Neoplasia & Oncologic Pathology
- 1. Neoplasia & Oncologic Pathology Marc Imhotep Cray, M.D.
- 2. Marc Imhotep Cray, M.D. 2 Basic Concepts in Oncology Nomenclature and Definitions Epidemiology of Cancer Classification of Cancer Cell Biology of Cancer
- 3. Marc Imhotep Cray, M.D. Neoplasia Nomenclature 3 Neoplasia means “new growth” and indicates presence of cells or tissues showing evidence of abnormally controlled or disordered growth Neoplasms comprise cells that show differentiation along one or more pathways of development Benign vs Malignant Benign neoplasms expand locally but do not invade adjacent tissues or spread to distant sites, while Malignant neoplasms (cancers) invade adjacent tissues and spread to distant sites
- 4. 4 Neoplasia Nomenclature (2) Preneoplastic and neoplastic cellular changes Neoplasia Uncontrolled, clonal proliferation of cells Can be benign or malignant Dysplasia Disordered, non-neoplastic cell growth Used only with epithelial cells Mild dysplasia is usually reversible Severe dysplasia usually progresses to carcinoma in situ Differentiation degree to which a malignant tumor resembles its tissue of origin Well-differentiated tumors closely resemble their tissue of origin Poorly differentiated look almost nothing like their tissue of origin Anaplasia Complete lack of differentiation of cells in a malignant neoplasm
- 5. Marc Imhotep Cray, M.D. Neoplasia Nomenclature (3) 5 Genetic and environmental factors influence development of neoplasia Most germline (i.e. inherited and present in all cells) genetic influences on neoplasm development are polygenic in nature, while; A minority of neoplasms occur in association with a clearly defined inherited defect in a single gene (monogenic) Neoplasms vary in their relative incidence betw. populations and different geographical areas as a result of differences in gene pools and environmental contributors to disease development
- 6. Marc Imhotep Cray, M.D. Neoplasia Nomenclature (4) 6 Neoplasm development is characterized by accumulation of genetic defects within neoplastic cells In some neoplasms, this sequence is well characterized In others specific genetic mutations are found sufficiently commonly that their detection may be used to confirm the diagnosis of tissue type or to help to determine likely biological behavior of neoplasm (i.e. how aggressively neoplasm is likely to grow)
- 7. Marc Imhotep Cray, M.D. Neoplasia Nomenclature (5) 7 Benign tumors may compress adjacent tissue but do not invade it Malignant tumors grow locally, infiltrate adjacent tissue and metastasize via lymphatic channels and blood vessels to distant sites Benign tumors can cause death by compressing vital structures (e.g. within brainstem) but otherwise generally possess a much better prognosis than malignant tumors
- 8. Marc Imhotep Cray, M.D. Neoplasia Nomenclature (6) 8 Malignant tumors commonly cause extensive local tissue damage but tumor metastasis to distant sites is often key process that causes death in advanced malignancy Benign and malignant tumors may also produce chemicals such as hormones and, therefore, be associated with clinical symptoms of hormone excess Called a “paraneoplastic syndrome”
- 9. Marc Imhotep Cray, M.D. 9 Paraneoplastic syndromes Syndrome Mechanism Example Cushing Syndrome ACTH-like substance Lung small cell anaplastic (oat cell) carcinoma Hypercalcemia Parathormone-like substance Lung (squamous cell) carcinoma Hyponatremia Inappropriate ADH secretion Lung small cell anaplastic (oat cell) carcinoma Polycythemia Erythropoietin-like substance Renal cell carcinoma Trousseau Syndrome Hypercoagulable state Various carcinomas Hypoglycemia Insulin-like substance Various carcinomas and sarcomas Carcinoid Syndrome 5-hydroxy-indoleacetic acid (5-HIAA) Metastatic malignant carcinoid tumors Neoplasia and Oncologic Pathology SDL Tutorial .pdf
- 10. Marc Imhotep Cray, M.D. Neoplasia Nomenclature (7) 10 Clinical and pathological features of neoplasms can indicate whether they are benign or malignant in nature Histopathological examination of malignant neoplasms is important to determine how aggressively neoplasm is likely to grow and metastasize Features such as tumor type grade (histological assessment of aggressiveness) size and presence of lymph node metastases are most commonly assessed features used to predict biological behavior of malignant neoplasms
- 11. Marc Imhotep Cray, M.D. Neoplasia Nomenclature (8) 11 Most cancers (>90%) arise from "epithelial" tissues, such as inside lining of colon, breast, lung or prostate These are referred to as carcinomas and usually affect older people Contrastly, sarcomas are tumors that arise from "mesenchymal" tissues such as bone, muscle, connective tissue, cartilage and fat
- 12. Marc Imhotep Cray, M.D. Neoplasia Nomenclature (9) 12 Certain neoplasms occur primarily in childhood e.g. neuroblastoma and nephroblastoma Elderly individuals develop wear-and-tear diseases osteoarthritis atherosclerosis-associated conditions e.g. ischemic heart disease [IHD]) and Elderly individuals are at increased risk of many neoplasms
- 13. Marc Imhotep Cray, M.D. Neoplasia Nomenclature (10) 13 Neoplasm development is commonly associated with genetic abnormalities within neoplastic tissue however, proportion of neoplasms that occur as a result of a single inherited germline genetic abnormality (i.e. a mutation present within all of cells making up an individual) is relatively low Examples include inherited predispositions to breast cancer and colorectal cancer o Although relatively uncommon, these inherited syndromes are important since affected individuals may develop cancer at a young age and sometimes develop multiple cancers o Identification of affected families may allow cancer prevention programs and/or detection of cancers at an early stage
- 14. Marc Imhotep Cray, M.D. More Definitions of Neoplasia 14 Neoplasia is new, uncontrolled growth of cells that is not under physiologic control A "tumor" or "mass lesion" is simply a "growth" or "enlargement" which may not be neoplastic (such as a granuloma) The term "cancer" implies malignancy, but neoplasms can be subclassified as either benign or malignant There is no single mechanism by which a neoplasm arises • Many different mechanisms give rise to neoplasms what makes diagnosis and treatment so challenging
- 15. Marc Imhotep Cray, M.D. “Cancer "or “Malignancy" 15 The word "cancer "or "malignancy" strikes fear in a patient hearing it for first time • likely nothing else will be heard or remembered soon after that • Provide empathy, "I know that this can be very difficult to hear" • At a follow-up session, more details regarding a treatment plan can be discussed Some urgency is required, as survival for average cancer, such as a breast cancer, decreases by 1% per month of delayed diagnosis and treatment Introduction to Cancer Animations_Mechanisms of Medicine
- 16. Marc Imhotep Cray, M.D. Epidemiology of Cancer 10 Leading causes of death in United States 16 Number of deaths for leading causes of death in 2017 1. Heart disease: 647,457 2. Cancer: 599,108 3. Accidents (unintentional injuries): 169,936 4. Chronic lower respiratory diseases: 160,201 5. Stroke (cerebrovascular diseases): 146,383 6. Alzheimer’s disease: 121,404 7. Diabetes: 83,564 8. Influenza and pneumonia: 55,672 9. Nephritis, nephrotic syndrome, and nephrosis: 50,633 10. Intentional self-harm (suicide): 47,173 Source: CDC National Vital Statistics Reports, Vol. 68, No. 6, June 24, 2019
- 17. Marc Imhotep Cray, M.D. Cancer Incidence and Leading Causes of Cancer Death in Men and Women in U.S. 17 Cancers w highest incidence in men: Prostate cancer Lung cancer Colorectal cancer Leading causes of cancer deaths in men: Lung cancer Prostate cancer Colorectal cancer Cancers w highest incidence in women: Breast cancer Lung cancer Colorectal cancer Leading causes of cancer deaths in women: Lung cancer Breast cancer Colorectal cancer Learn more: Cancer Facts and Figures, 2020_American Cancer Society.pdf Important Note: Incidence of lung cancer and colorectal cancer both exceed incidence of prostate and breast cancer when data for men and women are combined.
