Early reperfusion in myocardial infarction
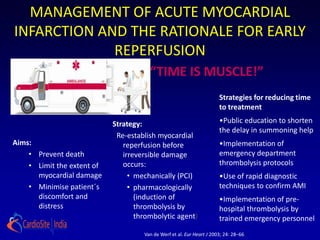
- 1. MANAGEMENT OF ACUTE MYOCARDIAL INFARCTION AND THE RATIONALE FOR EARLY REPERFUSION “TIME IS MUSCLE!” Strategies for reducing time to treatment Strategy: •Public education to shorten the delay in summoning help Re-establish myocardial Aims: reperfusion before •Implementation of • Prevent death irreversible damage emergency department • Limit the extent of occurs: thrombolysis protocols myocardial damage • mechanically (PCI) •Use of rapid diagnostic • Minimise patient´s • pharmacologically techniques to confirm AMI discomfort and (induction of •Implementation of pre- distress thrombolysis by hospital thrombolysis by thrombolytic agent) trained emergency personnel Van de Werf et al. Eur Heart J 2003; 24: 28–66.
- 2. Primary PCI for AMI AMI Treatment Objectives General Objectives: 1) Stabilize patient 2) Open artery to TIMI-3 flow 3) Reduce cardiac work 4) Prevent recurrent thrombosis Reduce Recurrent Triggering Specific Objectives - Bed Rest Restore Epicardial Flow - BP Control Promote Vasodilation - - Blocker - Nitrate - Calcium channel blocker Treat and Prevent Complications of Acute Open Artery Ischemic or Necrotic - Fibrinolysis Myocardium - Primary PCI - - Blocker Prevent Thrombosis - Anti-arrhythymic - Anti-Platelet Agents - GP IIb/IIIa Inhibitor - Anti-Thrombin Agent Adapted from Califf RM. In: Braunwald E., ed. Atlas of Heart Diseases. 1996:Ch. 1, with permission
- 3. Drop of rt-PA breaking down the surface of thrombus. Scanning electron microscopic photograph by Dr. h.c Lennart Nilsson
- 4. For every 30-minute delay from onset of symptoms to primary PCI, there is an 8 percent increase in the relative risk of 1-year mortality Importance of time to reperfusion in patients undergoing primary percutaneous coronary intervention (PCI) for ST segment elevation myocardial infarction (STEMI). This plot is based on the pooled data from 1791 patients undergoing primary PCI for STEMI. After adjusting for baseline risk, there is a curvilinear relationship between the time elapsed from the onset of symptoms to balloon inflation and the rate of mortality at 1 year. For every 30-minute delay from onset of symptoms to primary PCI, there is an 8 percent increase in the relative risk of 1-year mortality. (From De Luca G, Suryapranata H, Ottervanger JP, et al: Time-delay to treatment and mortality in primary angioplasty for acute myocardial infarction: Every minute counts. Circulation 109:1223, 2004
- 6. Generally caused by a Results from stabilization of a platelet aggregate at site of Pathophysiology of STEMI1 completely occlusive thrombus in a coronary artery plaque rupture by fibrin mesh Platelet RBC Fibrin mesh GPIIb/IIIa RBC=red blood cell • The common link between UA/NSTEMI and STEMI is that thrombus formation occurs secondary to the rupture or fissuring of an atherosclerotic plaque in the coronary arteries • This leads to thrombotic occlusion of the coronary artery with interruption of blood flow, resulting in myocardial ischemia and/or necrosis (death of myocardial cells) • Patients with ACS are at high risk of subsequent life-threatening atherothrombotic events such as MI, stroke or vascular death 1. Adapted from Antman EM. In: Califf RM, ed. Atlas of Heart Diseases, VIII. Philadelphia, PA: Current Medicine, 1996.
