2016 BDSRA Gray & Rozenberg CLN1
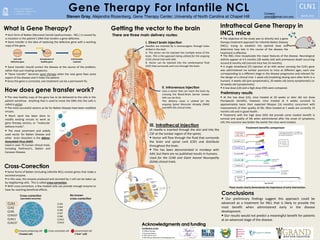
- 1. Each form of Batten (Neuronal Ceroid Lipofuscinoses - NCL-) is caused by a mutation in the patient’s DNA that renders a gene defective. Gene transfer is the idea of replacing the defective gene with a working copy of the gene. The INCL mice recapitulate the major features of the disease. Neurological deficits appear at 4.5 months (20 weeks old) with premature death occurring around 8 months old (normal mice live 24 months). A single intrathecal (IT) injection of an AAV vector carrying the CLN1 gene was administered via lumbar puncture to mice at different ages, each one corresponding to a different stage in the disease progression and relevant for the design of a clinical trial: 1 week old (modeling dosing soon after birth in a human), 4 weeks old (pre-symptomatic), 20 weeks old (early-symptomatic) or 26 weeks old (symptomatic). A low dose (LD) and a high dose (HD) were compared. I. Direct brain injection Needles are inserted by a neurosurgeon through holes drilled in the skull. A. The vector can be injected into multiple areas of the brain. This is the approach being utilized for the ongoing CLN2 clinical trial with AAV. B. Vector can be injected into the cerebrospinal fluid (CSF) that surrounds and runs through the brain. II. Intravenous injection Uses a vector that can reach the brain by crossing the Blood-Brain barrier (newer AAV can do this). This delivery route is utilized for the ongoing Spinal Muscular Atrophy (SMA) and MPS IIIA clinical trials. Getting the vector to the brain Cross-Correction Some forms of Batten (including Infantile NCL) involve genes that make a secreted enzyme. In this case, the enzyme produced and secreted by 1 cell can be taken up by neighboring cells. This is called cross-correction. With cross-correction, a few treated cells can provide enough enzyme to have far-reaching beneficial effects. Cross-correction (secreted enzyme) No known cross-correction CLN1 CLN2 CLN5 CLN10? CLN11 CLN13? CLN3 CLN4 CLN6 CLN7 CLN8 CLN12 CLN14 Enzyme producing cell (Treated cell) Cross-corrected cell Uncorrected cell (“Sick” cell) At the low dose (LD), mice treated at 20 weeks or later did not show therapeutic benefits; however, mice treated at 4 weeks survived to approximately twice their expected lifespan (16 months) concurrent with improvement of their quality of life. Mice treated at 1 week are currently 15 months old and in good health. Treatment with the high dose (HD) did provide some modest benefit in survival and quality of life when administered after the onset of symptoms; still, the outcome was better the earlier the mice were treated. Preliminary results Our preliminary findings suggest this approach could be advanced as a treatment for INCL that is likely to provide the most benefit when administered early in the disease development. Our results would not predict a meaningful benefit for patients at an advanced stage of the disease. Gene Therapy For Infantile NCL Steven Gray, Alejandra Rozenberg, Gene Therapy Center, University of North Carolina at Chapel Hill What is Gene Therapy? The new healthy copy of the gene has to be delivered to the cells in the patient somehow. Anything that is used to move the DNA into the cells is called a vector. The most successful vectors so far for Batten Disease have been modified viruses. The most prominent and widely used vector for Batten Disease and other brain disorders is the Adeno- Associated Virus (AAV). Used in over 75 human clinical trials, including Parkinson’s, Batten and Canavan Disease. How does gene transfer work? Contact : graysj@email.unc.edu Acknowledgments and funding Clifford Heindel Courtney Santos CLN1 BDSRA 2016 Intrathecal Gene Therapy in INCL mice The objective of the study was to directly test a gene therapy treatment approach for Infantile Batten Disease (INCL), trying to establish the optimal dose and determine how late in the course of the disease the treatment is effective. Conclusions There are three main delivery routes: Gene transfer should correct the disease at the source of the problem, rather than just treating symptoms. “Gene transfer” becomes gene therapy when the new gene fixes some aspect of the disease and it helps the patient. Since the gene is corrected, one treatment can be a permanent fix. 0 4 8 12 16 20 24 DeathBirth Onset of symptoms Onset of symptoms DeathBirth Death Currently alive (11 months) Birth Onset of symptoms DeathBirth Onset of symptoms DeathBirth Onset of symptoms Birth Onset of symptoms Birth Currently alive (15 months) and healthy Birth Death Untreated INCL mice Normal mice 26 weeks- LD IT injection LD Low Dose HD High Dose Age (months) 1 week-LD 4 weeks-LD 20 weeks-LD 26 weeks-HD 20 weeks-HD * Schematic representation. 1 week-HD and 4 weeks-HD treatments not shown (mice still too young to see benefits). Treatment benefits comparison* Much work has been done to modify existing viruses to work as gene therapy vectors, or “molecular delivery trucks”. cell makes protein using new gene These results clearly demonstrate the importance of early intervention. Cerebrospinal fluid Cell with defective gene Introduction of healthy gene Cell function restored UNC Vector Core UNC Mouse Behavioral Phenotyping Core Contributors at UNC III. Intrathecal injection (A needle is inserted through the skin and into the CSF at the lumbar region of the spine). Vector will flow through the fluid that surrounds the brain and spinal cord (CSF) and distribute throughout the brain. This has been demonstrated in monkeys with AAV, but there are no published results in humans. Used for the CLN6 and Giant Axonal Neuropathy (GAN) clinical trials.
