Keto- Regulator of fat Metabolism
•
0 j'aime•11 vues
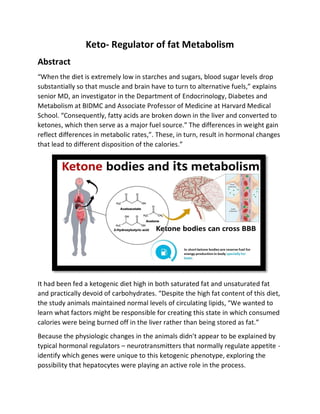
“When the diet is extremely low in starches and sugars, blood sugar levels drop substantially so that muscle and brain have to turn to alternative fuels,” explains senior MD, an investigator in the Department of Endocrinology, Diabetes and Metabolism at BIDMC and Associate Professor of Medicine at Harvard Medical School. “Consequently, fatty acids are broken down in the liver and converted to ketones, which then serve as a major fuel source.” The differences in weight gain reflect differences in metabolic rates,”. These, in turn, result in hormonal changes that lead to different disposition of the calories.”
Signaler
Partager
Signaler
Partager
Télécharger pour lire hors ligne
Recommandé
Recommandé
benefits of ketogenic diet247 ketogenic diet and its role in eliminating 

247 ketogenic diet and its role in eliminating GGS Medical College/Baba Farid Univ.of Health Sciences.
Contenu connexe
Similaire à Keto- Regulator of fat Metabolism
benefits of ketogenic diet247 ketogenic diet and its role in eliminating 

247 ketogenic diet and its role in eliminating GGS Medical College/Baba Farid Univ.of Health Sciences.
Similaire à Keto- Regulator of fat Metabolism (14)
A ketogenic diet to lose weight and fight metabolic disease

A ketogenic diet to lose weight and fight metabolic disease
Plus de AleenaKhan778846
Plus de AleenaKhan778846 (20)
Dernier
Falcon stands out as a top-tier P2P Invoice Discounting platform in India, bridging esteemed blue-chip companies and eager investors. Our goal is to transform the investment landscape in India by establishing a comprehensive destination for borrowers and investors with diverse profiles and needs, all while minimizing risk. What sets Falcon apart is the elimination of intermediaries such as commercial banks and depository institutions, allowing investors to enjoy higher yields.Unveiling Falcon Invoice Discounting: Leading the Way as India's Premier Bill...

Unveiling Falcon Invoice Discounting: Leading the Way as India's Premier Bill...Falcon Invoice Discounting
Dernier (20)
Berhampur 70918*19311 CALL GIRLS IN ESCORT SERVICE WE ARE PROVIDING

Berhampur 70918*19311 CALL GIRLS IN ESCORT SERVICE WE ARE PROVIDING
Uneak White's Personal Brand Exploration Presentation

Uneak White's Personal Brand Exploration Presentation
Lundin Gold - Q1 2024 Conference Call Presentation (Revised)

Lundin Gold - Q1 2024 Conference Call Presentation (Revised)
Horngren’s Cost Accounting A Managerial Emphasis, Canadian 9th edition soluti...

Horngren’s Cost Accounting A Managerial Emphasis, Canadian 9th edition soluti...
Berhampur CALL GIRL❤7091819311❤CALL GIRLS IN ESCORT SERVICE WE ARE PROVIDING

Berhampur CALL GIRL❤7091819311❤CALL GIRLS IN ESCORT SERVICE WE ARE PROVIDING
Kalyan Call Girl 98350*37198 Call Girls in Escort service book now

Kalyan Call Girl 98350*37198 Call Girls in Escort service book now
How to Get Started in Social Media for Art League City

How to Get Started in Social Media for Art League City
Unveiling Falcon Invoice Discounting: Leading the Way as India's Premier Bill...

Unveiling Falcon Invoice Discounting: Leading the Way as India's Premier Bill...
UAE Bur Dubai Call Girls ☏ 0564401582 Call Girl in Bur Dubai

UAE Bur Dubai Call Girls ☏ 0564401582 Call Girl in Bur Dubai
Quick Doctor In Kuwait +2773`7758`557 Kuwait Doha Qatar Dubai Abu Dhabi Sharj...

Quick Doctor In Kuwait +2773`7758`557 Kuwait Doha Qatar Dubai Abu Dhabi Sharj...
JAJPUR CALL GIRL ❤ 82729*64427❤ CALL GIRLS IN JAJPUR ESCORTS

JAJPUR CALL GIRL ❤ 82729*64427❤ CALL GIRLS IN JAJPUR ESCORTS
Falcon Invoice Discounting: Unlock Your Business Potential

Falcon Invoice Discounting: Unlock Your Business Potential
Call 7737669865 Vadodara Call Girls Service at your Door Step Available All Time

Call 7737669865 Vadodara Call Girls Service at your Door Step Available All Time
Nashik Call Girl Just Call 7091819311 Top Class Call Girl Service Available

Nashik Call Girl Just Call 7091819311 Top Class Call Girl Service Available
GUWAHATI 💋 Call Girl 9827461493 Call Girls in Escort service book now

GUWAHATI 💋 Call Girl 9827461493 Call Girls in Escort service book now
Getting Real with AI - Columbus DAW - May 2024 - Nick Woo from AlignAI

Getting Real with AI - Columbus DAW - May 2024 - Nick Woo from AlignAI
Marel Q1 2024 Investor Presentation from May 8, 2024