- 18. Marc Imhotep Cray, M.D. Percent distribution of the 10 leading causes of death, by sex: United States, 2017. 18
- 19. Marc Imhotep Cray, M.D. Classification of Cancer Grade and Stage of a Neoplasm 19 Grade: Grade of a neoplasm is determined pathologically Refers to cellular characteristics of neoplasm, especially degree of differentiation of cells involved Grading of a neoplasm requires tissue usually from a biopsy Grades are reported by roman numerals I to III or I to IV, w higher numbers representing more poorly differentiating cellular patterns
- 20. Marc Imhotep Cray, M.D. 20 Grading of Malignant Neoplasms Grade Definition I Well differentiated II Moderately differentiated III Poorly differentiated IV Nearly anaplastic Neoplasia and Oncologic Pathology SDL Tutorial .pdf
- 21. Marc Imhotep Cray, M.D. Grade and Stage of a Neoplasm cont’ed. 21 Stage: Stage of a cancer is determined clinically but often takes into account information provided by pathologist (the grade) Stage is most frequently reported using tumor-node-metastasis (TNM) system: • T: Tumor size. T refers to local growth measured by size of primary tumor • N: Nodes. N refers to involvement of regional lymph nodes and is an indication of extent of tumor spread • M: Metastases. M refers to the presence or absence of distant metastases o Presence of metastases confers a high stage Staging is most helpful in determining prognosis Higher stages indicate a poorer prognosis
- 22. 22 Staging of Malignant Neoplasms Stage Definition Tis In situ, non-invasive (confined to epithelium) T1 Small, minimally invasive within primary organ site T2 Larger, more invasive within the primary organ site T3 Larger and/or invasive beyond margins of primary organ site T4 Very large and/or very invasive, spread to adjacent organs N0 No lymph node involvement N1 Nearby lymph node involvement N2 Regional lymph node involvement N3 More distant lymph node involvement M0 No distant metastases M1 Distant metastases present Neoplasia and Oncologic Pathology SDL Tutorial .pdf In the diagram below utilizing a lung carcinoma as an example, the principles of staging are illustrated:
- 23. Marc Imhotep Cray, M.D. Comprehension Check Up 23 5. A 48-year-old woman goes to her physician for a routine physical examination. A 4 cm diameter non-tender mass is palpated in her right breast. The mass appears fixed to the chest wall. Another 2 cm non-tender mass is palpable in the left axilla. A chest radiograph reveals multiple 0.5 to 2 cm nodules in both lungs. Which of the following classifications best indicates the stage of her disease? A. T1 N1 M0 B. T1 N0 M1 C. T2 N1 M0 D. T3 N0 M0 E. T4 N1 M1
- 24. Marc Imhotep Cray, M.D. E. She has a large invasive (high T) primary tumor mass with axillary node (N > 0) and lung metastases (M1). 24 https://www.slideshare.net/shaifalyrustagi/the-anatomy-of-pectoral-region
- 25. Marc Imhotep Cray, M.D. Classification of Cancer cont’ed. 25 Based upon origin: Malignant neoplasms arising from tissue embryologically derived from ectoderm or endoderm are usually carcinomas Examples include: • Squamous cell carcinoma of cervix • Adenocarcinoma of stomach • Hepatocellular carcinoma • Renal cell carcinoma Malignancies arising from mesoderm (connective tissues) are usually sarcomas Examples include: • Leiomyosarcoma • Chondrosarcoma • Osteosarcoma • Liposarcoma
- 26. Marc Imhotep Cray, M.D. Classification of Cancer cont’ed. 26 Neoplasms with more than one cell type but arising from only one germ layer are called "mixed tumors“ • Best example is benign mixed tumor (also called pleomorphic adenoma) of salivary gland Neoplasms with more than one cell type and arising from more than one germ layer are called teratomas • Common in ovary Neoplasms ending in "-blastoma" resemble primitive embryonic tissues, which are often pediatric neoplasms Examples include: • Retinoblastoma • Neuroblastoma • Hepatoblastoma • Medulloblastoma
- 27. Marc Imhotep Cray, M.D. Classification of Cancer cont’ed. 27 Not all malignant neoplasms have benign counterparts: • Hematopoietic and lymphoid cells (as in bone marrow and lymph node) give rise to leukemias and lymphomas o They have no benign counterpart Gliomas (astrocytomas, oligodengrogliomas, glioblastoma, etc) arise from glial cells in CNS They have no benign counterpart
- 28. Marc Imhotep Cray, M.D. Carcinomas 28 Carcinomas arise from epithelial surfaces in gastrointestinal tract, in respiratory tract, in urogenital tract, in biliary tract, in skin and in organs w epithelial-lined ducts (breast, pancreas, salivary gland, liver, etc) Endocrine glands, including testis and ovary, may also give rise to carcinomas In general, carcinomas are composed of polygonal-shaped cells • Carcinomas that form glandular configurations are called adenocarcinomas • Carcinomas that form solid nests of cells with distinct borders, intercellular bridges, and pink keratinized cytoplasm (Cytokeratins=A tumor marker) are called squamous cell carcinomas
- 29. Marc Imhotep Cray, M.D. Sarcomas 29 Sarcomas arise from soft tissues • connective tissues such as cartilage, bone, or fascia, • smooth or skeletal muscle, • blood vessels, • lymph vessels, • coverings of organs such as mesothelium In general, sarcomas are composed of very pleomorphic spindle- shaped cells Sarcomas are generally big and bad