- 7. GOALS IN REPERFUSION IN AMI RAPID COMPLETE - TIMI III - EPICARDIAL ARTERY INTEGRITY OF MICROCIRCULATION MYOCARDIAL PERFUSION TIMI GRADE - III SUSTAINED
- 8. 12 M 10 9.3% o r 8 p = 0.003 vs TIMI 0/1 t a 6.1 % 6 p < 0.0001 vs TIMI 0/1 l P < 0.0001 vs TIMI 2 i 4 3.7 % t y 2 % 0
- 9. TITAN Other Angiographic Efficacy Endpoint: TIMI Myocardial Perfusion (TMP) Grades 8 TMP Grade 3 TMP Grade 2 TMP Grade 1 TMP Grade 0 7 Normal ground glass Stain present Dye strongly persistent No or minimal blush appearance of blush Blush persists 6 Dye mildly persistent at end of washout on next injection at end of washout Gone by next injection 6.2% 5 p = 0.05 5.1% 4 3 4.4% 2 2.0% 1 n = 46 n = 79 n = 434 n = 203 0 © TIMI 2005. Duplication prohibited by law Gibson et al, Circulation 2000
- 10. Assessing Reperfusion Options for Patients with STEMI1 STEP 1: Assess time and risk (time from symptom onset, risk of STEMI, risk of thrombolysis, time for transport to PCI lab) STEP 2: Determine whether fibrinolysis or invasive strategy is preferred* Fibrinolysis preferred if: Invasive strategy preferred if: • Early presentation (<3 hours) • Skilled PCI lab with surgical backup • Invasive strategy not an option available • Delay to invasive strategy • High risk (i.e. cardiogenic shock) • Contraindications to fibrinolysis • Late presentation (>3 hours) • Diagnosis of STEMI is in doubt *If presentation is <3 hours from onset and there is no delay to an invasive strategy, there is no preference for either strategy 1. Antman EM et al. Circulation 2004; 110: 588–636.
- 11. Because the benefits of fibrinolytic therapy are directly related to the time from symptom onset, treatment benefit is maximized by the earliest possible application of therapy1 Contraindications and cautions for fibrinolysis in patients with STEMI include:1 1. Any prior intracranial hemmorrhage (ICH) 2. A known structural cerebral vascular lesion or malignant intracranial neoplasm 3. Ischemic stroke within 3 months EXCEPT acute ischemic stroke within 3 hours 4. Suspected aortic dissection 5. Active or recent bleeding or bleeding diathesis 6. Systolic blood pressure (SBP) >180 mm Hg or diastolic blood pressure (DBP) >110 mm Hg 7. Current use of anticoagulants 8. Traumatic or prolonged cardiopulmonary resuscitation (CPR) or major surgery Reference 1. Antman EM et al. Circulation 2004; 110: 588–636.
- 12. Common thrombolytics regimens for STEMI1 Initial treatment Co-therapy Contraindications Streptokinase (SK) 1.5 million U in 100 mL None or iv Prior SK or D5W or NS over 3060 min heparin x 2448 hrs anistreplase Alteplase (tPA) 15 mg iv bolus, then iv heparin x 2448 hrs 0.75 mg/kg over 30 min, then 0.5 mg/kg iv over 60 min Total dose not over 100 mg Reteplase (r-PA) 10 U + 10 U iv bolus given iv heparin x 2448 hrs 30 min apart Tenecteplase Single iv bolus iv heparin x 2448 hrs (TNK-tPA) 30 mg if <60 kg 35 mg if 60 kg to <70 kg 40 mg if 70 kg to <80 kg 45 mg if 80 kg to <90 kg 50 mg if ≥90 kg Note: ASA should be given to all patients without contraindications 1. Van de Werf F et al. Eur Heart J 2003; 24: 2866.
