Lecture 11 2013_
•Télécharger en tant que PPT, PDF•
0 j'aime•1,576 vues
laser fundamentals
Signaler
Partager
Signaler
Partager
Recommandé
Recommandé
Contenu connexe
Tendances
Tendances (20)
B.Tech sem I Engineering Physics U-IV Chapter 1-ATOMIC PHYSICS

B.Tech sem I Engineering Physics U-IV Chapter 1-ATOMIC PHYSICS
LASERS, CHARACTERISTICS, STIMULATED ABSORPTION, SPONTANEOUS EMISSION, STIMULA...

LASERS, CHARACTERISTICS, STIMULATED ABSORPTION, SPONTANEOUS EMISSION, STIMULA...
M.Sc.Part-II Sem- III (Unit - IV) Nuclear Magnetic Resonance Spectroscopy

M.Sc.Part-II Sem- III (Unit - IV) Nuclear Magnetic Resonance Spectroscopy
Photoelectric Effect And Dual Nature Of Matter And Radiation Class 12

Photoelectric Effect And Dual Nature Of Matter And Radiation Class 12
Class 12th Physics Photoelectric effect dual nature of matter radiations

Class 12th Physics Photoelectric effect dual nature of matter radiations
En vedette
En vedette (20)
Similaire à Lecture 11 2013_
Similaire à Lecture 11 2013_ (20)
Lecture 04; spectral lines and broadening by Dr. Salma Amir

Lecture 04; spectral lines and broadening by Dr. Salma Amir
Interaction of radiation with Matter - Dr. Vandana

Interaction of radiation with Matter - Dr. Vandana
Analytical class atomic absorption spectroscopy, P K MANI

Analytical class atomic absorption spectroscopy, P K MANI
Plus de Gabriel O'Brien
Plus de Gabriel O'Brien (20)
Superconducting qubits for quantum information an outlook

Superconducting qubits for quantum information an outlook
Quantum computer based on color centers in diamond

Quantum computer based on color centers in diamond
Fox m quantum_optics_an_introduction_photon antibunching1

Fox m quantum_optics_an_introduction_photon antibunching1
Fox m quantum_optics_an_introduction_optical cavities

Fox m quantum_optics_an_introduction_optical cavities
Invited review article single photon sources and detectors

Invited review article single photon sources and detectors
Quantum jumps of light recording the birth and death of a photon in a cavity

Quantum jumps of light recording the birth and death of a photon in a cavity
Dernier
+971581248768 Mtp-Kit (500MG) Prices » Dubai [(+971581248768**)] Abortion Pills For Sale In Dubai, UAE, Mifepristone and Misoprostol Tablets Available In Dubai, UAE CONTACT DR.Maya Whatsapp +971581248768 We Have Abortion Pills / Cytotec Tablets /Mifegest Kit Available in Dubai, Sharjah, Abudhabi, Ajman, Alain, Fujairah, Ras Al Khaimah, Umm Al Quwain, UAE, Buy cytotec in Dubai +971581248768''''Abortion Pills near me DUBAI | ABU DHABI|UAE. Price of Misoprostol, Cytotec” +971581248768' Dr.DEEM ''BUY ABORTION PILLS MIFEGEST KIT, MISOPROTONE, CYTOTEC PILLS IN DUBAI, ABU DHABI,UAE'' Contact me now via What's App…… abortion Pills Cytotec also available Oman Qatar Doha Saudi Arabia Bahrain Above all, Cytotec Abortion Pills are Available In Dubai / UAE, you will be very happy to do abortion in Dubai we are providing cytotec 200mg abortion pill in Dubai, UAE. Medication abortion offers an alternative to Surgical Abortion for women in the early weeks of pregnancy. We only offer abortion pills from 1 week-6 Months. We then advise you to use surgery if its beyond 6 months. Our Abu Dhabi, Ajman, Al Ain, Dubai, Fujairah, Ras Al Khaimah (RAK), Sharjah, Umm Al Quwain (UAQ) United Arab Emirates Abortion Clinic provides the safest and most advanced techniques for providing non-surgical, medical and surgical abortion methods for early through late second trimester, including the Abortion By Pill Procedure (RU 486, Mifeprex, Mifepristone, early options French Abortion Pill), Tamoxifen, Methotrexate and Cytotec (Misoprostol). The Abu Dhabi, United Arab Emirates Abortion Clinic performs Same Day Abortion Procedure using medications that are taken on the first day of the office visit and will cause the abortion to occur generally within 4 to 6 hours (as early as 30 minutes) for patients who are 3 to 12 weeks pregnant. When Mifepristone and Misoprostol are used, 50% of patients complete in 4 to 6 hours; 75% to 80% in 12 hours; and 90% in 24 hours. We use a regimen that allows for completion without the need for surgery 99% of the time. All advanced second trimester and late term pregnancies at our Tampa clinic (17 to 24 weeks or greater) can be completed within 24 hours or less 99% of the time without the need surgery. The procedure is completed with minimal to no complications. Our Women's Health Center located in Abu Dhabi, United Arab Emirates, uses the latest medications for medical abortions (RU-486, Mifeprex, Mifegyne, Mifepristone, early options French abortion pill), Methotrexate and Cytotec (Misoprostol). The safety standards of our Abu Dhabi, United Arab Emirates Abortion Doctors remain unparalleled. They consistently maintain the lowest complication rates throughout the nation. Our Physicians and staff are always available to answer questions and care for women in one of the most difficult times in their lives. The decision to have an abortion at the Abortion Clinic in Abu Dhabi, (United Arab Emirates)+971581248768+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...?#DUbAI#??##{{(☎️+971_581248768%)**%*]'#abortion pills for sale in dubai@
Dernier (20)
Locating and isolating a gene, FISH, GISH, Chromosome walking and jumping, te...