- 30. Marc Imhotep Cray, M.D. Environmental causes: Chemicals: including those that are man-made (such as aniline dyes and bladder cancer), drugs (cigarette smoke, and lung cancer), and natural compounds (aflatoxins and liver cancer) which are carcinogenic • Controversy: Glyphosate and Non-Hodgkin Lymphoma o Research evidence: Chiu BC, Dave BJ, Blair A, Gapstur SM, Zahm SH, Weisenburger DD. Agricultural pesticide use and risk of t(14;18)-defined subtypes of non-Hodgkin lymphoma. Blood. 2006;108(4):1363-1369. doi:10.1182/blood-2005-12-008755 Causes of Neoplasia 30 Origin for many neoplasms is obscure however, some factor are known to causes a cell to be transformed to a neoplastic cell
- 31. Marc Imhotep Cray, M.D. Causes of Neoplasia cont’ed. 31 Oncogenic viruses: such as human papillomavirus (HPV) implicated in most squamous cell carcinomas of cervix and anogenital squamous papillomas, Epstein-Barr virus (EBV) implicated in African Burkitt's lymphoma, hepatitis B virus (HBV) implicated in development of hepatocellular carcinomas and Herpesvirus 8 causes Kaposi sarcoma Of Note: The human T-lymphotropic virus, human T-cell lymphotropic virus, or human T-cell leukemia-lymphoma virus (HTLV) family of viruses are a group of human retroviruses known to cause a type of cancer called adult T-cell leukemia/lymphoma (=HTLV type 1)
- 32. Marc Imhotep Cray, M.D. Causes of Neoplasia cont’ed. 32 Radiation: Including; • Ultraviolet (UV) light that induces pyrimidine dimers in DNA and promotes skin cancers (Goodsell, DS. The Molecular Perspective: Ultraviolet Light and Pyrimidine Dimers. The Oncologist 2001;6:298-299) o Most skin cancers are a result of exposure to the UV rays in sunlight o Both basal cell and squamous cell cancers (most common types of skin cancer) are found on sun-exposed parts of body their occurrence is related to lifetime sun exposure o The signature mutation caused by UV light is a CC to TT mutation, caused when a CC dimer is mispaired with two adenine bases during replication b/c of these mutations, connection betw. UV damage to DNA and cancer is quite clear NB: Xeroderma pigmentosum. An autosomal recessive disease in which increased sensitivity to sunlight is accompanied by a high incidence of skin cancers, including basal cell carcinoma, squamous cell carcinoma, and malignant melanoma
- 33. Marc Imhotep Cray, M.D. Causes of Neoplasia cont’ed. 33 • Ionizing radiation (such as gamma radiation/ x-rays) induces mutations in DNA promotes malignancies such as leukemia, thyroid, lung, colon, & breast CA National Research Council (US) Committee on the Biological Effects of Ionizing Radiation (BEIR V). Health Effects of Exposure to Low Levels of Ionizing Radiation: Beir V. Washington (DC): National Academies Press (US); 1990. 2, Genetic Effects of Radiation.
- 34. Marc Imhotep Cray, M.D. Causes of Neoplasia: Chemical carcinogenesis 34 Chemical carcinogenesis • There are two steps: initiation and promotion • An initiating carcinogenic agent irreversibly damages cell DNA (it is mutagenic) to start the process Examples of carcinogenic initiators include: o alkylating agents like cyclophosphamide o polycyclic aromatic hydrocarbons like epoxides found in smoked foods o aromatic amines or azo dyes used in food coloring o aflatoxins in moldy peanuts o nitrosamines in pickled foods See: Carcinogens. IN: First AID for the USMLE Step 1 2020, Pg. 225.
- 35. Marc Imhotep Cray, M.D. Causes of Neoplasia (2)Chemical carcinogenesis 35 A promoting agent (may be same as carcinogen) then acts (reversibly) to cause proliferation of a neoplastic cell clone, but there is a "dose-threshold" conc. of promoter below which neoplasia will not occur Examples of promoters include: hormones such as estrogen, drugs such as diethylstilbesterol, and chemicals
- 36. Marc Imhotep Cray, M.D. Causes of Neoplasia(3)Chemical carcinogenesis 36 An example of chemical carcinogenesis involves grilled meats • Meats exposed to high temperatures (above 162°C, or 325°F), either in an oven or over an open flame (grilling) undergo changes in proteins that form compounds called heterocyclic amines and polycyclic aromatic hydrocarbons can be carcinogenic (A char on meat indicates risk) o Following ingestion of these compounds, not only directly exposed tissues such as stomach and colon have an increased risk for cancer, but also sites elsewhere, including pancreas, breast, and prostate
- 37. Marc Imhotep Cray, M.D. Causes of Neoplasia: Hereditary 37 Hereditary causes: Chromosomes which have absent or defective anti-oncogenes that control growth (retinoblastoma results from defective chromosome 13) Obscure defects: racial predilections (American women have breast cancer more often than Japanese women; Japanese men have stomach cancer far more often than American men) Age: older persons have a greater propensity to develop neoplasms from lack of effective control mechanisms…see next slide
- 38. 38 Cancer and Ageing Key Concepts: Ageing is key risk factor for cancer and it promotes cancer All normal cells undergo cellular senescence, a permanent growth‐arrested phase Cellular senescence results due to intrinsic factors such as telomere shortening or extrinsic factors such as stress Induction of senescence by intrinsic or extrinsic factors is mediated by p53/p21 and p16INK4A/pRB tumor suppressor pathways Tumor suppressor pathways not only regulate cancer but also regulate organismic ageing Cellular senescence contributes to ageing and cancer via its cell autonomous and nonautonomous effects Cellular senescence can suppress or promote cancer via cell autonomous and nonautonomous mechanisms Source: Dimri, M. and Dimri, G.P. (2014). Cancer and the Ageing Process. In eLS , John Wiley & Sons, Ltd (Ed.). Molecular mechanism of cellular senescence.
