Filter aids
- 1. Filter Aids - Devyani Joshi
- 2. • Mechanism of filtration basically involves a two step process ▫ 1. The filter paper itself resists the flow of solid material while permitting the passage of liquid. ▫ 2. During the course of filtration the suspended solid material builds up on the filter medium and thereby forms a filter bed which acts as a second and often more efficient filter medium.
- 3. • Rate of flow is directly proportional to (driving force) (cross sectional area) Resistance
- 4. Filter Paper • Filter paper is a semi- permeable paper barrier placed perpendicular to a liquid or air flow. It is used to separate fine solids from liquids or air • The important parameters are wet strength, porosity, particle retention, flow rate, compatibility, efficiency and capacity.
- 5. When possible first few milliliters of filtrate should be discarded to eliminate contamination of pharmaceutical product by fibers associated with most of the filter papers.
- 6. Types of laboratory filter papers 1.Qualitative Filter Papers • 100% cotton linter cellulose • pH tolerant: 0 to 12 • Thermostable: up to 120°C • Ash Content: 0.1% ▫ APPLICATIONS • Clarify and remove precipitates • Preparation for qualitative analysis
- 7. 2.Quantitative/Hardened Filter Papers • 100% cotton linter cellulose • Ash Content: 0.01% • Acid washed: Double acid washed in hydrochloric then hydrofluoric acid, then rinsed with ultrapure water to neutralize. ▫ APPLICATIONS • Gravimetric analysis • Environmental monitoring
- 8. 3.Chromatography Papers • High quality papers are carefully tested for spot formation, capillary action, water flow rate and absorption speed to assure uniformity and reproducibility • Better resolution with slower flow rate papers APPLICATIONS • Chromatography • Electrophoresis and blotting • Separation of heavily loaded solutes
- 10. Cotton filters ▫ A small pledged of absorbent cotton, loosely inserted in the neck of funnel, adequately serves to remove large particles of extraneous material from clear liquid. ▫ It is sometimes necessary to return the liquid a number of times to secure perfect transparency.
- 11. •Glass-wool filters ▫ When solutions of highly reactive chemicals such as strong acids, are to be filtered, filter paper cannot be used. In its place glass wool may be used. This material is resistant to ordinary chemical action, and when properly packed into the neck of funnel it constitutes very effective filtering medium.
- 12. • Sintered glass filters ▫ These filters have as a filtering medium a flat or convex plate consisting of particles of jenna glass powdered and sifted to produce granules of uniform size that are molded together. They vary in porosity depending on size of granules used in the plate. A vacuum attachment is necessary to facilitate the passage of liquid through the filter plate.
- 13. •Funnels ▫ They are conical shaped utensils widely used to support the filter media. Buchner funnel is largely used today. A piece of round filter paper is laid on the perforated porcelain diaphragm and the filtration is conducted. This funnel is specially applicable for vacuum filtration.
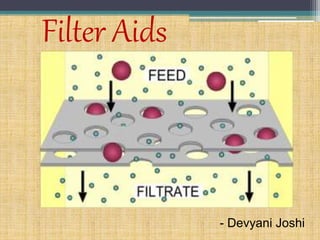
- 14. Aids to filtration • Addition of insoluble adsorbent powder to a liquid prior to filtration greatly increases the efficiency of the process. • Purified talc, siliceous earth (kieselgurh), clays, charcoal, paper pulp, magnesium carbonate, bentonite, silica gel have been used for this purpose.
- 15. • Talc- it is nonadsorbent to materials in solution and is chemically inert medium for filtering any liquid provided it is not impalpably fine variety that will pass through the filter paper. • Keiselgurh- It is almost pure silica (SiO2).It is as applicable as talc for general filtration purposes with no danger of removing active constituents by adsorption. • • Siliceous earths or clays-like fuller’s earth or kaolin in hydrated form which is produced when they are brought into contact with aqueous liquids are safe for general use only in filtering fixed oils.
- 16. • Charcoal- it possesses adsorptive properties not only towards colour but for many active constituents such as alkaloids or glycosides. Thus charcoal should never be used as filtering medium unless removal of such materials is desirable. • Chalk and magnesium carbonate- they react readily with acids and possess finite solubility in water and aqueous fluids. When they are added to alkaloidal preparation prior to filtration will precipitate and remove all the alkaloidal constituents.
