and increased E/e′, pre- and periprocedural anticongestive medication is important and effective for preventing congestive heart failure after ASD closure in elderly patients monitore pulmonary capillary wedge pressure (PCWP) during the procedure to avoid the acute congestive heart failure caused by ASD closure in elderly patients. If mean PCWP increased > 10 mmHg from the baseline value during balloon occlusion of the defect (test balloon occlusion), or PCWP increased >20 mmHg, such ASD closure could lead to the development of pulmonary edema and the procedure should be abandoned Especially in patients who had a history of heart failure, we considered to be hemodynamically high-risk patients. Creation of a fenestration hall in the device may avoid the abrupt hemodynamic change after the transcatheter closure of ASD. However, the optimal fenestration size has not been evaluated, and the experiences are still limited SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 12
- 13. Special groups who may be suitable for consideration of balloon occlusion trial 1. RV dilatation 2. significant PAH’ but responsive to PVT 3. Significant LV impairment ,off-loading across ASD which may decompensate once ASD closed SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 13
- 14. ASD ECHO transesophageal echocardiography (TEE) has long been the standard modality for ASD closure. However, intracardiac echocardiography (ICE) is gradually replacing the role of TEE recently. Transthoracic echocardiography (TTE) may also be used especially in patients with good windows for echocardiography such as small children SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 14
- 15. ASD ECHO SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 15 PSAX- IAS separates Rt & lt atrium and runs posteriorly from NCC of aortic valve. L Not seen in entirety as a result of drop out artefact APICAL 4C- Posterior aspect of I nteratrial septum is clearly delineated in this view but drop out artefact is seen in region of fossa ovalis. Pulmonary venous drainage- 3 veins draining to LA APICAL 5C VIEW- Anterior aspect of interatrial septum
- 16. when to suspect ASD in 2d echo SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 16 • right ventricular dilation • abnormal motion of IVS: brisk anterior movement in early systole or flattened movement throughout diastole • SUB COSTAL 4C VIEW- is useful in patients with COPD and ventilated patients, viewed with breath held in inspiration- index marker in 3o' clock position. No IAS drop outs • SUB COSTAL SHORT AXIS- Index marker at 12o'clock position and sweeping the transducer from midline to Rt side of patient
- 17. SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 17 The Relative Atrial Index (RAI) If you detect IAS drop out in apical 4c view ,then determine RELATIVE ATRIAL INDEX A Simple, Reliable, and Robust Transthoracic Echocardiographic Indicator of Atrial Defects
- 18. ASD ECHO SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 18 I En face view in 2D First the apical 4c view was taken. The image index marker was at approximately kept at 1 o'clock. Keeping the atrial septum and ASD in the region of interest, the transducer was rotated counterclockwise approximately 45° to 60°.
- 19. ASD ECHO SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 19 For contrast echocardiography APICAL 4C VIEW is used AGITATED SALINE USED- 5m1 in each 10m1 syringe. 0.5m1 of air taken in the syringe and agitated to create microbubbles. arrow shows negative contrast effect ,a direct evidence of shunt(non contrast blood in RA) Extent of shunting tend to focus on numbers of bubbles seen in a single still frame in the left atrium. Shunt grading incorporates : Grade 1: 5 bubbles Grade 2: 5 to 25 bubbles Grade 3: >25 bubbles Grade 4: Opacification of chamber
- 20. ASD echo 1. Detect size from multiple views (SC frontal, SC sagital, parasternal short axis, apical 4C ) 2. Determine Total Septal length 3. Determine the Rims: Mitral Valve (apical 4c) Aortic (PS short) SVC / pulmonary veins SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 20
- 21. ASD ECHO Other important views SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 21 • To visualize SVC: Suprasternal short axis -index marker in 4 o'clock position • L-SVC is seen from left supraclavicular fossa or suprasternal short axis • Suprasternal short axis :to visualise the the pulmonary veins draining into left atrium • Cleft mitral valve in AVCD in 12o'clock position in PSAX
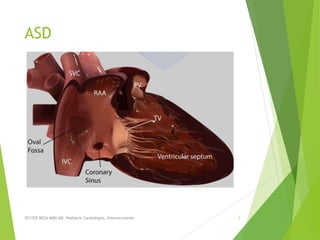
- 22. ASD ECHO :RIMS OF ASD SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 22 the rims of a secundum ASD are labeled as: 1. aortic (superoanterior) 2. atrioventricular (AV) valve (mitral or inferoanterior) 3. superior venacaval (SVC or superoposterior) 4. inferior venacaval (IVC or inferoposterior),most important 5. posterior (from the posterior free wall of the atria, coronary sinus rim).