- 39. Marc Imhotep Cray, M.D. Causes of Neoplasia: Altered DNA 39 Altered DNA: All of the above causes of neoplasia mentioned are mediated by cause, whatever it is, producing a mutation in, or damage to, cell DNA There can be mutations involving tumor suppressor genes (such as p53) fail to exert a controlling influence on growth activation Majority of human neoplasms arise via this mechanism NB: The p53 protein (“guardian of the genome”) is a tumor suppressor gene protein responsible for promoting apoptosis (regulated cell death) in cells with excessive DNA mutations or cellular damage.
- 40. Marc Imhotep Cray, M.D. Causes of Neoplasia: Altered DNA cont’ed. 40 In some cases these mutations are mediated by proto-oncogenes (genes which control cellular growth) undergo mutation to oncogenes which give rise to neoplasia • Proto-oncogenes can be activated by 1) point mutations, 2) translocations, and 3) gene amplification An example of this is chronic myelogenous leukemia (CML) neoplastic proliferation of WBCs o All cases of CML have "Philadelphia chromosome" which is a translocation betw chromosomes 9 and 22 • Translocation juxtaposes proto-oncogene ABL with breakpoint cluster region (BCR) on chromosome 22 • The chimeric ABL-BCR gene leads to production of a mutant protein w enhanced tyrosine kinase activity • Protein plays a role in regulation of cell growth in CML
- 41. Marc Imhotep Cray, M.D. Causes of Neoplasia: Altered DNA cont’ed. 41 About 15 to 20% of human cancers have been linked to oncogenic activity • The ras oncogene is transforming gene found most frequently in human cancers Oncogenic viruses may bring oncogenes w them, so-called viral oncogenes (typical of RNA containing "retroviruses" such as human T-lymphotropic viruses (HTLV's) Growth factors such as epidermal growth factor (EGF), platelet- derived growth factor (PDGF) and colony-stimulating factor-1 (CSF-1) assist oncogene activity • Transforming growth factor (TGF-α) also promotes tumor growth
- 42. Marc Imhotep Cray, M.D. Causes of Neoplasia: Altered DNA cont’ed. 42 DNA repair mechanisms may be affected • There are DNA excision repair genes that can be mutated, introducing genomic instability and a greater likelihood that mutations in other genes will occur to drive oncogenesis Examples include: o DNA mismatch repair genes: defective nucleotide "spell checker" introducing "microsatellite instability" of tandem repeat sequences in DNA. Seen in hereditary non-polyposis colon cancer (HNPCC) o Nucleotide excision repair genes: defective function in xeroderma pigmentosa, allowing DNA damage from pyrimidine dimer formation induced by ultraviolet light
- 43. Marc Imhotep Cray, M.D. Familial vs Sporadic Neoplasia 43 Most human cancers are "sporadic" b/c there is no identifiable inherited gene involved but cancers developed as a result of environmental factors carcinogens such as cigarette smoke that randomly induced mutations in cells led to uncontrolled growth Such factors are encountered throughout life and act over a long period of time hence, most sporadic cancers occur in adults Most affected persons have one primary site, and that site is where you would expect most cancers to be (breast, lung, prostate, colon, etc.)
- 44. Marc Imhotep Cray, M.D. Familial vs Sporadic Neoplasia cont’ed. 44 Familial CAs have a specific gene w a defined inheritance pattern Thus, one is born w "one strike" and it is a matter of years before another event triggers cancer growth • Tend to occur at a younger age than sporadic cancers One form of classic familial cancer syndrome involves a tumor suppressor gene, w the "two hit" hypothesis • A person inherits a bad p53 gene, for example, (first hit) but still has another functional copy of this gene on other chromosome • Sometime later, a mutation wipes out good gene (second hit) and growth control is lost allowing a clone of neoplastic cells to arise multiple organs can be affected Thus, familial cancers often involve more than one organ • Affected individuals can have more than one cancer, and • Malignancies other than epithelial are more likely (soft tissue sarcomas, leukemias/lymphomas, nervous system tumors)
- 45. Marc Imhotep Cray, M.D. Familial vs Sporadic Neoplasia cont’ed. 45 The rare Li Fraumeni Syndrome illustrates difference betw. "familial" and "sporadic" cancers In Li Fraumeni syndrome there is an inherited mutation in p53 gene, and a variety of cancers arise in persons w this mutation However, as indicated above, p53 mutations are most common mutations in sporadic cancers too
- 46. Marc Imhotep Cray, M.D. Biologic Characteristics of Neoplasms: Oncogenesis 46 Companion Video Edu: Intro to Cancer Animations_Mechanisms of Medicine
- 47. Marc Imhotep Cray, M.D. Oncogenesis 47 What drives a cell into uncontrolled growth? Figuratively, as you drive your cell down the highway of life, you have several factors controlling that journey You have an accelerator to speed up proliferation You have brakes to stop growth forward You have fuel in the tank to keep going Thus, you can have: Gene activation: accelerated cell proliferation • When you have activation of an oncogene, accelerator can be stuck open Loss of tumor suppression: loss of the brakes on cell growth • Tumor suppressor genes act as brakes associated with cell cycle regulation Persistence: accumulation of cells from lack of apoptosis • Apoptotic mechanisms also serve as brakes on growth Replication: acquisition of ability to keep dividing Cancer cells often acquire telomerase activity ("immortality" )
- 48. Marc Imhotep Cray, M.D. Oncogene and Tumor Suppressor Gene 48 An oncogene is a mutated form of a normal gene (proto- oncogene) Proto-oncogenes code for proteins that can lead to cellular proliferation and neoplasia However, proto-oncogenes are normally silent (unexpressed) Activating mutations convert proto-oncogenes into oncogenes and predispose to neoplasia b/c only a single gene needs to be mutated, oncogenes are seen as dominant mutations An example of a proto-oncogene is a gene for a growth factor receptor that, when mutated, could lead to inappropriate activation of receptor leading to uncontrolled cellular proliferation in absence of excessive levels of that growth factor
- 49. Marc Imhotep Cray, M.D. 49 Oncogenesis Mechanism Action Example Growth Promotion Overexpression of growth factor receptors (such as epidermal growth factor, or EGF) making cells more sensitive to growth stimuli HER2 (c-erb-B2) Increased growth factor signal transduction by an oncogene that lacks the GTPase activity that limits GTP induction of cytoplasmic kinases that drive cell growth RAS Overexpression of a gene product by stimulation from an oncogene (such as RAS) C-SIS Lack of normal gene regulation through translocation of a gene where it is controlled by surrounding genes to a place where it is no longer inhibited BCR-ABL Binding of oncogene product to the nucleus with DNA transcriptional activation to promote entry into the cell cycle C-MYC Neoplasia and Oncologic Pathology SDL Tutorial .pdf
