cfDNA Screening for Fetal Aneuploidy- Positive predictive value
•
1 j'aime•834 vues
Cell-free DNA Screening for Fetal Aneuploidy, Positive predictive value, aneuploidy , screening, pregnancy, NIPT, Negative Predictive Value, Non-invasive screening,
Signaler
Partager
Signaler
Partager
Télécharger pour lire hors ligne
Recommandé
Evolution of molecular prognostic testing in ER positive breast cancerEvolution of molecular prognostic testing in ER positive breast cancer

Evolution of molecular prognostic testing in ER positive breast cancerBell Symposium & MSP Seminar
Recommandé
Evolution of molecular prognostic testing in ER positive breast cancerEvolution of molecular prognostic testing in ER positive breast cancer

Evolution of molecular prognostic testing in ER positive breast cancerBell Symposium & MSP Seminar
Gene Profiling in Clinical Oncology - Slide 10 - H. Rugo - Why genomic tools ...

Gene Profiling in Clinical Oncology - Slide 10 - H. Rugo - Why genomic tools ...European School of Oncology
Gene Profiling in Clinical Oncology - Slide 9 - F. André - Genomic evaluation...

Gene Profiling in Clinical Oncology - Slide 9 - F. André - Genomic evaluation...European School of Oncology
Pranešimas XVI kompiuterininkų konferencijos sekcijoje „Duomenų tyryba ir jos taikymai“,
„Kompiuterininkų dienos – 2013“, Šiauliai 2013-09-20
Niakšu, Olegas ; Kurasova, Olga ; Gedminaitė, Jurgita „Duomenų tyryba BRCA1 g...

Niakšu, Olegas ; Kurasova, Olga ; Gedminaitė, Jurgita „Duomenų tyryba BRCA1 g...Lietuvos kompiuterininkų sąjunga
Contenu connexe
Tendances
Gene Profiling in Clinical Oncology - Slide 10 - H. Rugo - Why genomic tools ...

Gene Profiling in Clinical Oncology - Slide 10 - H. Rugo - Why genomic tools ...European School of Oncology
Gene Profiling in Clinical Oncology - Slide 9 - F. André - Genomic evaluation...

Gene Profiling in Clinical Oncology - Slide 9 - F. André - Genomic evaluation...European School of Oncology
Pranešimas XVI kompiuterininkų konferencijos sekcijoje „Duomenų tyryba ir jos taikymai“,
„Kompiuterininkų dienos – 2013“, Šiauliai 2013-09-20
Niakšu, Olegas ; Kurasova, Olga ; Gedminaitė, Jurgita „Duomenų tyryba BRCA1 g...

Niakšu, Olegas ; Kurasova, Olga ; Gedminaitė, Jurgita „Duomenų tyryba BRCA1 g...Lietuvos kompiuterininkų sąjunga
Tendances (20)
Gene Profiling in Clinical Oncology - Slide 10 - H. Rugo - Why genomic tools ...

Gene Profiling in Clinical Oncology - Slide 10 - H. Rugo - Why genomic tools ...
Beyond BRCA Mutations: What's New in the World of Genetic Testing?

Beyond BRCA Mutations: What's New in the World of Genetic Testing?
Gene Profiling in Clinical Oncology - Slide 9 - F. André - Genomic evaluation...

Gene Profiling in Clinical Oncology - Slide 9 - F. André - Genomic evaluation...
Efrat Levy Lahad : Genetic testing for breast and ovarian cancer

Efrat Levy Lahad : Genetic testing for breast and ovarian cancer
Niakšu, Olegas ; Kurasova, Olga ; Gedminaitė, Jurgita „Duomenų tyryba BRCA1 g...

Niakšu, Olegas ; Kurasova, Olga ; Gedminaitė, Jurgita „Duomenų tyryba BRCA1 g...
Molecular testing and ihc surrogates for breast carcinoma

Molecular testing and ihc surrogates for breast carcinoma
En vedette (7)
Top 5 Deep Learning and AI Stories - October 6, 2017

Top 5 Deep Learning and AI Stories - October 6, 2017
Similaire à cfDNA Screening for Fetal Aneuploidy- Positive predictive value
UOG Journal Club: Maternal blood cell-free DNA testing in early screening for...