- 23. SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 23
- 24. ASD RIMS By conventional definition, a margin 5 mm is considered to be adequate. Podnar et al. defined 10 morphological variations of defects, the most common type being the defect with deficient aortic rim (42.1%). The other variants included central defects (24.2%), deficient inferoposterior rim (12.1%), perforated aneurysm of the septum (7.9%), multiple defects (7.3%), combined deficiency of mitral and aortic rims (4.1%), Deficient SVC rim (1%), and deficient coronary sinus rim (1%). SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 24
- 25. The locations of rim deficiencies (A) aortic rim, (B) IVC rim, (C) SVC rim deficiency, (D) posterior rim, (E) atrioventricular valve rim. SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 25 Specific rims of the sASD are measured on 2D imaging Consideration should be given to the integrity of the rim tissue. Thin flimsy tissue is unlikely to have the strength to hold the device disc despite appearing an adequate length.
- 26. ASD rims Traditional teaching allows the aortic rim to be absent since the ASD closure device is able to anchor using both left and right discs by splaying across the aortic root. a number of erosions resulting in cardiac perforations have led to caution in such ,usually arises within the first 12 months although has been reported as late as 8 years post procedure. High risk features appear to include absent or limited aortic and superior vena cava rims ,compounded by significant device oversizing, and motion (‘see-saw’ movement) where the waist of the device (or the disc edges protrude into the aortic wall) is in continual contact with the aortic root, perhaps causing distortion of its wall SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 26
- 27. The locations of rim deficiencies In atrioventricular valve rim deficiency, encroachment of device onto the mitral and/or tricuspid valve is a potential problem. This is a concern especially in infants and young children because of the inherent design of Amplatzer-type devices which have a relatively larger disk-rim width in smaller devices. In case of device encroachment onto the valve, it is generally recommended not to implant a device. There is an extremely rare documented case of erosion on mitral valve SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 27
- 28. The rim deficiencies Deficiency in the surrounding rim(s) is frequently associated with large defects, and may potentially increase the risk of complications such as device embolization, erosion and encroachment of device onto nearby cardiac structures. Aortic rim (antero-superior rim, Figure 1A) deficiency is most common rim deficiency and device implantation is frequently interfered by LA disk prolapse. Erosion risk is higher in aortic rim deficiency as well as device oversizing , thus device selection has to be refrained from undue oversizing. IVC rim (posteroinferior rim, Figure 1B) deficiency is second most common among rim deficiencies and associated with higher risk of device embolization . In case with this rim deficiency, under-sizing of the device may further increase the risk of device embolization, and should be avoided. It is difficult to visualize IVC rim with TEE guidance, so ICE is preferable imaging modality in patients with IVC rim deficiency ,however, so called ‘modified retroflexed view’ may be helpful to visualize IVC rim with TEE guidance Superior vena cava (SVC) rim (posterosuperior rim, Figure 1C) deficiency is a rare condition and may interfere with device positioning When the rim deficiency is extended from SVC rim to aortic rim, this indicates the defect is located superiorly in the atrial roof and may carry higher risk of erosion In case with posterior rim (Figure 1D) deficiency, the feasibility of device closure may be decided by the extent of rim deficiency (Figure 2). In the presence of rim deficiency in large area from IVC to posterior rim, the risk of device embolization is very high and this condition may preclude device closure.SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 28
- 29. 3D echo for ASD 3D echo provides instantaneous appreciation of defect position, orientation, size and spatial relationships to surrounding structures and may offer a superior approach to ASD measurements. Correct anatomical orientation of the 3D image is essential and ideally performed and presented for viewing in a standardised manner. a major advantage of 3D echo, is its ability to simultaneously align anatomical understanding between team members (interventional, surgical and imaging) enabling more effective dialogue and an efficiently performed procedure. 3D multi-plane reconstruction allows a detailed assessment of the ASD rims ,simplifying the 2D incremental 10°–15° sweep performed to assess the precise nature of each rim region. Accurate orientation of the 3D defect in its true long and short axis can be rapidly achieved . 3D TEE ASD measurements appear to be more accurate when compared to 2D since they allow precise orientation with the defect axes SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 29