- 50. Marc Imhotep Cray, M.D. 50 Oncogene Associated Neoplasms HER2 Breast and ovarian carcinomas RAS Many carcinomas and leukemias C-SIS Gliomas BCR-ABL Chronic myelogenous leukemia, acute lymphocytic leukemia C-MYC Lymphomas BRCA-1 Breast and ovarian carcinomas APC Colonic adenocarcinomas NF-1 Neurofibromas and neurofibrosarcomas Rb Retinoblastomas, osteosarcomas, small cell lung carcinomas P53* Many carcinomas BCL-2 Chronic lymphocytic leukemia, lymphomas *T Soussi and KG Wiman. TP53: an oncogene in disguise. Cell Death and Differentiation (2015) 22, 1239–1249. Neoplasia and Oncologic Pathology SDL Tutorial .pdf NB: In reference to Oncogenes and Tumor Suppressor Genes, for Board exam purposes, you should use the classification provided on Pg. 224. of First AID for the USMLE Step 1, 2020.
- 51. Marc Imhotep Cray, M.D. Oncogene & Tumor Suppressor Gene cont’ed. 51 A tumor suppressor gene is a normally active gene (in contrast with a proto-oncogene) Tumor suppressor genes encode proteins that suppress cellular proliferation; therefore, if its function is lost (disinhibited), cancerous cells are permitted to survive Mutations in tumor suppressor genes are said to be recessive b/c mutation must occur in both alleles in order to completely knock out the function of gene
- 52. Marc Imhotep Cray, M.D. 52 Loss of Tumor Suppressor Gene Function Loss of normal growth inhibition BRCA-1 Lack of regulation of cell adhesion with loss of growth control through cell interaction APC Loss of down-regulation of growth promoting signal transduction NF-1 Loss of regulation of cell cycle activation through sequestation of transcriptional factors RB Loss of regulation of cell cycle activation through lack of inhibition of cell proliferation that allows DNA repair p53 Limitation of Apoptosis Overexpression of gene, activated by translocation, prevents apoptosis BCL-2 Oncogenesis Mechanism Action Example Neoplasia and Oncologic Pathology SDL Tutorial .pdf
- 53. Marc Imhotep Cray, M.D. Oncogene & Tumor Suppressor Gene cont’ed. Familial cancer syndromes mutations 53 Tumor suppressor genes are more commonly involved in familial cancer syndromes b/c an embryo w one mutated copy of a tumor suppressor gene is likely to develop normally However, a second mutation may be acquired at a later date leading to cancer (loss of heterozygosity) • Eg., Knudson’s “two-hit hypothesis” On the other hand, an embryo is less likely to survive if it has inherited an oncogene b/c effects of this dominant negative mutation are typically much more disruptive to development
- 54. Marc Imhotep Cray, M.D. Tumor Suppressor Gene cont’ed Knudson’s “two-hit hypothesis” 54 Alfred Knudson studied retinoblastoma, which was later found to be a form of cancer caused by mutation of retinoblastoma (Rb) tumor suppressor gene He observed that in familial forms of retinoblastoma, tumors formed in pts at a much younger age than was typical for those arising in pts with no family history of the disease He theorized that in familial cases, a germ-line mutation was inherited (the first hit) and that a second sporadic mutation occurred early in life (the second hit) thus deactivating both tumor suppressor genes and causing tumor formation • Theory was therefore called the two-hit hypothesis In nonfamilial cases, however, mutations of both tumor suppressor genes (both hits) had to occur sporadically and thus took much longer to manifest as tumor formation
- 55. Marc Imhotep Cray, M.D. Apoptotic mechanisms 55 Along with tumor suppressor genes, apoptotic mechanisms also serve as brakes on growth There are cell receptors that, when contacting a ligand such as a hormone, will signal the cell to self destruct There are genes encoding proteins that oppose this process (antiapoptotic proteins)- thus, if you turn off apoptotic genes, or turn on antiapoptotic genes then cells will continue to accumulate (sort of like having all of your friends and relatives come to your house, but never leave)
- 56. Marc Imhotep Cray, M.D. What about the fuel of oncogenesis? 56 One of most important cellular resources is DNA in order to keep dividing, cells need to maintain their chromosomes However, the ends of chromosomes tend to "wear down" w repeated divisions enzyme telomerase can maintain ends of chromosomes • Ordinarily, only human stem cells have active telomerase • Cancer cells often acquire telomerase activity makes them "immortal" b/c they continue cell division indefinitely Neoplastic cells also need local resources to continue growth, in the form of structural support and blood supply
- 57. Marc Imhotep Cray, M.D. Cellular Transformation 57 Some factor, as discussed above, causes a cell to be transformed to a neoplastic cell not controlled by normal body processes • Most transformed cells die b/c they are too abnormal to function or are abnormal enough for body's immune system to destroy them • If, however, factors promoting neoplasia persist a transformed cell may some day give rise to a clone that does continue to grow
- 58. Marc Imhotep Cray, M.D. Cellular Transformation cont’ed. 58 Malignant neoplasms do not tend to arise from benign neoplasms (e.g., malignant melanomas do not come from benign nevi) However, in some cases such as adenomas of colon, appearance of benign neoplasm is a step toward possible malignancy, b/c oncogenic forces are at work producing additional abnormalities in DNA in existing lesions
- 59. Marc Imhotep Cray, M.D. Cellular Transformation cont’ed. 59 There are "pre-cancerous" conditions in which malignant neoplasia is more likely to occur (but not in every case): liver cirrhosis, chronic ulcerative colitis, atrophic gastritis, epidermal actinic keratosis, and oral leukoplakia (all are examples of "pre-cancerous” lesions) In these cases, there is ongoing cellular proliferation for repair of damaged tissue (often from ongoing inflammation) abnormal cell proliferation leads to a greater likelihood for mutation to occur
- 60. Marc Imhotep Cray, M.D. Clonality 60 Neoplastic cells tend to be monoclonal, or similar in genetic makeup, indicating origin from a transformed cell Non-neoplastic proliferations (such as reactions to inflammation) have cells that are polyclonal in origin The concept of "tumor progression" holds that subclones may arise over time from original malignant clone Subclones may differ from original clone in characteristics such as invasiveness, metastatic potential, and response to therapy Subclones may arise from acquisition of additional mutations NB: Clonality is a key characteristic of neoplastic cellular proliferations that distinguishes them from reactive proliferations (such as inflammation).