- 13. Prothrombotic effects of fibrinolytic therapy. Coronary thrombus is composed of a platelet core with fibrin-thrombin admixture (“white” and “red” clot)
- 14. CONTRAINDICATIONS TO FIBRINOLYTIC THERAPY Absolute contraindications Hemorrhagic stroke or stroke of unknown origin at anytime Ischemic stroke in preceding 6 months Central nervous system trauma or neoplasms Recent major trauma/surgery/head injury (within preceding 3 weeks) Gastrointestinal bleeding within the last month Known bleeding disorder Aortic dissection Non-compressible punctures (e.g. liver biopsy, lumbar puncture)
- 15. Primary PCI versus fibrinolysis for MI Meta analysis of 23 trials 15 14 10 Percentage 8 7 7 5 5 3 2 1 0 Death Re MI Total Stroke Total fibrinolysis prim PCI P<0.0001 Keeley EC. Lancet 2003;361:13-20
- 16. PAMI VS THROMB.META ANALYSIS
- 17. Contemporary Mortality Differences Between Primary PCI and Thrombolysis in STEMI 5,295 Belgian STEMI patients stratified by TIMI risk profile. Primary PCI Thrombolysis In-Hospital Mortality (n = 4,574) (n = 721) P Value High Risk 23.7% 30.6% 0.03 Intermediate Risk 2.9% 3.1% 0.30 Low Risk 0.3% 0.4% 0.60 The mortality benefit of primary PCI over early thrombolysis was offset if the door-to-balloon time exceeded 60 min. Conclusion: Primary PCI in patients with STEMI reduces in-hospital mortality compared with initial thrombolysis, but the benefit is restricted to high-risk patients. Claeys MJ, et al. Arch Intern Med. 2011;171:544-549.
- 18. IMPACT OF TIME-TO-TREATMENT AND 30 DAY MORTALITY : PCI VS. THROMBOLYSIS Thrombolysis PCI 12 30-35-DAY MORTALITY (%) 10 8 6 4 2 0 1 2 8 3 4 5 6 7 TIME FROM ONSET OF PAIN TO THERAPY IN HOURS The figure shows the increase in mortality over time in relation to the start of reperfusion therapy with pharmacological vs. mechanical means, compiling data from a meta-analysis of thrombolysis trials and the NRMI-2 results for mechanical reperfusion. Cannon et al. J Thromb Thrombol 1994; 1: 27–34. Cannon et al. JAMA 2000; 283: 2941–2947. Huber et al. Eur Heart J 2005; 26: 1063–1074.
- 19. CONTRAINDICATIONS TO FIBRINOLYTIC THERAPY Relative contraindications Transient ischaemic attack in preceding 6 months Oral anticoagulant therapy Pregnancy or within 1 week post-partum Refractory hypertension (systolic blood pressure > 180 mm Hg and/or diastolic blood pressure > 110 mm Hg) Advanced liver disease Infective endocarditis Active peptic ulcer Refractory resuscitation
- 20. Strategies for improving pharmacological reperfusion • Despite the success of thrombolysis in clinical practice, various strategies have been investigated in order to improve the effectiveness of pharmacological reperfusion. • The following sections consider the experience to date with strategies such as: 1. Improved fibrinolytic agents offering increased convenience and safety 2. Improved antithrombotic co-therapies 3. Improved antiplatelet co-therapies.
