Enzyme linked receptors (1)
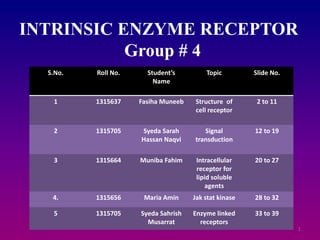
- 1. 1 INTRINSIC ENZYME RECEPTOR Group # 4 S.No. Roll No. Student’s Name Topic Slide No. 1 1315637 Fasiha Muneeb Structure of cell receptor 2 to 11 2 1315705 Syeda Sarah Hassan Naqvi Signal transduction 12 to 19 3 1315664 Muniba Fahim Intracellular receptor for lipid soluble agents 20 to 27 4. 1315656 Maria Amin Jak stat kinase 28 to 32 5 1315705 Syeda Sahrish Musarrat Enzyme linked receptors 33 to 39
- 2. STRUCTURE OF CELL RECEPTOR What is receptor ? A receptor is a protein molecule usually found inside or on the surface of a cell that receives chemical signals from outside the cell. When such chemical signals bind to a receptor, they cause some form of cellular/tissue response, e.g. change in the electrical activity of the cell. Therefore, a receptor is a protein molecule that recognizes and responds to endogenous chemical signals, e.g. the acetylcholine receptor recognized and responds to its endogenous ligand, acetylcholine. 2
- 3. However sometimes, the term is also used to include other proteins that are drug targets, such as enzymes, transporters and ion channels. 3
- 4. Receptor proteins are embedded in either the cell's plasma membrane (cell surface receptors), the cytoplasm (cytoplasmic receptors), or in the nucleus (nuclear receptors). A molecule that binds to a receptor is called a ligand, and can be a peptide (short protein) or another small molecule such as a neurotransmitter, hormone, pharmaceutical drug, or toxin. 4
- 5. The endogenously designated molecule for a particular receptor is referred to as its endogenous ligand. E.g. the endogenous ligand for the nicotinic acetylcholine receptor is acetylcholine but the receptor can also be activated by nicotine and blocked by curare. Each receptor is linked to a specific cellular biochemical pathway. While numerous receptors are found in most cells, each receptor will only bind with ligands of a particular structure, much like how locks will only accept specifically shaped keys. When a ligand binds to its corresponding receptor, it activates or inhibits the receptor's associated biochemical pathway. 5
- 6. TYPES OF RECEPTORS The structures of receptors are very diverse and can broadly be classified into the following catagories. 1. Type 1: L (ionotropic receptors) : These receptors are typically the targets of fast neurotransmitters such as acetylcholine (nicotinic) and GABA and activation of these receptor results in changes in ion movement across the membrane. They have a hetero structure. Each subunit consists of the extracellular ligand-binding domain and a transmembrane domain where the transmembrane domain in turn includes four transmembrane alpha helixes. The ligand binding cavities are located at the interface between the subunits. 6
- 7. 2. Type 2: G protein coupled receptors (metabotropic) This is the largest family of receptors and includes the receptors for several hormones and slow transmitters e.g. dopamine, metabotropic glutamate. They are composed of seven transmembrane alpha helices. The loops connecting the alpha helices form extracellular and intracellular domains. The binding site for larger peptidic ligands is usually located in the extracellular domain whereas the binding site for smaller non-peptidic ligands is often located between the seven alpha helices and one extracellular loop. These receptors are coupled to different intracellular effector systems via G-proteins. 7
- 8. 8
- 9. 3. Type 3: kinase linked and related receptors : These receptors are composed of an extracellular domain containing the ligand binding site and an intracellular domain, often with enzymatic function, linked by a single transmembrane alpha helix. E.g. the insulin receptor. 4. Type 4: nuclear receptors: While they are called nuclear receptors, these are actually located in the cytosol and migrate to the nucleus after binding with their ligands. They are composed of a C-terminal ligand binding region, a core DNA-binding domain (DBD) and an N-terminal domain that contains the AF1(activation function 1) region. 9
- 10. The core region has two zinc fingers that are responsible for recognizing the DNA sequences specific to this receptor. The N-terminal interacts with other cellular transcription factors in a ligand independent manner and depending on these interactions it can modify the binding/activity of the receptor. Steroid and thyroid hormone receptors are examples of such receptors. 10
- 11. 11
- 12. Signal Transduction • Signal transduction is also known as cell signaling. • It is the transmission of molecular signals from a cell's exterior to its interior • Signals received by cells must be transmitted effectively into the cell to ensure an appropriate response. • This step is initiated by cell-surface receptors. • Signal transduction occurs when an extracellular signaling molecule activates a specific receptor located on the cell surface or inside the cell. 12