UOG Journal Club: Maternal blood cell-free DNA testing in early screening for...International Society of Ultrasound in Obstetrics and Gynecology (ISUOG)
Analysis of cell-free DNA in maternal blood in screening for fetal aneuploidies: updated meta-analysis
M. M. Gil, M. S. Quezada, R. Revello, R. Akolekar and K. H. Nicolaides
Volume 45, Issue 3, pages 249–266, March 2015
http://onlinelibrary.wiley.com/doi/10.1002/uog.14791/fullUOG Journal Club: Analysis of cell-free DNA in maternal blood in screening fo...

UOG Journal Club: Analysis of cell-free DNA in maternal blood in screening fo...International Society of Ultrasound in Obstetrics and Gynecology (ISUOG)
NY Prostate Cancer Conference - S. Stone - Session 1: Cell cycle progression ...

NY Prostate Cancer Conference - S. Stone - Session 1: Cell cycle progression ...European School of Oncology
Similaire à cfDNA Screening for Fetal Aneuploidy- Positive predictive value (20)
Discordant noninvasive prenatal testing & cytogenetic results: 109 consecutiv...

Discordant noninvasive prenatal testing & cytogenetic results: 109 consecutiv...
Diagnóstico prenatal no invasivo en sangre materna

Diagnóstico prenatal no invasivo en sangre materna
Cell free fetal dna testing for prenatal diagnosis

Cell free fetal dna testing for prenatal diagnosis
UOG Journal Club: Maternal blood cell-free DNA testing in early screening for...

UOG Journal Club: Maternal blood cell-free DNA testing in early screening for...
ACOG screening for fetal chromosomal abnormalities 2020

ACOG screening for fetal chromosomal abnormalities 2020
20160918 - F. Prefumo - Ecografia, screening biochimico, NIPT e diagnosi pren...

20160918 - F. Prefumo - Ecografia, screening biochimico, NIPT e diagnosi pren...
UOG Journal Club: Analysis of cell-free DNA in maternal blood in screening fo...

UOG Journal Club: Analysis of cell-free DNA in maternal blood in screening fo...
Cf dna for fetal aneuploidy risk assessment toma sept 2015 usb

Cf dna for fetal aneuploidy risk assessment toma sept 2015 usb
NY Prostate Cancer Conference - S. Stone - Session 1: Cell cycle progression ...

NY Prostate Cancer Conference - S. Stone - Session 1: Cell cycle progression ...
Practice Bulletin #226, Screening for Chromosomal Abnormalities

Practice Bulletin #226, Screening for Chromosomal Abnormalities
Plus de Asha Reddy
Plus de Asha Reddy (20)
LIKELIHOOD RATIOS -Agathokleous 2013 Meta-analysis 2nd trimester T 21.

LIKELIHOOD RATIOS -Agathokleous 2013 Meta-analysis 2nd trimester T 21.
Dindigul district cervical screening study, india acceptability, effectivenes...

Dindigul district cervical screening study, india acceptability, effectivenes...
Repeated failed non-invasive prenatal testing in autoimmune disease owing to...

Repeated failed non-invasive prenatal testing in autoimmune disease owing to...
A case of repeated failed non-invasive prenatal testing in a woman with immun...

A case of repeated failed non-invasive prenatal testing in a woman with immun...
Recurrent Third-Trimester Fetal Loss and Maternal Mosaicism for Long-QT Syndrome

Recurrent Third-Trimester Fetal Loss and Maternal Mosaicism for Long-QT Syndrome
Antiphospholipid syndrome - ACOG 2015 Recommendations for Heparin

Antiphospholipid syndrome - ACOG 2015 Recommendations for Heparin
Recommended Thromboprophylaxis for Pregnancies Complicated by Inherited Throm...