- 30. TEE for ASD A systematic approach includes a scout at 0 degrees in transverse plane, imaging the AS through its entire length (high, mid, low esophageal levels allowing a quick overview of the defect) then rotating through 0°–180° preferably in 10°–15° increments where specific rim assessments are viewed at 45°, 90°, 135°. Limitations of TEE imaging should be remembered and include poor echo windows; especially low esophageal views where the inferior border of the AS moves away from the esophageal wall and may be difficult to image. LA size effects the angle of view and ensuring the patient is appropriately volume loaded is essential to optimize the widest angle of view as possible. Where image quality remains poor or esophageal intubation is contra-indicated or where the TEE probe cannot be tolerated (e.g., general anaesthesia not advisable) then ICE imaging may be considered. SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 30
- 31. Multiple ASD understanding the accurate anatomy and properties of surrounding/intervening rims of multiple defects is the cornerstone of successful device closure. To overcome these problems, proper use of real time 3-dimensional (RT3D) echocardiography may be helpful RT3D echocardiography enables visualization of the wide ranged septum in a single view in the echocardiography and provides instantaneous understanding of the anatomy as well as identification of complex morphology and spatial relationship between multiple defects Temporary balloon occlusion test may also be useful to investigate compliance of surrounding rims and intervening septum, as well as to predict changes of the defects and rims after device implantation. Also, a careful observation of fluoroscopic images with balloon sizing may provide additional information on the spatial relationship between the defects and intervening septum SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 31
- 32. Check list for assessing ASD for device closure a) Confirm defect is secundum type b) Would surgery be a better option? a) Presence or anomalous pulmonary venous drainage b) Significant mitral valve pathology necessitating surgical intervention c) Detailed assessment of defect a) Defect size b) Number of defects c) Surrounding rims d) Alrial septal aneurysm d) Is there a risk for interference during device placement a) Eustachian valve (less commonly Eustachian ridge) b) Chiari network SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 32
- 33. ASD closure Suitable for closure RV dilatation with or without symptoms with L-R shunt and without significant PAH Paradoxical embolism Platypnoea / orthodeoxia Syndrome Suitable anatomy (adequate n‘ms. detect 535 mm. LA size allows device. no associated lesions) Eisenmenger syndrome with lit-L shunt. significant PAH unresponsive to PVT Unsuitable for closure Current Sepsis Contra-indication to anti- platelet therapy Nickel allergy Unsuitable anatomy inadequate rims defect >36 mm LA size too small associated lesions‘ SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 33
- 34. balloon sizing (BS) at the time of the procedure is an option. However unless the defect opens (folds back) noticeably once the guide wire is placed, reliance can be placed on echo measurements without the need for BS. If BS is employed then this should be a ‘stop-flow’ technique .This approach ensures sizing in line with TEE avoiding oversizing and its subsequent risks .When performed correctly the method relies on TEE imaging. The shaft of the balloon, as it is inflated, is visualized through its entirety (confirming alignment through the central long axis for accurate sizing) and monitored using CFD. When the flow on CFD stops balloon inflation ceases This method prevents oversizing and a prominent balloon waist is avoided (suggesting over-inflation). The narrowest central portion of the balloon is measured on TEE and compared to fluoroscopy. The measured diameter equates to the device size. It is recommended not to oversize more than 2–4 mm in the case of deficient anterosuperior rim due to an increased risk of erosion. SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 34