- 21. Strategies for improving pharmacological reperfusion Improved antiplatelet co-therapy i.v. glycoprotein IIb/IIIa inhibitors Clopidogrel Improved Improved fibrinolytic antithrombotic Agents co-therapy convenience Direct thrombin inhibitors (tenecteplase, retepla) (hirudin, bivalirudin) Low mol. Risk of major bleeds weight heparins (tenecteplase) (enoxaparin)
- 22. Enoxaparin improves infarct-related artery patency at 90 minutes Enoxaparin improves infarct- TIMI 2 related artery patency at 90 100 minutes TIMI 3 In the HART II study, 90 % of patients 80 27.2 minutes after starting 60 27.5 therapy, patency rates (TIMI 40 flow grade 2/3) were 80.1% 20 52.9 and 75.1% in the enoxaparin 47.6 0 and UFH groups, respectively. Enoxaparin Overall, enoxaparin Unfractionated heparin appeared to be at least as effective as UFH as an TIMI flow at 90 minutes adjunct to thrombolysis, with a trend toward higher recanalization rates and less reocclusion at 5 to 7 days. Therefore, such a regimen was evaluated in ASSENT-3, the first large-scale trial to compare the two antithrombotics in combination with fibrinolysis. The ASSENT-3 Investigators. Efficacy and safety of tenecteplase in combination with enoxaparin, abciximab, or unfractionated heparin: the ASSENT-3 randomised trial in acute myocardial infarction. Lancet 2001; 358: 605–13. Ross et al, Circulation 2001
- 23. TRANSFER – AMI STUDY DESIGN - I • 52 sites in canad with no pci capability • randomised, nonblinded • within 12 hours of chest pain • Higher risk killip class ii or iii • BP < 100 AND HR > 100,RV inf. • Shock, cabg exclude N. Eng. J. Med. 2009, 360, 2705
- 24. TRANSFER – AMI STUDY DESIGN - I AMI Stent 1059 Patients TR. TO PCI CENTER IMM. Standard treatment Early pci within 6 hours (522) + Stent TNK, CLOP. ± GP II b / III a Blockers ASP, HEP. Angio meantime 32.5 HRS (89 %) PCI – 67 % N. Eng. J. Med. 2009, 360, 2705
- 25. CLINICAL ENDPOINTS – TRANSFER - AMI End Point Standard Routine Early Relative Risk with Treatment PCI (N=536) Routine Early PCI (N=522) (95 % CI) P Value Efficacy end points at 30 days – no (%) Primary end point 90 (17.2) 59 (11.0) 0.64 (0.47-0.87) 0.004 Death 18 (3.4) 24 (4.5) 1.30 (0.71-2.36) 0.39 Reinfarction 30 (5.7) 18 (3.4) 0.57 (0.33-1.04) 0.06 Death or reinfarction 47 (9.0) 38 (7.1) 0.79 (0.52-1.19) 0.25 Recurrent ischemia 11 (2.1) 1 (0.2) 0.09 (0.01-0.68) 0.003 Death, reinfarction, or 58 (11.1) 39 (7.3) 0.65 (0.44-0.96) 0.03 recurrent ischemia New or worsening 29 (5.6) 16 (3.0) 0.54 (0.30-0.98) 0.04 congestive heart failure Cardiogenic shock 16 (3.1) 24 (4.5) 1.46 (0.79-2.72) 0.23 N. Eng. J. Med. 2009, 360, 2705
- 26. 1.0 0.12 TRANSFER-AMI Standard treatment Cumulative Incidence of Death or Reinfarction 0.10 0.8 0.08 0.06 Routine early PCI 0.6 0.04 0.02 0.4 0.00 0 1 2 3 4 5 6 P=0.36 0.2 0.0 0 1 2 3 4 5 6 Months from Randomization No. at risk Standard treatment 522 473 465 462 462 460 458 Routine early PCI 537 497 487 487 484 483 481 N. Eng. J. Med. Kaplan-Meir Curves for Death or Reinfarction and for Reinfarction Only at 6 Months 2009, 360, 2705