Recommended Thromboprophylaxis for Pregnancies Complicated by Inherited Throm...
Human Milk Banking Guidelines -INDIAN ACADEMY OF PEDIATRICS

Human Milk Banking Guidelines -INDIAN ACADEMY OF PEDIATRICS
Suture ligation of supernumerary digits and tags :An outmoded practice

Suture ligation of supernumerary digits and tags :An outmoded practice
Dernier
Dernier (20)
Call Girls Jabalpur Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Jabalpur Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Gwalior Just Call 8617370543 Top Class Call Girl Service Available

Call Girls Gwalior Just Call 8617370543 Top Class Call Girl Service Available
Call Girls Dehradun Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Dehradun Just Call 9907093804 Top Class Call Girl Service Available
Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...

Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...
Call Girls Haridwar Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Haridwar Just Call 8250077686 Top Class Call Girl Service Available
Top Rated Hyderabad Call Girls Erragadda ⟟ 6297143586 ⟟ Call Me For Genuine ...

Top Rated Hyderabad Call Girls Erragadda ⟟ 6297143586 ⟟ Call Me For Genuine ...
Call Girls Bangalore Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Bangalore Just Call 8250077686 Top Class Call Girl Service Available
VIP Hyderabad Call Girls Bahadurpally 7877925207 ₹5000 To 25K With AC Room 💚😋

VIP Hyderabad Call Girls Bahadurpally 7877925207 ₹5000 To 25K With AC Room 💚😋
Call Girls Nagpur Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Nagpur Just Call 9907093804 Top Class Call Girl Service Available
Call Girls Bhubaneswar Just Call 9907093804 Top Class Call Girl Service Avail...

Call Girls Bhubaneswar Just Call 9907093804 Top Class Call Girl Service Avail...
All Time Service Available Call Girls Marine Drive 📳 9820252231 For 18+ VIP C...

All Time Service Available Call Girls Marine Drive 📳 9820252231 For 18+ VIP C...
VIP Service Call Girls Sindhi Colony 📳 7877925207 For 18+ VIP Call Girl At Th...

VIP Service Call Girls Sindhi Colony 📳 7877925207 For 18+ VIP Call Girl At Th...
(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...

(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...
Call Girls Cuttack Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Cuttack Just Call 9907093804 Top Class Call Girl Service Available
Bangalore Call Girls Nelamangala Number 9332606886 Meetin With Bangalore Esc...

Bangalore Call Girls Nelamangala Number 9332606886 Meetin With Bangalore Esc...
Night 7k to 12k Chennai City Center Call Girls 👉👉 7427069034⭐⭐ 100% Genuine E...

Night 7k to 12k Chennai City Center Call Girls 👉👉 7427069034⭐⭐ 100% Genuine E...
💎VVIP Kolkata Call Girls Parganas🩱7001035870🩱Independent Girl ( Ac Rooms Avai...

💎VVIP Kolkata Call Girls Parganas🩱7001035870🩱Independent Girl ( Ac Rooms Avai...
Call Girls Aurangabad Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Aurangabad Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Ooty Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Ooty Just Call 8250077686 Top Class Call Girl Service Available
Pondicherry Call Girls Book Now 9630942363 Top Class Pondicherry Escort Servi...