Marel Q1 2024 Investor Presentation from May 8, 2024
Keto- Regulator of fat Metabolism
- 1. Keto- Regulator of fat Metabolism Abstract “When the diet is extremely low in starches and sugars, blood sugar levels drop substantially so that muscle and brain have to turn to alternative fuels,” explains senior MD, an investigator in the Department of Endocrinology, Diabetes and Metabolism at BIDMC and Associate Professor of Medicine at Harvard Medical School. “Consequently, fatty acids are broken down in the liver and converted to ketones, which then serve as a major fuel source.” The differences in weight gain reflect differences in metabolic rates,”. These, in turn, result in hormonal changes that lead to different disposition of the calories.” It had been fed a ketogenic diet high in both saturated fat and unsaturated fat and practically devoid of carbohydrates. “Despite the high fat content of this diet, the study animals maintained normal levels of circulating lipids, “We wanted to learn what factors might be responsible for creating this state in which consumed calories were being burned off in the liver rather than being stored as fat.” Because the physiologic changes in the animals didn't appear to be explained by typical hormonal regulators – neurotransmitters that normally regulate appetite - identify which genes were unique to this ketogenic phenotype, exploring the possibility that hepatocytes were playing an active role in the process.
- 2. It using microarray gene analysis, they discovered that their hunch was correct: FGF21, a liver-derived fibroblast growth factor gene, was significantly increased in the mice that had been fed ketogenic diets. “FGF21 had previously been identified as a potential metabolic regulator by scientists at Eli Lilly, who showed that transgenic mice that overexpressed FGF21 were protected from diet-induced obesity, had smaller fat cells and had more brown adipose tissue, "But little was actually known about FGF21's physiologic roles. Working with Jeffrey Flier's lab, we were able to show that FGF21 is essential for fatty acid oxidation.” Circulating/ Regulating Furthermore, when FGF21 was inhibited, the mice developed a massive accumulation of fat in the liver and an extreme increase in circulating lipids. A second study by Maratos-Flier and colleagues published in the June 2007 issue of the American Journal of Physiology further elucidates the unique metabolic changes that occur with the consumption of a ketogenic diet. “Although the purpose of both of these studies was to glean insights into metabolic physiology, our findings suggest that increased levels of FGF21 may be a potential mechanism behind low-carbohydrate diets' beneficial properties when it comes to lipid metabolism,” says Maratos-Flier. “Diets that limit carbohydrates and eliminate trans fats, and at the same time emphasize fiber and good fats, appear to be healthiest, especially among individuals who are predisposed to developing diabetes.” Metabolic Pathways Modifications in the macronutrient composition of diets or food components have been shown to affect hormones, metabolic pathways, gene expression, and appetite control. Several studies have provided evidence for the appetite suppressant effect of ketogenic diets and shown that for as long as participants are in a state of ketosis, appetite does not
- 3. increase, despite weight loss. It has been consistently shown by our research group and others that under ketogenic conditions, feelings of hunger, measured using validated visual analogue scales (VASs), do not increase, even when massive weight loss is achieved (up to 17% or initial body weight). Studies where hunger feelings have been evaluated through other methods, such as the Three Factor Eating Questionnaire [14], the Food Craving Questionnaire [15], Non validated questionnaires, semi- structured interviews, [19] and case reports [20,21] have also confirmed that ketogenic diets are associated with diminished or absent feelings of hunger, a reduced desire to eat and decreased overall appetite. From our knowledge, all the available evidence shows that the secretion of the hunger hormone ghrelin, which is upregulated in response to diet- induced weight loss, is blunted under ketogenic conditions. Moreover, once participants are no longer in ketosis, upon refeeding and re-introduction of CHO, the expected surge in ghrelin secretion and hunger feelings above baseline levels is seen. The effect of ketogenic diets on the release of satiety hormones, namely cholecystokinin (CCK), glucagon-like peptide-1 (GLP-1) and peptide YY, remains unfortunately inconclusive. Our group has found no changes in the postprandial release of these satiety peptides with a 17% weight loss induced using VLED, independently of the participants being in or out of ketosis.
- 4. Whether appetite responses to ketosis differ between males and females is yet to be fully determined. However, our research group found that the basal and postprandial secretion of GLP-1 in response to weight loss induced by a VLED was modulated by sex, with a decrease in basal GLP-1 concentrations seen only in males, and an increase in postprandial concentration of GLP-1 happening only in females. The threshold of nutritionally induced ketosis needed to achieve appetite suppression using ketogenic diets has yet to be determined. However, the results from the systematic review and meta-analysis performed by Gibson and colleagues indicate that a restriction of CHO below 50 g/day does not appear to be necessary. This raises the question of whether such a restriction should be applied in ketogenic diets, because the severity of the CHO restriction is counterintuitive to healthy eating recommendations. Entire core food groups, such as whole grains, legumes, reduced-fat dairy, fruits, and vegetables need to be severely constrained or eliminated due to the need to significantly reduce CHO intake. This restriction and elimination may have a negative impact on bone health and increase the risk of developing certain types of cancer and cardiovascular disease. Furthermore, in addition to the CHO restriction, fiber intake is also reduced in ketogenic diets and likely explains why constipation is one of the most commonly reported side effects of these diets. Given that high amounts of circulating ketones have been seen in participants who followed diets with a CHO intake of up to 192 g/day, a greater allowance of CHO in ketogenic diets might and should be considered in future formulations. The literature on this topic seems to
- 5. suggest that 100 g/day of CHO should be the cut off for ketogenic diets. A more liberal CHO allowance can, for instance, enable the incorporation of CHO-rich vegetables that are higher in fiber content pivotal to alleviate constipation in those undergoing ketogenic diets and allow for the consumption of calcium-containing dairy products with fortified vitamin D and calcium to hinder bone loss that has been seen as a long-term side effect of VLED. Conclusion: Harvey and colleagues reported recently that equicaloric diets containing up to 25% total energy from CHO can result in mean ßHB ≥ 0.5 mmol/L, with no clinically relevant differences in symptoms of CHO withdrawal between diets containing from 5% to 25% total energy from CHO [9].