- 35. ASD ECHO There has been debate on the necessity of balloon sizing for selection of device size. Balloon sizing may be skipped in suitable defects with sufficient surrounding rims ,however, it has long been regarded as an essential step of the procedure .Indeed, balloon sizing may provide more information than averaged size of the defect including compliance of surrounding rims and presence of additional defect. While the balloon stretched diameter or balloon occlusive diameter were used in balloon sizing in the past, currently stop flow diameter (SFD) is recommended as the standard measurement to avoid oversizing In self-centering devices such as the Amplatzer Septal Occluder (ASO) the recommended device size is the same or slightly larger (<2 mm) than the SFD. In cases with aortic rim deficiency, the usual recommendation is to avoid an “oversized” device because of the potential risk of erosion in cases with inferior vena cava (IVC) rim deficiency with higher risk of device embolization, use of an “undersized” device should be avoided. In case of using a non-self-centering device such as the Gore Septal Occluder (GSO) ,a device twice the size of the defect is recommended and the GSO is not recommended for defects >18 mm SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 35
- 36. Illustration of feasible (A), borderline (B) and unfeasible (C) defect for device closure in posterior rim deficiency. For the defects with posterior rim deficiency, the extent of rim deficiency determines feasibility of device closure, so the clear anatomic definition is very important SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 36
- 37. Multiple ASD In case of device closure of multiple defects using multiple devices, the optimal combination of devices based on the comprehensive information from RT3D echocardiography and balloon occlusion test is required to prevent unfavorable interference between multiple devices Usually, a small additional defect adjacent to a larger defect (<7 mm in distance) can also be closed by implantation of a single device in the major defect When the additional defect is also sizable or defects are in distance each other, use of multiple devices is required. For multi-fenestrated defects with a large septal aneurysm, patch-like closure using a non-self-centering device may be a good option Several discrete defects may necessitate multiple device implantations or fenestrations (multiple small ‘pepper pot’ defects) may warrant a large device with a narrow waist placed as centrally as possible on the AS to ensure coverage of the entire fossa ovalis (FO). SEYYED REZA MIRI.MD, Pediatric Cardiologist, Interventionist 37
- 38. ASD closure History Transcatheter closure Initially attempted in adult dogs ,Work expanded to human patients in1976 Amplatzer device first implanted in humans,in 1995, by Hijazi Z
- 39. ASD device History-1 King and Mills, in 1974, originally described and subsequently demonstrated feasibility of closing ASD using a device. Their device consisted of Dacron-covered stainless steel umbrellas, but it required a large delivery sheath and complicated maneuvering during deployment. William Rashkind in 1977 developed a device that Single disk anchoredby “fish hooks”14-16f delivery sheath, size 25, 30 and 35mm initially had 3 stainless steel arms covered with medical grade foam and subsequently a device with 6 arms, all of which ended with miniature hooks. Due to difficulty in deploying the double-umbrella, Lock et al. modified the Rashkind device in 1989 into a clamshell occluder by introducing a second spring in the arms. Rome et al. and Hellenbrand et al. reported clinical experience with this device in the 1990s.
- 40. ASD device History-2 In 1990, Sideris et al. described a buttoned device that consisted of an occluder and a counter- occluder In 1993, Pavenik et al. designed a monodisk device, which consisted of a stainless steel ring covered with 2 layers of wire mesh and hollow pieces of braided stainless steel wire. Babic et al. in 1991, Siveret et al. in 1995, and Hausdorf in 1996 described the use of a double umbrella device called an ASD occluding system deployed via an arterio-venous wire loop. In 1993, Das et al. described a self-centering device delivered transvenously called Das Angel Wing with a subsequent modified device that was called Guardian Angel Wing. When the clamshell device was withdrawn due to stress fractures of the stainless steel arms, the device was modified by using MP35N, a nonferrous alloy, with an additional bend in the arms and the device was called Cardio-SEAL (2nd generation:Clamshell) in 1996.
- 41. ASD device History-3 In 1997, Amplatz developed a self-expanding prosthesis made of nitinol (nickel and titanium alloy) wire mesh with 2 round disks and a connecting short waist. This device was called Amplatzer septal occluder (ASO) and was approved by the FDA in December 2001. In 1998, the device was further modified by using a self-centering mechanism with microsprings between the umbrellas and the device was called STARFlex The Helex occluder is made of a 0.012-inch nitinol wire covered with an ultra- thin expanded polytetrafluoroethylene membrane, and once deployed, the device forms 2 round flexible disks on either side of the septal defect. The FDA approved the Helex occluder in 2006. The STARFlex device was subsequently further modified by using bioabsorbable material to replace the Dacron, and the devices were called BioSTAR (NMT Medical Inc.) and BioTREK (NMT Medical Inc.)
