Cellular Energy pt.1
•Télécharger en tant que PPT, PDF•
1 j'aime•590 vues
Signaler
Partager
Signaler
Partager
Recommandé
Recommandé
Contenu connexe
Tendances
Tendances (20)
Chemical dynamics, intro,tst, lindemann theory by dr.y. s. thakare

Chemical dynamics, intro,tst, lindemann theory by dr.y. s. thakare
Chem 2 - Chemical Kinetics VIII: The Arrhenius Equation, Activation Energy, a...

Chem 2 - Chemical Kinetics VIII: The Arrhenius Equation, Activation Energy, a...
En vedette
En vedette (8)
Similaire à Cellular Energy pt.1
Similaire à Cellular Energy pt.1 (20)
Passive vs. Active transport.mp4.downloadPassive vs. Active tra

Passive vs. Active transport.mp4.downloadPassive vs. Active tra
Passive vs. Active transport.mp4.downloadPassive vs. Active tra.docx

Passive vs. Active transport.mp4.downloadPassive vs. Active tra.docx
Chapter 7Energy and Metabolism© Cengage Learning 2015.docx

Chapter 7Energy and Metabolism© Cengage Learning 2015.docx
Biological oxidation Bioenergetics and general concepts (part - I)

Biological oxidation Bioenergetics and general concepts (part - I)
UNIT1.2 bioenergetic in introduction of biochemistry

UNIT1.2 bioenergetic in introduction of biochemistry
BCM 201_Energetics of life and the functioning of living organisms

BCM 201_Energetics of life and the functioning of living organisms
Plus de Jolie Yu
Plus de Jolie Yu (20)
Dernier
https://app.box.com/s/7hlvjxjalkrik7fb082xx3jk7xd7liz3TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...Nguyen Thanh Tu Collection
https://app.box.com/s/x7vf0j7xaxl2hlczxm3ny497y4yto33i80 ĐỀ THI THỬ TUYỂN SINH TIẾNG ANH VÀO 10 SỞ GD – ĐT THÀNH PHỐ HỒ CHÍ MINH NĂ...

80 ĐỀ THI THỬ TUYỂN SINH TIẾNG ANH VÀO 10 SỞ GD – ĐT THÀNH PHỐ HỒ CHÍ MINH NĂ...Nguyen Thanh Tu Collection
Making communications land - Are they received and understood as intended? webinar
Thursday 2 May 2024
A joint webinar created by the APM Enabling Change and APM People Interest Networks, this is the third of our three part series on Making Communications Land.
presented by
Ian Cribbes, Director, IMC&T Ltd
@cribbesheet
The link to the write up page and resources of this webinar:
https://www.apm.org.uk/news/making-communications-land-are-they-received-and-understood-as-intended-webinar/
Content description:
How do we ensure that what we have communicated was received and understood as we intended and how do we course correct if it has not.Making communications land - Are they received and understood as intended? we...

Making communications land - Are they received and understood as intended? we...Association for Project Management
Dernier (20)
TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...
Beyond_Borders_Understanding_Anime_and_Manga_Fandom_A_Comprehensive_Audience_...

Beyond_Borders_Understanding_Anime_and_Manga_Fandom_A_Comprehensive_Audience_...
Python Notes for mca i year students osmania university.docx

Python Notes for mca i year students osmania university.docx
80 ĐỀ THI THỬ TUYỂN SINH TIẾNG ANH VÀO 10 SỞ GD – ĐT THÀNH PHỐ HỒ CHÍ MINH NĂ...

80 ĐỀ THI THỬ TUYỂN SINH TIẾNG ANH VÀO 10 SỞ GD – ĐT THÀNH PHỐ HỒ CHÍ MINH NĂ...
Kodo Millet PPT made by Ghanshyam bairwa college of Agriculture kumher bhara...

Kodo Millet PPT made by Ghanshyam bairwa college of Agriculture kumher bhara...
Making communications land - Are they received and understood as intended? we...

Making communications land - Are they received and understood as intended? we...
This PowerPoint helps students to consider the concept of infinity.

This PowerPoint helps students to consider the concept of infinity.
Unit 3 Emotional Intelligence and Spiritual Intelligence.pdf

Unit 3 Emotional Intelligence and Spiritual Intelligence.pdf
On National Teacher Day, meet the 2024-25 Kenan Fellows

On National Teacher Day, meet the 2024-25 Kenan Fellows
Cellular Energy pt.1
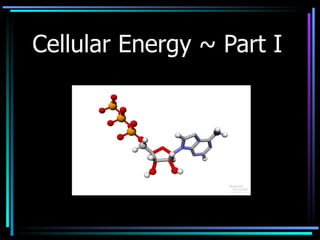
- 1. Cellular Energy ~ Part I
- 2. Endergonic vs. Exergonic Reactions
- 3. Energy Diagram for a Reversible Reaction
- 27. The ATP – ADP Cycle