Electronegativity, Bond Type and Drawing Lewis Structures
•Télécharger en tant que PPT, PDF•
1 j'aime•2,339 vues
Great graphics depicting how electronegativity affects bond type and Lewis structure of compounds, including a table comparing/contrasting ionic, covalent and metallic bonds with a guided notes, blank chart for students.
Signaler
Partager
Signaler
Partager
Recommandé
Recommandé
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...christianmathematics
Contenu connexe
Plus de Emma Wise
Plus de Emma Wise (20)
Dernier
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...christianmathematics
Dernier (20)
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...
IGNOU MSCCFT and PGDCFT Exam Question Pattern: MCFT003 Counselling and Family...

IGNOU MSCCFT and PGDCFT Exam Question Pattern: MCFT003 Counselling and Family...
Disha NEET Physics Guide for classes 11 and 12.pdf

Disha NEET Physics Guide for classes 11 and 12.pdf
Z Score,T Score, Percential Rank and Box Plot Graph

Z Score,T Score, Percential Rank and Box Plot Graph
Interactive Powerpoint_How to Master effective communication

Interactive Powerpoint_How to Master effective communication
Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...

Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...
Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...

Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...
BAG TECHNIQUE Bag technique-a tool making use of public health bag through wh...

BAG TECHNIQUE Bag technique-a tool making use of public health bag through wh...
Call Girls in Dwarka Mor Delhi Contact Us 9654467111

Call Girls in Dwarka Mor Delhi Contact Us 9654467111
Electronegativity, Bond Type and Drawing Lewis Structures
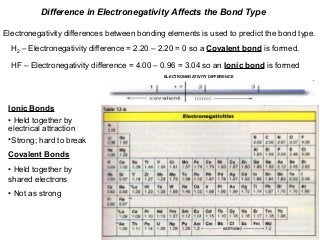
- 1. Difference in Electronegativity Affects the Bond Type Electronegativity differences between bonding elements is used to predict the bond type. H2 – Electronegativity difference = 2.20 – 2.20 = 0 so a Covalent bond is formed. HF – Electronegativity difference = 4.00 – 0.96 = 3.04 so an Ionic bond is formed ELECTRONEGATIVITY DIFFERENCE Ionic Bonds • Held together by electrical attraction •Strong; hard to break Covalent Bonds • Held together by shared electrons • Not as strong
- 2. 4 Step Process: Drawing Lewis Dot Structures Key consideration – The octet rule Step 1: Arrange atoms • Central atom lower electronegativity Step 2: Add up the valence electrons of all atoms Step 3: Draw single bonds • Subtract 2 electrons for each single bond from total counted in Step 2 Step 4: Deal out remaining electrons • Satisfy octet rule (except for Hydrogen)
- 4. Comparing and Contrasting Ionic and Covalent Bonds Ionic Bond Electrons Bond Electronegativity Differences State Melting Pt. Conductivity Water Solubility Covalent Bond