- 61. 61 Clonal expansion Source: Momna Hejmadi. Introduction to Cancer Biology, 2nd Ed. 2010, Fig. 1.1, Pg. 8. Cancer is a multi-gene, multi-step disease originating from single abnormal cell (clonal origin) Changes in DNA sequences result in cell progressing slowly to mildly aberrant stage Successive rounds of mutation & natural selection leads to a mass of abnormal cells called tumors Some cells in tumor undergo further rounds of mutations leading to formation of malignant cells which cause metastasis
- 62. Marc Imhotep Cray, M.D. Tumor Genetics 62 Neoplasms have a greater tendency to demonstrate karyotypic abnormalities such as translocations, deletions, and gene amplifications (which are also activators of proto-oncogenes) Leukemias and lymphomas are well known with chronic myelogenous leukemia, and t(8:14) translocation in Burkitt lymphoma being exemplary
- 63. Marc Imhotep Cray, M.D. Tumor Genetics cont’ed. 63 The process of neoplasia begins w cell transformation • A variety of chemical carcinogens , as stated above, i.e., benzene, cigarette smoke, and nitrites can initiate and/or promote the cell transformation process • As explained above, radiation, either as low level long-term environmental gamma rays or as higher dose therapeutic radiation, can also produce genetic mutations in cells • And we said persistent infectious agents such as HPV can lead to cellular transformation as well
- 64. Marc Imhotep Cray, M.D. Tumor Genetics cont’ed. 64 Genetic damage w DNA alterations leads to point mutations of genes, translocations of genetic material betw. chromosomes, and gene reduplication w amplification • These alterations transform proto-oncogenes into oncogenes o Proto-oncogenes within cells may play a role in growth promotion and regulation in normal cells (eg. embryogenesis) but are "turned off" in adults o Proto-oncogenes are "turned on" by transformation lead to uncontrolled growth that defines neoplasia
- 65. Marc Imhotep Cray, M.D. Tumor Genetics cont’ed. 65
- 66. Marc Imhotep Cray, M.D. Tumor growth 66 In general, less differentiated a neoplasm, faster it grows The cell cycle of neoplastic cells is not shortened, rather growth fraction of cells proliferating is increased this is offset by neoplastic cell death Tumor growth is expressed as a "doubling time" or time to increase twice in volume (e.g., from 1 to 1.3 cm diameter) • An aggressive malignant neoplasm doubles in 1 to 3 months, while benign neoplasms double in years
- 67. Marc Imhotep Cray, M.D. Tumor growth cont’ed. 67 Some neoplastic growth is influenced by host factors Estrogenic hormones aid growth of breast fibroadenomas or carcinomas and uterine leiomyomas b/c tumor cells have hormone receptors Growth is also dependent upon ability of tumor to develop a blood supply Factors secreted by neoplastic cells promote angiogenesis and fibroblast proliferation Cancers must elaborate growth factors in order to produce an environment for continued growth vascular endothelial growth factor (VEGF) promotes angiogenesis that keep neoplasm supplied w bld VEGF increases expression of ligands that active Notch signaling pathway
- 68. Marc Imhotep Cray, M.D. Characteristics of Transformed (Neoplastic) Cells 68 Neoplastic cell growth is not inhibited by contact w surrounding cells and is not dependent on anchorage to a solid surface Neoplastic cells are discohesive and transplantable favoring invasion and metastasis Neoplastic cells can bind to laminin and fibronectin in connective tissues then secrete collagenases or proteases, and then invade Neoplastic cells may attain "immortality" or ability to keep dividing indefinitely
- 69. The Six Hallmarks of Cancer A heterotypic model proposed by Douglas Hanahan and Robert Weinberg in 2000, manifested as the six common changes in cell physiology that results in cancer: 1. Immortality: Continuous cell division and limitless replication 2. Produce ‘Go’ signals (growth factors from oncogenes) 3. Override ‘Stop’ signals (anti-growth signals from tumor suppressor genes) 4. Resistance to cell death (apoptosis) 5. Angiogenesis: Induction of new blood vessel growth 6. Metastasis: Spread to other sites Acquired Capabilities of Cancer Hanahan, D ; Weinberg, R. The Hallmarks of Cancer. Cell, Vol. 100, 57–70, January 7, 2000.
- 70. 70 The six hallmarks of CA (2) Almost all cancers share some or all 6 traits described, depending on the tumor Some tumors may show all these changes b/c of mutations in one key gene (e.g. the p53 gene controls at least 4 of the traits) whereas other tumors may need more than 1 mutation for progression In Re of the Graphic: Arrows on right (orange and red) show signals that regulate normal cell behavior Green arrows on left indicate abnormal growth triggered by cancer cells The green boxes outline the 6 key characteristics of cancer cells Source: Momna Hejmadi. Introduction to Cancer Biology, 2nd Ed. 2010, Fig. 1.5, Pg. 15.