- 27. 1.0 0.20 TRANSFER-AMI Cumulative Incidence of Primary End Point Standard treatment 0.15 0.8 0.10 Routine early PCI 0.6 0.05 0.4 0.00 0 1 2 3 4 5 6 0.2 P=0.04 0.0 0 5 10 15 20 25 30 Days from Randomization No. at riskc Standard treatment 522 442 434 434 433 433 432 Routine early PCI 537 488 486 483 481 480 478 Primary End Points at 30 Days N. Eng. J. Med. Composite of Death, reinfarction, worsening heart failure or cardiogenic shock 2009, 360, 2705
- 28. Primary Endpoint: 30-Day Death, re- MI, CHF, Severe Recurrent Ischemia, Shock 18 16.6 16 14 OR=0.537 (0.368, 0.783); p=0.0013 % of Patients 12 10.6 10 8 6 4 Standard (n=496) 2 Pharmacoinvasive (n=508) 0 0 5 10 15 20 25 30 Days from Randomization n=496 422 415 415 414 414 412 n=508 468 466 463 461 460 457
- 29. TRANSFER-AMI Summary • Compared with ‘Standard Treatment’, a ‘Pharmacoinvasive Strategy’ of routine early PCI within 6 hrs after thrombolysis is associated with a 6% absolute (46% relative) reduction in the composite of death, reinfarction, recurrent ischemia, heart failure and shock • The pharmacoinvasive strategy is not associated with any increase in transfusions, severe bleeding or intracranial hemorrhage despite high use of GP IIb/IIIa inhibitors during PCI • In contrast to older trials, routine early PCI after thrombolysis using stents and contemporary pharmacotherapy is safe and effective – Benefit seen despite high cath/PCI rates in Standard Treatment group (including ~40% rescue PCI) N. Eng. J. Med. 2009, 360, 2705
- 30. DANAMI-2: Study Design High-risk ST elevation MI patients (>4 mm elevation), Sx < 12 hrs 5 PCI centers (n=443) and 22 referring hospitals (n=1,129), transfer in < 3 hrs Lytic therapy Primary PCI Primary PCI Front-loaded tPA with transfer without transfer 100 mg (n=567) (n=223) (n=782) Death / MI / Stroke at 30 Days Stopped early by safety and efficacy committee
- 31. DANAMI-2: transfer for primary PCI vs on-site Alteplase (n=1572) P=0.0003 15.0% Primary angioplasty 13.7% Thrombolysis P=0.35 10.0% 7.8% P<0.001 8.0% 6.6% 6.3% p=0.002 5.0% 1.6% 2.0% 1.1% 0.0% Death Re-MI Stroke Any event Anderson 2003;349:733
- 32. PRAGUE-2 study design • Randomised 850 patients with acute ST-elevation myocardial infarction (STEMI) presenting within 12 h of symptom onset to the nearest community hospital. • Thrombolysis group, n=421 or • Immediate transport for primary percutaneous coronary intervention (PCI group, n=429).
- 33. Prague-2: Transfer for PCI vs on-site thrombolysis in acute MI (n=850) Mortality at 30 days 20% Transfer for PCI 15.3% 15% Streptokinase 10.0% 10% 6.8% 7.3% 7.4% 6.0% 5% p=0.12 p=0.02 0% All patients Rx <3hrs of Rx >3hrs of symptoms symptoms Symptoms to balloon 277 min Symptom to lysis 195 min Planned 1200 patients Widimsky, Eur Heart J 2003;24:94
- 34. Transfer for primary PCI vs on-site lytic Quantitative review of 5 trials* *LIMI, Prague I & II, Air PAMI, DANAMI-2 Primary PCI (n=1466) 15.0% 15.0% Thrombolysis (n=1443) 10.0% 8.9% P<0.0001 8.2% 7.0% 6.7% 5.0% P=0.057 P<0.0001 1.8% 2.2% 1.1% 0.0% Death Re-MI Stroke Any event Keeley, Lancet 2003;361:13
- 35. REACT TRIAL DESIGN STEMI – Within 6 hours of chest pain 427 with failed thrombolysis RPCI 144 Rethrombolysis Routine Within 12 hours 142 Treatment For symptoms 141 Heparin
- 36. REACT Trial (Rescue PCI) Event Free Survival 100 R-PCI 81.5 % (95 % CI 74.0%-87.0%) Conservative 67.5 % (95 % CI 58.9 %-74.6%) 80 Event Free Survival 60 Repeated thrombolysis 64.1 % (95 % CI 55.5 %-71.5 %) 40 20 P=0.004 0 0 40 80 120 160 200 240 280 320 360 400 Days after Randomization JACC 2009, 54, 118