- 13. 13
- 14. • In turn, this receptor triggers a biochemical chain of events inside the cell, creating a response. • Depending on the cell, the response alters the cell's metabolism, shape, gene expression, or ability to divide. • The signal can be amplified at any step. Thus, one signaling molecule can cause many responses. 14
- 15. Products For Signal Transduction • Calcium Signaling • Complement • Cytokine and NF-κB Signaling • G Proteins (Heterotrimeric) • G Proteins (Small) • Gap Channels • Growth Factor Receptors • Heat Shock Proteins • Hedgehog Signaling • Inositol and cAMP Signaling • MAPK Signaling • Nitric Oxide Signaling • Notch Signaling • PI 3-Kinase/Akt Signaling • Post-translational Modifications • Proteasome • Transcription Factors • Translocation, Exocytosis & Endocytosis • Wnt Signaling 15
- 16. Signal Transduction Pathways • Transmission is continued either by a series of biochemical changes within the cell or by modification of the cell membrane potential by the movement of ions in or out of the cell. • Receptors that initiate biochemical changes can do so either directly via intrinsic enzymatic activities within the receptor or by activating intracellular messenger molecules. • Signal transducing receptors are of four general classes: • Receptors that penetrate the plasma membrane and have intrinsic enzymatic activity or are enzyme associated (Enzyme-linked Receptors) • Receptors that are coupled, inside the cell, to G proteins (7-TM Receptors) • Receptors that are found intracellularly and upon ligand binding directly alter gene transcription (Nuclear Receptors) • Ligand-gated ion channels 16
- 17. 17
- 18. • The intracellular component of signal transduction is highly receptor specific, thereby maintaining the specificity of the incoming signal inside the cell. • Signal transduction pathways amplify the incoming signal by a signaling cascade using a network of enzymes. • These enzymes act on one another in specific ways to ultimately generate a precise and appropriate physiological response by the cell. • Signal transduction involves altering the behavior of proteins in the cascade, in effect turning them on or off like a switch. • Adding or removing phosphates is a fundamental mechanism for altering the shape, and therefore the behavior, of a protein. • Several small molecules within the cell act as intracellular messengers (also known as second messengers). These include cAMP, cGMP, nitric oxide, lipids and Ca2+ ions. • Activated receptors stimulate second messenger production, which in turn activate other enzymes and so the cascade continues. 18
- 19. 19
- 20. Intracellular receptor for lipid soluble agents Receptors for steroid and thyroid hormones are located inside target cells, in the cytoplasm or nucleus, and function as ligand- dependent transcription factors. That is to say, the hormone- receptor complex binds to promoter regions of responsive genes and stimulates or sometimes inhibits transcription from those genes. The mechanism of action of steroid hormones is to modulate gene expression in target cells. 20
- 21. Structure of Intracellular Receptors • these receptors are composed of a single polypeptide chain that has, in the simplest analysis, three distinct domains: • The amino-terminus: In most cases, this region is involved in activating or stimulating transcription by interacting with other components of the transcriptional machinery. The sequence is highly variable among different receptors. • DNA binding domain: Amino acids in this region are responsible for binding of the receptor to specific sequences of DNA. • The car boxy-terminus or ligand- binding domain: This is the region that binds hormone 21
- 22. Hormone-Receptor Binding and Interactions with DNA • Receptor activation is the term used to describe conformational changes in the receptor induced by binding hormone. The major consequence of activation is that the receptor becomes competent to bind DNA. • Activated receptors bind to "hormone response elements", which are short specific sequences of DNA which are located in promoters of hormone-responsive genes. In most cases, hormone-receptor complexes bind DNA in pairs, as shown in the figure below. • Transcription from those genes to which the receptor is bound is affected. Most commonly, receptor binding stimulates transcription. The hormone-receptor complex thus functions as a transcription factor. 22
- 24. Example • As a specific example, consider glucocorticoids, a type of steroid hormone that probably affects the physiology of all cells in the body. The image to the right depicts a pair of glucocorticoid receptors (blue and green on the top) bound to their DNA hormone response element (bottom). The two steroid hormones are not visible in this depiction. 24