Locating and isolating a gene, FISH, GISH, Chromosome walking and jumping, te...
9999266834 Call Girls In Noida Sector 22 (Delhi) Call Girl Service

9999266834 Call Girls In Noida Sector 22 (Delhi) Call Girl Service
Module for Grade 9 for Asynchronous/Distance learning

Module for Grade 9 for Asynchronous/Distance learning
+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...
chemical bonding Essentials of Physical Chemistry2.pdf

chemical bonding Essentials of Physical Chemistry2.pdf
Pests of mustard_Identification_Management_Dr.UPR.pdf

Pests of mustard_Identification_Management_Dr.UPR.pdf
Connaught Place, Delhi Call girls :8448380779 Model Escorts | 100% verified

Connaught Place, Delhi Call girls :8448380779 Model Escorts | 100% verified
Vip profile Call Girls In Lonavala 9748763073 For Genuine Sex Service At Just...

Vip profile Call Girls In Lonavala 9748763073 For Genuine Sex Service At Just...
300003-World Science Day For Peace And Development.pptx

300003-World Science Day For Peace And Development.pptx
Pulmonary drug delivery system M.pharm -2nd sem P'ceutics

Pulmonary drug delivery system M.pharm -2nd sem P'ceutics
The Mariana Trench remarkable geological features on Earth.pptx

The Mariana Trench remarkable geological features on Earth.pptx
Forensic Biology & Its biological significance.pdf

Forensic Biology & Its biological significance.pdf
Thyroid Physiology_Dr.E. Muralinath_ Associate Professor

Thyroid Physiology_Dr.E. Muralinath_ Associate Professor
Justdial Call Girls In Indirapuram, Ghaziabad, 8800357707 Escorts Service