- 37. REACT Trial Longer-Term Mortality 100 R-PCI 80.6 % (95 % CI 60.2%-91.2%) 80 Event Free Survival 60 Repeated thrombolysis 76.0% (95 % CI 67.4%-82.6%) 40 Conservative 72.2% (95 % CI 60.6%-80.9%) 20 P=0.026 0 0 1 2 3 4 5 6 7 Years from Randomization Numbers at risk R-PCI 144 132 131 119 84 40 14 0 Repeated thrombolysis 142 119 117 104 66 35 8 0 Conservative 141 118 112 100 72 33 8 0 JACC 2009, 54, 118
- 38. REACT Trial (Rescue PCI) 1-Year Revascularization HRs Repeated thrombolysis HR=1.05 vs Conservative 95% 0.68 to 1.62 R-PCI vs HR=1.05 Repeated thrombolysis 95% 0.32 to 0.86 HR=0.50 R-PCI vs Conservative 95% 0.30 to 0.83 0 0.2 0.4 0.6 0.8 1 1.2 1.4 1.6 1.8 2.0 Favors Comparative Group Favors Reference Group Hazard Ratio Absolute revascularization : Repeat thrombolysis n=41; Conservative n=4(20); R-PCI n=25 JACC 2009, 54, 118
- 39. RESCUE PCI – Death Odds Ratio, random model Bilateral CI, 95 % for trials, 95 % for MA Treatment Better Control Better Conservative vs rescue balloon PTCA Invasive Cons. 5/74 6/75 LIMI 1/16 4/12 Belinkie RESCUE 4/78 7/73 Total 0.53 p=0.18 10/160 17/168 Conservative vs rescue stent PCI REACT 7/141 15/144 MERLIN 15/153 17/154 Total 0.68 p=0.19 22/294 32/298 Total 32/462 49/458 0.63 p=0.055 Relative Risk 0 0.5 1 1.5 2 2.5 3 events / size Metanalysis Collect et al, JACC
- 40. RESCUE PCI – Death or Reinfarction Odds Ratio, random model Bilateral CI, 95 % for trials, 95 % for MA Treatment Better Control Better Conservative vs rescue balloon PTCA Invasive Cons. 10/74 12/75 LIMI 2/16 4/12 Belinkie RESCUE 4/78 7/73 Total 0.62 p=0.18 16/168 23/160 Conservative vs rescue stent PCI REACT 8/141 22/144 MERLIN 26/153 32/154 Total 0.60 p=0.033 34/294 54/298 Total 50/462 77/458 0.60 p=0.012 Relative Risk 0 0.5 1 1.5 2 2.5 3 events / size Metanalysis Collect et al, JACC 2006, 48, 136
- 41. Major components of time delay between onset of infarction and restoration of flow in the infarct-related artery.
- 42. The previous figure shows infarction and restoration of flow in the infarct- related artery. Plotted sequentially from left to right are the time for patients to recognize symptoms and seek medical attention, transportation to the hospital, in-hospital decision-making and implementation of reperfusion strategy, and time for restoration of flow once the reperfusion strategy has been initiated. The time to initiate fibrinolytic therapy is the “ door-to-needle” (D-N) time; this is followed by the period of time required for pharmacological restoration of flow. More time is required to move the patient to the catheterization laboratory for a percutaneous coronary interventional (PCI) procedure, referred to as the “ door-to-balloon” (D-B) time, but restoration of flow in the epicardial infarct-related artery occurs promptly after PCI. At the bottom are shown a variety of methods for speeding the time to reperfusion along with the goals for the time intervals for the various components of the time delay. (Adapted from Cannon CP, Antman EM, Walls R, Braunwald E: Time as adjunctive agent to thrombolytic therapy. J Thromb Thrombolysis 1:27, 1994.)
- 43. Thank You!