Unit 1 lesson 14 intro to periodic table
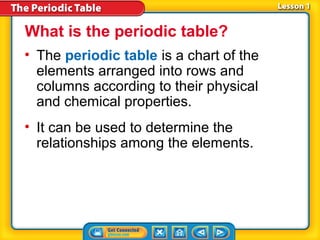
- 1. What is the periodic table? • The periodic table is a chart of the elements arranged into rows and columns according to their physical and chemical properties. • It can be used to determine the relationships among the elements.
- 2. Atomic Number Atomic Number 11 11 Na Na Element symbol Element symbol Sodium Sodium 22.990 22.990 Element name Element name Atomic Mass Atomic Mass 11 11 Atomic # = ______ 22.990 22.990 Atomic Mass = _________ 23 23 Mass # = _________ 11 11 # of Protons = ________ 12 12 # of Neutrons = ________ 11 11 # of Electrons = ________
- 3. Arranged according Arranged according to similarities to similarities or patterns or patterns
- 4. Today’s Periodic Table You can identify many of the properties of an element from its placement on the periodic table.
- 5. The table is organized into columns, rows, and blocks, which are based on certain patterns of properties.
- 6. Today’s Periodic Table (cont.) • A group is a column on the periodic table. • Elements in the same group have similar chemical properties and react with other elements in similar ways.
- 7. Today’s Periodic Table (cont.) What can you infer about the properties of two elements in the same group?
- 8. Today’s Periodic Table (cont.) • The rows on the periodic table are called periods. • The atomic number of each element increases by one as you read from left to right across each period.