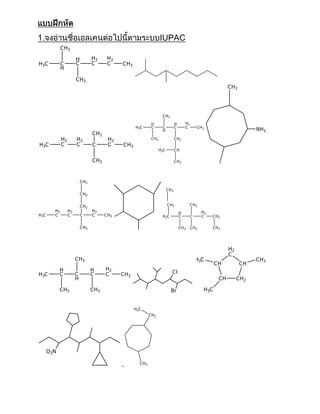
แบบฝึกหัดารอ่านชื่อแอลเคน แอลีน อแลไคน์
- 1. 1. IUPAC C H3 H H2 H2 H 3C C C C C C H3 H C H3 C H3 C H3 H H H2 H 3C C C C C C H3 H NH 2 C H3 H2 H2 H2 C H3 C H2 H 3C C C C C C H3 H 3C CH C H3 C H3 C H3 C H3 C H2 C H2 C H3 C H3 H2 H2 H2 H2 H H 3C C C C C C H3 H 2C C C C C H2 C H3 C H3 C H3 C H3 H2 C C H3 H 3C C H3 CH CH H H H2 H 3C C C C C C H3 Cl H CH C H2 C H3 C H3 Br H 3C H 3C C H2 O 2N C H3
- 2. C H3 F H 3C CH C H3 H H2 H 2C C C C C H2 C H3 C H3 C H3 Br Br C H2C H2C H2C H3 C H 3 C H 2 C H 2 C H 2 C HC H 2 C H 2 C H 2 C H 3 F Br C H 3 C HC H 2 C H 3 C 2H 5 C H 3 C H 2 C H 2 C H 2 C HC H 2 C H 2 C H 2 C H 3 C H3 C H3 C C H3 C H 3 C H 2 C H 2 C H 2 C HC H 2 C H 2 C H 2 C H 3 2. Write the structural formula for the following: a) 5-ethyl-2,8-dimethyldecane b) 2,2,4,4,- tetramethylpentane c) 2,2,6-trimethyloctane d) 3-ethyl-3,5- dimethylheptane e) 3- methyl pentane e) 2,2-dimethyl propane 3. Draw the line formula for each of the following cycloalkanes:
- 3. a) ethylcyclobutane b) 1,1-dimethylcyclopentane c) 1,3,5-trimethylcyclohexane 4. Write the line formula for each of the following haloalkanes: a) 1,1-dichloro-2,2-diflouoroethane b) 2,4-dichloro- 2,4-difluorohexane c) 1,1,3,5-tetrachlorocyclohexane d) 2- fluorooctane 5. What is the molar mass of this substance? 2 . . ………………………………………………………………………… ……… . 3 Br2 2.1
- 4. 2.2 2.3 ............................. ........ 4 STP 5 C H3 H2 H2 H H2 H2 H2 H2 H2 H2 H2 H2 H 3C C C C C H 3 H 3C C C C C C H3 H 3C C C C C C C H3 C H3 C H3 A B C 6.
- 5. 7. IUPAC NAME H2 H H2 H 3C C C C C C C H3 H H H 3C C H3 C H3 C H3 H H 3C C C C C H2 H H C H3 C H3 C H3 C H3 H H2 C H2 C H3 H 3C C C C C C C H3 H H H2 H H 3C C C C C C C H3 C H3 H H C H3
- 6. Br H 2C C H2 C H3 C H3 C H3 H2 H2 H2 H 3C C C C C C C C H2 C H3 C H3 C H2 C H2 C H2 H2 H2 C H3 C C C C C C H3 C H3 C H3 C H3 C H3 C H2 C H3 8 g dm3 STP
- 7. 3. --- 9. C5H10 10 IUPAC C H3 C H3 C H3 C H2 H 2C C H3 H2 H2 H2 H 3C C C CH H 3C C C CH HC C C CH C H3 C H3 H 3C C H2 C H3 C H3 C H3 H2 H H2 H2 H 3C C C C C C C C C H3 H H2 H2 C H2 C H2 C H C H3 C C C C C C H3 C H3 C H3 C H3 H 2C C H3 H 3C C H3 11
- 8. 12. Cl2 Br2