- 42. ASD Closure Devices (current and historical) FDA approved • Amplatzer Septal Occluder (AGA) • Amplatzer Cribriform Septal Occluder (AGA) • HELEX Septal Occluder (Gore) Other devices (current and historical): • Figulla ASD Occluder (Occlutech) • AtriaSept ASD Device (CARDIA) • BioSTAR - Bioabsorbable Septal Repair Implant (NMT) • Sideris Buttoned Device • ASDOS • DAS Angel Wings • Transcatheter Patch
- 43. Asd closure steps Step 1: TTE Assessment Step 2: TEE or ICE imaging Step 3: Hemodynamic assessment • venous access - 8Fr sheath (+/- Arterial line) • Right heart cath: qp:qs; pvr etc Step 4: Balloon size and device selection “stop flow” waist +/- 2 mm Step 5: Load device, advance across ASD and open Step 6: Assess by ECHO and “Wiggle” Step 7: release and reassess enhanced atrial to septal alignment using a modified Mullins sheath achieving good septal apposition during delivery of both components
- 44. Aspirin 325 mg is usually administered before the procedure and Clopidogrel 600 mg loading dose at the end of the procedure. Femoral venous access is obtained in bilateral groins (or 2 sheaths in the same vein), one of which ,9 Fr 35-cm sheath is for the ICE catheter to easily traverse the iliac vein into the inferior vena cava, particularly for left femoral vein insertion. Heparin is administered to maintain ACT >250 seconds and a dose of intravenous antibiotic is administered prior to device deployment.
- 45. ASD closure is usually performed in the cardiac catheterization laboratory with conscious sedation and fluoroscopic +/- ICE guidance For complex septal anatomy, such as multiple ASDs, TEE may be preferred. Advantages of ICE over TEE include no need for general anesthesia or additional cardiologists to perform the procedure, better views of the posteroinferior part of the interatrial septum, and shorter procedure times. Most operators use the AcuNav ICE catheter
- 46. Management and Indications for Atrial Septal Defect Closure In patients with PAH, pulmonary vasodilator testing to assess for reversibility and test occlusion of ASD should be performed. Inhaled nitric oxide is used commonly as a pulmonary vasodilator. A positive vasoreactivity response is defined as a reduction of mean PAP of >10 mm Hg with resultant mean PAP of 40 mm Hg or less, without fall in cardiac output. Closure in such patients may be performed if there is net left-to-right shunting, PA pressure <2/3 systemic levels, PVR <2/3 SVR, or when responsive to either pulmonary vasodilator testing or test occlusion. A favorable response is indicated by a fall in mean pulmonary artery pressure with test occlusion with no decrease in cardiac output and no rise in right atrial pressure.
- 47. Management and Indications for Atrial Septal Defect Closure In patients with PAH, when pulmonary vasodilator testing has unfavorable response, pulmonary vasodilator therapy should be initiated and hemodynamics reassessed a few months later. ASD closure is also indicated in presence of paradoxical embolism and documented platypnea orthodeoxia. An absolute contraindication (Class III) for closure is irreversible PAH and no evidence of left-to-right shunt.
- 48. Procedural Details ASD closure Balloon sizing is the next step and is usually performed with an AGA sizing balloon or NuMed sizing balloon. Under fluoroscopic and ICE guidance, the balloon catheter is placed in the defect over the extra-stiff guidewire and the balloon is gently inflated until no flow is visualized by color Doppler on ICE imaging. It is very important to stop inflating when flow ceases (stop-flow diameter) to avoid oversizing the defect. This diameter is measured on ICE as well as fluoroscopy. For ASO, device size should be equal to but no larger than 1 to 2 mm above the stop-flow diameter. Helex septal occluder size should be at least twice the stop-flow diameter. For defects >18 mm, ASO is preferable over Helex device.
- 49. Sizing the ASD Two major approaches exit, TEE sizing or balloon sizing (BS). 2D TEE provides high-resolution images where the defect measures slightly smaller than the actual size required for device sizing. Colour flow Doppler (CFD) clearly demonstrates flow and the boundaries of the ASD, being comparable to surgical and device sized measurements When the measurements in all four TEE planes [0, 45, 90, 135] are similar (1–2 mm) the largest is taken as the ASD size. If the measurements are significantly different (≥3 mm) a mental reconstruction will allow an understanding of the overall 3D shape, although 3D TEE has superseded this requirement. When the defect is circular a single diameter measurement is taken. When oval in shape the short and long axes are averaged and correlate with device sizing. The device size chosen is the measured ASD size plus 20% ,If additional features exist [such as atrial septal aneurysm (ASA) or absent aortic rim] ,it is advised usually add 25% to the measured size of the defect thereby allowing for adequate grip of the surrounding rims.
