Key Word Definition Card Sort
•Télécharger en tant que DOC, PDF•
1 j'aime•470 vues
For Science Teachers
Signaler
Partager
Signaler
Partager
Contenu connexe
Tendances
Tendances (14)
35 ppt v2_chemistry_10_metals and non-metals basic physical and chemical prop...

35 ppt v2_chemistry_10_metals and non-metals basic physical and chemical prop...
En vedette
En vedette (20)
CRC Network | Implications of the CRC for the Public Sector

CRC Network | Implications of the CRC for the Public Sector
Reusing historical newspapers of KB in e-humanities - Case studies and exampl...

Reusing historical newspapers of KB in e-humanities - Case studies and exampl...
Similaire à Key Word Definition Card Sort
Junior cycle science chemistry metals. By Theresa Lowry-Lehnen. Science Teacher.

Junior cycle science chemistry metals. By Theresa Lowry-Lehnen. Science Teacher.Theresa Lowry-Lehnen
Similaire à Key Word Definition Card Sort (20)
Reaction of metals and non metals with water,Introduction to reactivity serie...

Reaction of metals and non metals with water,Introduction to reactivity serie...
Core & Extension Metals I Reactivity Series & Redox.pptx

Core & Extension Metals I Reactivity Series & Redox.pptx
Ncert class 10 - science - chapter 3 - metals and non-metals

Ncert class 10 - science - chapter 3 - metals and non-metals
Junior cycle science chemistry metals. By Theresa Lowry-Lehnen. Science Teacher.

Junior cycle science chemistry metals. By Theresa Lowry-Lehnen. Science Teacher.
Plus de amr hassaan
Plus de amr hassaan (20)
Frio invierno هكذا يكون الشتاء الروسي russian winter

Frio invierno هكذا يكون الشتاء الروسي russian winter
الارض الفضاء - الكون - من اجمل ماترى فى حياتك - اعرف حجم همومك الحقيقي

الارض الفضاء - الكون - من اجمل ماترى فى حياتك - اعرف حجم همومك الحقيقي
Dernier
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"National Information Standards Organization (NISO)
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"National Information Standards Organization (NISO)
APM Welcome, APM North West Network Conference, Synergies Across Sectors

APM Welcome, APM North West Network Conference, Synergies Across SectorsAssociation for Project Management
Dernier (20)
9548086042 for call girls in Indira Nagar with room service

9548086042 for call girls in Indira Nagar with room service
Beyond the EU: DORA and NIS 2 Directive's Global Impact

Beyond the EU: DORA and NIS 2 Directive's Global Impact
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"
social pharmacy d-pharm 1st year by Pragati K. Mahajan

social pharmacy d-pharm 1st year by Pragati K. Mahajan
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"
Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...

Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...
Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...

Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...
APM Welcome, APM North West Network Conference, Synergies Across Sectors

APM Welcome, APM North West Network Conference, Synergies Across Sectors
Disha NEET Physics Guide for classes 11 and 12.pdf

Disha NEET Physics Guide for classes 11 and 12.pdf
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
Interactive Powerpoint_How to Master effective communication

Interactive Powerpoint_How to Master effective communication
Key Word Definition Card Sort
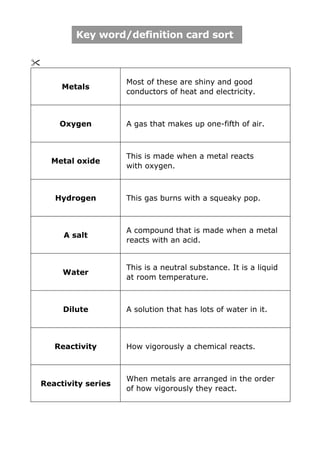
- 1. Key word/definition card sort Most of these are shiny and good Metals conductors of heat and electricity. Oxygen A gas that makes up one-fifth of air. This is made when a metal reacts Metal oxide with oxygen. Hydrogen This gas burns with a squeaky pop. A compound that is made when a metal A salt reacts with an acid. This is a neutral substance. It is a liquid Water at room temperature. Dilute A solution that has lots of water in it. Reactivity How vigorously a chemical reacts. When metals are arranged in the order Reactivity series of how vigorously they react.
- 2. Metals and metal salt solutions What to do 1 Put small samples of each 2 Add 3–5 drops of zinc metal in the wells of a sulphate solution to each spotting tile, as shown below. metal in row 1. 3 Add 3–5 drops of iron 4 Add 3–5 drops of copper sulphate solution to each sulphate solution to each metal in row 2. metal in row 3. 5 Add 3–5 drops of magnesium 6 For each well decide whether nitrate solution to each metal or not a chemical reaction in row 4. has happened. Write down your results.
- 3. Metals and metal salt solutions Put a tick in the boxes where you did see signs of a reaction. Put a cross in the boxes where you did not see signs of a reaction. Magnesium Iron Copper Zinc Zinc sulphate solution Iron sulphate solution Copper sulphate solution Magnesiu m nitrate solution Cross out the words in bold that are wrong: Magnesium is the most reactive metal that we used. It did/did not react with solutions of the salts of zinc, iron and copper. Copper is the least reactive metal that we used. It did/did not react with solutions of the salts of magnesium, zinc and iron. Metals react with the salts of metals that are more/less reactive than they are.