Justdial Call Girls In Indirapuram, Ghaziabad, 8800357707 Escorts Service
Lecture 11 2013_
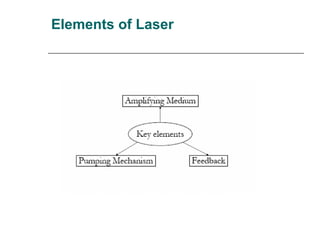
- 2. Working Principle of Laser
- 3. Working Principle of Laser Einstein’s assumptions & implications Thermodynamic equilibrium at arbitrary temperature T exists between the radiation field and the atoms The spectral density u(ν) of the radiation energy has the distribution characteristics of a blackbody at temperature T The atom population densities Nl and Nu at energy levels El and Eu, respectively, are distributed according to the Boltzman distribution at that temperature Population densities Nl and Nu are constant in time
- 4. Working Principle of Laser The radiative process and assumptions above, it follows that the rate of change of atoms in level Eu is given by ( ) ( )υυ uBNuBNAN dt dN lululuulu u +−−== 0 The spectral energy density can be written as ( ) 1 18 /3 33 − = kTh ec hn u υ υπ υ The Boltzman distribution ( ) kTh l ukTEE l u l u e g g e g g N N lu // υ−−− == …..(1) …..(2) …..(3)
- 5. Working Principle of Laser Solving eq.(1) in terms of u(υ) and substituting Nu/Nl from eq. (3), we obtain ( ) ( ) ul kTh lu u l ul ulullu ul BeB g g A BNNB A u − = − = // υ υ Compare it to eq. (2), gives 3 33 8 c hn B A ul ul υπ = lululu BgBg = ……(5) ……(6) ……(5)
- 6. Working Principle of Laser The importance of eqs. (5) and (6) cannot be underestimated. They tell us that: (i) The fundamental Einstein’s coefficients Aul, Bul and Blu are all inter-related. (ii) guBul = glBlu , i.e. stimulated emission and absorption are inverse processes. However note that the rate dNu/dt and dNl/dt differ depending on the population densities Nu and Nl. If Nu > Nl it leads to an increase in u(υ), an amplification. And if Nl > Nu it leads to a decrease in u(υ), an attenuation. For laser to operate, it is necessary that Nu be greater than Nl – a condition called population inversion. (iii) Since Bul/Aul is proportional to the reciprocal of the cube of the frequency, the higher the frequency the smaller Bul becomes in comparison with Aul. Since Bul is related to stimulated emission and Aul is related to spontaneous emission, it would seem that lasers of short wavelength radiation would be more difficult to build and operate.Two important ideas for the successful operation of a laser emerge from a review of Einstein’s study of the interaction of electromagnetic radiation with matter are, stimulated emission and population inversion.
- 7. Lasing condition Population inversion Necessary condition for amplification. The case of the upper level being more populated than the lower level. If stimulated emission rate exceeds absorption rate, net optical gain. The relationship for the intensity at a specific distance z into medium at a frequency ν and width Δν can be expressed as,
- 9. Population inversion If the value of the exponent is positive, the beam will increase in intensity and so amplification will occur. If it is negative, the beam will decrease in intensity and absorption will occur. The values of σul and z are always positive, thus amplification will occur only if This condition is not normal under thermal equilibrium l l u N g g N =u
- 11. Emission Broadening Homogeneous Broadening Due to the isotropic collisions with other atoms, which also causes non-radiative decay The processes lead to a Lorentzian distribution of emitting frequencies All of the atoms in level u have an equal probability of participating in the emission at any frequency ν of that emission shape – all atoms behave the same way
- 12. Emission Broadening Homogeneous Broadening The process can decrease either the decay time τu of the atoms residing in the excited level u OR affect the linewidth – depending on collision intervals Dephasing collisions interrupts the phase of radiating atoms without increasing their population decay rate
- 13. Emission Broadening Inhomogeneous Do not affect the lifetime, but do affect the linewidth The processes include Amorphous Crystal broadening, Doppler broadening and Isotope broadening Emission processes that lead to a Gaussian distribution of emitting frequencies Specific portions of the population density Nu contribute to different portions of the emission linewidth
- 14. Emission Broadening Amorphous Crystal Broadening Glass materials have various small regions oriented in slightly different directions Thus, each of the glass molecules can have slightly different energy levels This leads to different radiating frequencies for different regions Since the emission line is composed of the sum of all of the individual lines, this leads to a much broader emission spectrum
- 15. Emission Broadening Doppler Broadening Due to random movements of radiating atoms in all directions with a range of velocities This causes frequency shifts depending on the directions of the movements The faster the atoms move on the average, the broader the bandwidth A single photon might be able to stimulate one atom to emit because that atom happened to be Doppler shifted to the photon’s frequency, but it might not be able to stimulate another atom because it had a different Doppler shift than the first I.e. different atoms contribute to the gain at different frequencies within the laser bandwidth
- 16. Emission Broadening Isotope Broadening Due to the presence of more than one isotropic form of the species These different isotopes consist of atoms having the same number of protons and electrons, but with different numbers of neutrons Atoms with slightly different numbers of neutrons within their nuclei exhibit small differences in energy level values The slightly different energy level values for different isotopes provide slightly different frequencies for the transitions