- 25. Glucocorticoid Action 1. GR exists in an inactive form in the cytoplasm complexed with heat shock protein 90 (hsp90). 2. Glucocorticoid (G) diffuses across cell membrane and enters cytoplasm 3. G binds to GR changes conformation dissociates from hsp90 4. exposes a nuclear localization signal on GR. 5. G-GR (hormone-receptor complex, HR) enters nucleus, dimerizes with another HR. 25
- 26. 6. HR dimmer binds to enhancer/hormone- response element upstream of hormone activated 7. Binding of HR dimmer to enhancer activates transcription 8. Most contain 2 zinc fingers (1) controls DNA binding, (2) controls demonization 26
- 28. JANUS KINASE JAK Kinase is a family of intracellular,nonA-receptor tyrosine kinases that transduced cytokine mediated signals via JAK-STAT pathway. These receptors differ in not having any catalytic domain .Mammals have four members of this family jak1, jak2, jak3, and tyrosine kinase 2. JAK STAT KINASE BINDING RECEPTOR 28
- 29. They were initially named as just another kinase. Janus kinase possess two near identical phosphate transferring domain. One exhibit kinase activity while another negatively regulates the kinase activity of the first. 29
- 30. JAK-STAT SIGNALLING Cytokine receptor typically consist of two chains each having an extracellular cytokine binding domain and an intra-cytoplasmic domain. It binds a member of family of protein tyrosine kinases called as janus kinases or JAK’S. Cytokine binding to the receptor stabilizes the dimmer. 30
- 31. 31
- 32. Jaks are brought together that are bound to cytoplasmic portion of each chain. Janus kinase phosphorylate the cytoplasmic tails of cytokine receptor. STAT binds to phosphorylated cytokines rp chains and themselves phosphorylated by JAKS. STAT dimmerizes and migrate to the nucleus where they can directly activate gene transcription. 32
- 34. •also known as a catalytic receptor •transmembrane receptor, where the binding of an extracellular ligand causes enzymatic activity on the intracellular side •integral membrane protein possessing both enzymatic catalytic and receptor functions •Upon ligand binding a conformational change is transmitted which activates the enzyme, initiating signaling cascades 34
- 35. Physiology and diseases • involved in growth, proliferation, differentiation, or survival • Because of this, their ligands are collectively called growth factors. • The effects of enzyme-linked receptors typically are slow requiring the expression of new genes • Mutations in receptor tyrosine kinases are responsible for a wide array of diseases, including cancers, neurodegeneration, achondroplasia and atherosclerosis. 35
- 36. This lecture will focus on: 1. Receptor tyrosine kinases 2. Tyrosine kinase-associated receptors 3. Receptor serine/threonine kinase 36
- 37. 1. Receptor serine/threonine kinases There are two types of serine/threonine kinase receptors, both of which contain an intracellular kinase domain. They are each dimeric proteins, so an active receptor complex is made up of four receptors. 1. Type I receptors • Inactive unless in complex with type II receptors. • Do not interact with ligand dimers. • Contain conserved sequences of serine and threonine residues near to their kinase domains. 2. Type II receptors • Constitutively active kinase domains (even in the absence of the bound ligand). • Able to phosphorylate and activate the type I receptor. 37
- 38. 2. Receptor tyrosine kinases (RTKs) • RTK ligands, such as fibroblast growth factor (FGF), epidermal growth factor (EGF), nerve growth factor (NGF) etc. bind as dimers. • Ligand binding to RTK monomers results in dimer formation. • Receptors possess an intracellular tyrosine kinase domain. Within the dimer the conformation is changed, locking the kinase into an active state. • The kinase of one receptor then phosphorylates a tyrosine residue contained in the "activation lip"of the second receptor. • This forces the activation lip out of the kinase active site, allowing ATP bind and resulting in enhanced kinase activity. • This induces phosphorylation at further tyrosine residues. • Phosphotyrosine is a conserved "docking site" for many intracellular signal transduction proteins that contain SH2 domains 38
- 39. 3. Tyrosine-kinase-associated receptors • Cytokines are the main ligands that signal through tyrosine kinase-associated receptors. • The intracellular side of each receptor is bound to a cytosolic tyrosine kinase protein. 1. Cytokines bind simultaneously to two receptor monomers. 2. This brings the two associated kinases closer together. 3. One kinase phosphorylates the other kinase in an area called the "activation lip" (similar to RTK activation). The activation lip moves out of the active site and binds ATP therefore enhancing kinase activity. 4. The enhanced kinase phosphorylates more tyrosine residues on the intracellular portion of the receptor. 5. Phosphotyrosines serve as "docking sites" for SH2 domain-containing proteins 39
- 40. QUESTIONS? 40