- 71. Updated to: The Eight Hallmarks of Cancer It appears that all cancers display eight fundamental changes in cell physiology, which are considered the hallmarks of cancer. Hanahan D, Weinberg RA: Hallmarks of cancer: the next generation. Cell 144:646, 2011. These changes consist of the following: 1. Self-sufficiency in growth signals 2. Insensitivity to growth-inhibitory signals 3. Altered cellular metabolism 4. Evasion of apoptosis 5. Limitless replicative potential (immortality) 6. Sustained angiogenesis 7. Invasion and metastasis 8. Evasion of immune surveillance Eight cancer hallmarks and two enabling factors (genomic instability and tumor-promoting inflammation). Most cancer cells acquire these properties during their development, typically due to mutations in critical genes. Kumar V and Abbas AK. Robbins Basic Pathology 10th ed. Phil: Saunders, 2018. Fig. 6.17;Pg. 205.
- 72. Marc Imhotep Cray, M.D. Development of cancer through stepwise accumulation of complementary driver mutations. The order in which various driver mutations occur is usually unknown and may vary from tumor to tumor. 72 Kumar V and Abbas AK. Robbins Basic Pathology 10th ed. Phil: Saunders, 2018. Fig. 6.17;Pg. 205. What is driver mutation and passenger mutation? Identifying which mutations contribute to cancer development is a key step in understanding tumor biology and developing targeted therapies. Mutations that provide a selective growth advantage, and thus promote cancer development, are termed driver mutations, and those that do not are termed passenger mutations
- 73. 73 The Cell Cycle and restriction point Most cells in human body are quiescent(i.e., not actively dividing) Exceptions include cells of the hematopoietic system, the integument, and the intestinal mucosa However, those cells that divide follow a well-defined cyclic pattern Major challenges in cell replication include high-fidelity DNA duplication (in the S phase) and segregation of chromosomes into daughter cells (during mitosis or the M phase) Different phases & checkpoints help address these issues S phase: DNA synthesis G2 phase: gap period 2 M phase: mitosis G1 phase: gap period 1 R: restriction point The restriction point occurs just prior to S phase important b/c once restriction point is passed, a cell is committed to complete cell cycle Multiple cellular factors interact to allow a cell to pass the restriction point (Fig. next slide)
- 74. Marc Imhotep Cray, M.D. Regulation of the cell cycle 74 Brown, TA; Shah, SJ. USMLE Step 1 Secrets 4th Ed. Saunders, 2017. Pg. 225.
- 75. 75 Cell Cycle Checkpoints (aka cell cycle arrests) Most important function of cell cycle arrest is to allow for repair of cellular damage and DNA sequences If improperly duplicated DNA were not repaired at cell cycle checkpoints, then DNA mutations would quickly propagate, and integrity of cellular homogeneity could not be maintained • A cell may also arrest in absence of proper nutrients, growth factors, or hormones Cyclins are proteins that regulate progression from one phase of cell cycle to next • Cyclins phosphorylate cyclin-dependent kinases (CDKs) to form cyclin-CDK complexes that must be in either an activated or inactivated state for various cell cycle stages to commence or terminate • CDKs are regulated by CDK inhibitors such as p16, p21, and p27 o If these CDK inhibitors are mutated cell cycle becomes deregulated and cancer can result Brown, TA; Shah, SJ. USMLE Step 1 Secrets 4th Ed. Saunders, 2017. Pg. 225.
- 76. 76 Cell Cycle and Cancer Phases, Hallmarks, and Development_Amboss Online Version
- 77. Marc Imhotep Cray, M.D. What is the importance of p53? 77 The p53 protein (“guardian of the genome”) is a tumor suppressor gene protein responsible for promoting apoptosis (regulated cell death) in cells w excessive DNA mutations or cellular damage p53 can activate DNA repair genes during DNA damage, initiate apoptosis in cells that cannot be repaired, activate CDK inhibitor p21, and prevent phosphorylation and inactivation of Rb protein Loss of p53 can lead to loss of apoptotic regulation and thus to uncontrolled cellular proliferation normal p53 gene can be inactivated by hypermethylation aberrant methylation of tumor suppressor genes is an epigenetic mechanism p53 gene (TP53) is most commonly mutated gene in human CAs
- 78. Marc Imhotep Cray, M.D. Function of retinoblastoma (Rb) protein? 78 Like p53, Rb is a tumor suppressor protein functions to prevent cell cycle progression from G1 to S phase by binding to and inhibiting transcription factor E2F Rb is heavily regulated via p53 and cyclin-CDK complexes When in a hypophosphorylated state (which is maintained by p53), Rb functions as a tumor suppressor inhibits cell cycle progression until cell is actively ready to divide During G1 to S transition, ↑ levels of cyclin D/CDK4 and CDK6 and cyclin E/CDK2 phosphorylate Rb and prevent its binding to E2F (see slide 74) E2F then activates additional proteins that push cell cycle to S phase Loss-of-function mutations of Rb present in several human cancers
- 79. Marc Imhotep Cray, M.D. 79 Family members: E2F is a group of genes that encodes a family of transcription factors (TF) in higher eukaryotes. Three of them are activators: E2F1, 2 and E2F3a. Six others act as suppressors: E2F3b, E2F4-8. All of them are involved in the cell cycle regulation and synthesis of DNA in mammalian cells. Legend •cyc A - Cyclin A binding domain •DNA - DNA-binding domain •DP1,2 - domain for dimerization with DP1, 2 •TA - transcriptional activation domain •PB - pocket protein binding domain https://www.wikiwand.com/en/E2F
- 80. 80 Overview of signal transduction pathways involved in apoptosis. Learn more via Video Edu: Cell Signaling_JJ Medicine
- 81. Marc Imhotep Cray, M.D. Tumor Markers 81 Marker Cancer Alpha-fetoprotein (AFP) Hepatocellular carcinoma, testicular carcinomas Cancer Antigen (CA) 15-3 Breast Cancer Antigen (CA) 19-9 Colon, pancreas, bladder Cancer Antigen (CA) 125 Ovary Carcinoembryonic antigen (CEA) Gastrointestinal carcinomas, other adenocarcinomas Hepatocyte growth factor Marker of cell proliferation with therapy resistance and metastasis in colorectal carcinomas Human Chorionic Gonadotrophin (HCG) Gestational trophoblastic disease, testicular carcinomas Monoclonal immunoglobulin Multiple myeloma Myeloperoxidase Marker of oxidative stress in colorectal carcinomas Osteopontin Marker of cancer progression, invasiveness and metastasis Prostate specific antigen (PSA) Prostatic adenocarcinoma Tissue Inhibitor of Matrix Metalloproteinases 1 (TIMP1) Pancreas Neoplasia and Oncologic Pathology SDL Tutorial .pdf
- 82. 82 The Cancer-Immunity Cycle Generation of immunity to cancer is a cyclic process that can be self propagating, leading to an accumulation of immune-stimulatory factors that in principle should amplify and broaden T cell responses. The cycle is also characterized by inhibitory factors that lead to immune regulatory feedback mechanisms, which can halt the development or limit the immunity. This cycle can be divided into seven major steps, starting with the release of antigens from the cancer cell and ending with the killing of cancer cells. Each step is described in the graphic, with the primary cell types involved and the anatomic location of the activity listed. Source: Chen DS and Mellman I. Oncology Meets Immunology: The Cancer-Immunity Cycle. Immunity 39, July 25, 2013. Abbreviations are as follows: APCs, antigen presenting cells; CTLs, cytotoxic T lymphocytes
- 83. 83 Cancer-Immunity Cycle 7 Steps_Amboss Online Version
- 84. Marc Imhotep Cray, M.D. 84 Reversible to Irreversible Cellular Alterations
- 85. 85 Cellular adaptations Reversible changes that can be physiologic (eg, uterine enlargement during pregnancy) or pathologic (eg, myocardial hypertrophy 2° to systemic HTN). If stress is excessive or persistent, adaptations can progress to cell injury (eg, significant LV hypertrophy injury to myofibrils HF) First AID for the USMLE Step 1 2020, Pg. 206.