Pondicherry Call Girls Book Now 9630942363 Top Class Pondicherry Escort Servi...
cfDNA Screening for Fetal Aneuploidy- Positive predictive value
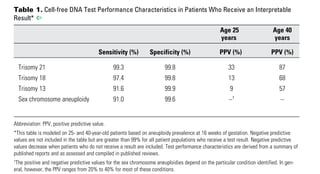
- 1. Committee Opinion No. 640 3 than trisomies 13, 18, or 21. One study of women with abnormal traditional screening test results who had diagnostic testing estimated that up to 17% of clini- cally significant chromosomal abnormalities would not be detectable with most of the current cell-free DNA techniques (23). Given the performance of conventional screening methods and the limitations of cell-free DNA, conventional screening methods remain the most appro- priate choice for first-line screening for most women in the general obstetric population. Testing for Other Genetic Conditions With Cell-free DNA Although all laboratories that currently offer cell-free DNA screening for aneuploidy include trisomies 13, 18, and 21 as part of their standard panel, the approach to the sex chromosomes and other chromosome abnormali- ties vary. Some laboratories offer routine sex chromo- some, microdeletion, and rare trisomy (eg, trisomy 16 or trisomy 22) assessment, whereas others require that sex chromosome and other assessments be requested in order for those results to be reported. Microdeletion syndromes occur sporadically or are due to other genetic Use in the General Obstetric Population Data on the performance of cell-free DNA testing in the general obstetric population have become available (1, 8, 11, 16, 17). The sensitivity and specificity in the general obstetric population are similar to the levels previously published for the aforementioned high-risk population. The positive predictive value, however, is lower in this population, given the lower prevalence of aneuploidy in the general obstetric population. That is, fewer women with a positive test result will actually have an affected fetus, and there will be more false-positive test results (Fig. 1). Another limitation of cell-free DNA screening in the general obstetric population is that trisomies 13, 18, and 21 comprise a smaller proportion of the chromosome abnormalities found in the general obstetric population (21–23). Traditional serum analyte screening methods allow for higher detection rates of these other chromo- some abnormalities as well as the risk of other adverse pregnancy outcomes. For example, a positive integrated screening test result may indirectly identify a fetus with an unbalanced rearrangement of a chromosome other Table 1. Cell-free DNA Test Performance Characteristics in Patients Who Receive an Interpretable Result* ^ Age 25 Age 40 years years Sensitivity (%) Specificity (%) PPV (%) PPV (%) Trisomy 21 99.3 99.8 33 87 Trisomy 18 97.4 99.8 13 68 Trisomy 13 91.6 99.9 9 57 Sex chromosome aneuploidy 91.0 99.6 --† -- Abbreviation: PPV, positive predictive value. *This table is modeled on 25- and 40-year-old patients based on aneuploidy prevalence at 16 weeks of gestation. Negative predictive values are not included in the table but are greater than 99% for all patient populations who receive a test result. Negative predictive values decrease when patients who do not receive a result are included. Test performance characteristics are derived from a summary of published reports and as assessed and compiled in published reviews. † The positive and negative predictive values for the sex chromosome aneuploidies depend on the particular condition identified. In gen- eral, however, the PPV ranges from 20% to 40% for most of these conditions. Applicability to clinical practice: Positive predictive value (defined as true positives divided by true positives plus false positives) is directly related to the prevalence of the condition in the population screened. Based on the sensitivity and specificity of the test, when a population with an overall preva- lence of 1/1,000 for trisomy 21 is screened, the positive predictive value of an abnormal result is 33%—only one in three women who get an abnormal result will have an affected fetus. If the prevalence is 1/75, the positive predictive value is 87%. Data from Gil MM, Quezada MS, Revello R, Akolekar R, Nicolaides KH. Analysis of cell-free DNA in maternal blood in screening for fetal aneuploidies: updated meta-analysis. Ultrasound Obstet Gynecol 2015;45:249–66; Porreco RP, Garite TJ, Maurel K, Marusiak B, Ehrich M, van den Boom D, et al. Noninvasive prenatal screening for fetal trisomies 21, 18, 13 and the common sex chromosome aneuploidies from maternal blood using massively parallel genomic sequencing of DNA. Obstetrix Collaborative Research Network. Am J Obstet Gynecol 2014;211:365.e1–365.12; Snijders RJ, Sebire NJ, Nicolaides KH. Maternal age and gestational age-specific risk for chromosomal defects. Fetal Diagn Ther 1995;10:356–67; Benn P, Cuckle H, Pergament E. Non-invasive prenatal testing for aneuploidy: current status and future prospects. Ultrasound Obstet Gynecol 2013;42:15–33; and Verweij EJ, de Boer MA, Oepkes D. Non-invasive prenatal testing for trisomy 13: more harm than good? Ultrasound Obstet Gynecol 2014;44:112–4.