- 50. Procedural Details 1. the intravenous administration of heparin (100 U/kg) 2. the stretch diameter of the ASD is measured with sizing balloon, either OBW by Meditech (20, 27 ,33 mm) or AGA Medical Corporation (24 , 34 mm) 3. Introduction into the left atrium is accomplished coaxially via catheter exchange with a guide wire located in the left superior pulmonary vein. 4. The balloon is filled in the left atrium with contrast material diluted with physiological serum until it reached a volume greater than the diameter of the defect, and is slowly removed under TEE-color Doppler control until the short circuit disappear entirely. 5. The balloon is then slowly deflated and drawn toward the right atrium until the jump of the balloon to the right atrium is noted. The stretch diameter is that of the balloon when the balloon passes from the left to the right atrium, occluding the ASD. 6. Once the balloon is outside the patient, the diameter of the balloon was tested by inflating it with the same amount in millimeters of contrast solution, assuming the stretch diameter to be that of the orifice through which the balloon passes with a certain degree of difficulty, according to the template recommended by the manufacturer.
- 51. Procedural Details ASD closure ICE assessment should include Doppler flow, which may still demonstrate flow through the waist (but should not be present around the disk), and evaluation for obstruction of adjacent structures including atrioventricular valves. If the positioning is unsatisfactory or there is impingement of adjacent structures, the device is retracted back into the delivery sheath and redeployed or replaced with a new device as appropriate. Device positioning can also be confirmed by angiography in LAO cranial projection, which allows separation of the left and right atrial disks. In cases where the device impinges or indents on the aortic root, there may be higher risk of erosion. Once satisfactory positioning is confirmed, the device is released by attaching the plastic vise to the delivery cable and rotating it counterclockwise.
- 52. ASD closure steps After completion of hemodynamic assessment, angiography, and ICE assessment, a Goodale-Loubin (GL) catheter is advanced with a 0.035-inch J-tipped guidewire into the SVC. The GL catheter is moved in a caudal direction and then directed toward the interatrial septum and the ASD crossed with or without the 0.035-inch J-tipped guidewire using ICE and fluoroscopic guidance. The catheter and guidewire are placed in the left superior pulmonary vein, taking care to ensure that the wire tip is not in the left atrial appendage to avoid perforation. The 0.035-inch J-tipped guidewire is exchanged for a 0.035-inch 1-cm Amplatz super-stiff wire, again taking care to ensure that the tip is not in the appendage.
- 53. ASD closure The next steps depend on the device used. For the ASO device, delivery sheath size ranges from 6 Fr to 12 Fr depending on device size chosen. The balloon-sizing catheter is removed, leaving the 0.035-inch wire in place. The delivery cable is passed through the loader and the device is screwed to the tip of the delivery cable. The device and loader are immersed in sterile saline solution and the device is pulled into the loader while flushing through the side arm. The delivery sheath is prepped and the dilator is inserted into the sheath. The short sheath in the femoral vein is removed and the delivery sheath/dilator is then advanced over the 0.035-inch wire, which has been placed in the left upper pulmonary vein. The dilator is removed once it reaches the right atrium and the sheath is de-aired. The sheath is then advanced over the wire into the left atrium, taking care to avoid suction of air in the system. The guidewire is removed and the sheath is flushed carefully. The loading device is then attached to the delivery sheath. Under fluoroscopic guidance, the device is advanced, carefully watching for any sign of air in the system. Once the device is at the tip of the delivery sheath in the left atrium, under fluoroscopic and echocardiography guidance the left atrial disk is deployed by retracting the sheath over the delivery cable. The device is gently pulled against the interatrial septum and with tension on the delivery cable; the sheath is retracted further to deploy the right atrial disk. After deployment, the position is checked by ICE and, if needed, a gentle “to and fro” motion (Minnesota wiggle) can be performed with the delivery cable to assure stable positioning