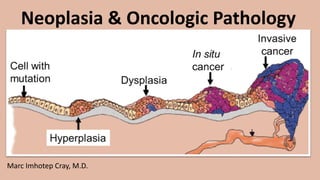
- 86. 86 Neoplasia and neoplastic progression: Uncontrolled, monoclonal proliferation of cells. Can be benign or malignant. Any neoplastic growth has two components: parenchyma (neoplastic cells) and supporting stroma (nonneoplastic; eg, blood vessels, connective tissue). 1. Normal cells Normal cells with basal apical polarity 2. Dysplasia Loss of uniformity in cell size and shape (pleomorphism); loss of tissue orientation; nuclear changes (eg, ↑ nuclear: cytoplasmic ratio) 3. Carcinoma in situ/ preinvasive Irreversible severe dysplasia that involves entire thickness of epithelium but does not penetrate the intact basement membrane 4. Invasive carcinoma Cells have invaded basement membrane using collagenases and hydrolases (metalloproteinases); Cell-cell contacts lost by inactivation of E-cadherin 5. Metastasis Spread to distant organ(s) via lymphatics or blood First AID for the USMLE Step 1 2020, Pg. 219.
- 87. 87 Left Ventricular Cardiac Hypertrophy Any increase in tissue size is not necessarily neoplasia Here is an example of left ventricular cardiac hypertrophy in which there has been an increase in size of myocardial fibers in response to an increased pressure load from hypertension With hypertrophy, the cells increase in size, but the cells do not increase in number Except for being larger, the cells are normal in appearance Alterations in cell growth can be physiologic (normal responses to stimuli) or pathologic Cardiac hypertrophy shown above is a pathologic response to abnormally elevated blood pressure Source: https://webpath.med.utah.edu/NEOHTML/NEOPLIDX.html
- 88. 88 Endometrial Hyperplasia Large fronds of endometrium seen in this uterus opened to reveal endometrial cavity are a result of hyperplasia This resulted from increased estrogen With hyperplasia, there is an increase in cell numbers to produce an increase in tissue size • However, cells are normal in appearance Sometimes hyperplasias can be "atypical" and cells not completely normal • Such conditions can be premalignant Source: https://webpath.med.utah.edu/NEOHTML/NEOPLIDX.html
- 89. Marc Imhotep Cray, M.D. Tissue evidence of carcinogenic factors at work 89 Metaplasia: an initial change from normal cells to a different cell type (such as chronic irritation of cigarette smoke causing ciliated pseudostratified epithelium to be replaced by squamous epithelium more able to withstand the insult) Dysplasia: an increasing degree of disordered growth or maturation of tissue (often thought to precede neoplasia) such as cervical dysplasia as a result of human papillomavirus infection • Dysplasia is still a reversible process however, once transformation to neoplasia has been made, process is not reversible…
- 90. Marc Imhotep Cray, M.D. …Thus, there is a natural history from metaplasia to dysplasia to neoplasia. This is best evidenced in development of uterine, cervix and respiratory tract neoplasms 90 https://sphweb.bumc.bu.edu/otlt/MPH-Modules/PH/PH709_Cancer/PH709_Cancer5.html
- 91. 91 Dysplasia Dysplasia is illustrated in histologic section from a biopsy of the uterine cervix shown to the right Area to the right encircled with red illustrates dysplasia; the cells have not yet progressed to cancer, but they are in danger of doing so, so they would be considered "pre-malignant" Dysplasia can revert back to hyperplasia, but it may also become malignant Therefore, dysplasia should be carefully monitored or treated Dysplasia on a Biopsy from the Uterine Cervix http://library.med.utah.edu/WebPath/FEMHTML/FEM008.html
- 92. Marc Imhotep Cray, M.D. Dysplasia on a Pap Smear 92 https://sphweb.bumc.bu.edu/otlt/MPH-Modules/PH/PH709_Cancer/PH709_Cancer5.html To continue this learning sequence see: Neoplasia and Oncologic Pathology SDL Tutorial. Pgs. 10-70.
- 93. 93 THE END See next slide for links to tools and resources for further study.
- 94. Marc Imhotep Cray, M.D. Further study: 94 eLearning (Cloud) Neoplasia Lecture Notes. pdf Neoplasia and Oncologic Pathology SDL Tutorial .pdf Oncologic Pathology: A Case-based Organ Systems Review Ppt. Oncologic Pathology Video Edu. Folder Atlas: Klatt EC. Robbins and Cotran Atlas of Pathology 3rd Ed. Elsevier- Saunders, 2015. Textbooks: Kumar V and Abbas AK. Robbins Basic Pathology 10th ed. Phil: Saunders, 2018. Rubin R and Strayer DS Eds. Essentials of Rubin's Pathology, 6th Ed.. Baltimore: Lippincott Williams & Wilkins, 2012.
