Sae reporting timelines and compensation as per new drugs and clinical trials rules 2019
•Télécharger en tant que PPTX, PDF•
4 j'aime•253 vues
Hey guys here is small presentation for SAE reporting timelines and compensation as per New Drugs and Clinical Trials Rules 2019 for India. Suggestions are welcome.
Signaler
Partager
Signaler
Partager
Recommandé
Recommandé
Contenu connexe
Tendances
Tendances (20)
Adverse Events and Serious Adverse Events - Katalyst HLS

Adverse Events and Serious Adverse Events - Katalyst HLS
Clinical trial protocol, ammendments, Protocol deviations and violations

Clinical trial protocol, ammendments, Protocol deviations and violations
Role & responsibilities of a clinical research coordinator

Role & responsibilities of a clinical research coordinator
Overview of Audits and Inspections in Clinical Research

Overview of Audits and Inspections in Clinical Research
New drugs and clinical trials rules, 2019_ Dilip Kawane

New drugs and clinical trials rules, 2019_ Dilip Kawane
Aggregate Reporting_Pharmacovigilance_Katalyst HLS

Aggregate Reporting_Pharmacovigilance_Katalyst HLS
KEY STAKEHOLDERS IN CLINICAL RESEARCH SUCHISMITA BANIK

KEY STAKEHOLDERS IN CLINICAL RESEARCH SUCHISMITA BANIK
Dernier
Presentation by Jared Jageler, David Adler, Noelia Duchovny, and Evan Herrnstadt, analysts in CBO’s Microeconomic Studies and Health Analysis Divisions, at the Association of Environmental and Resource Economists Summer Conference.Effects of Extreme Temperatures From Climate Change on the Medicare Populatio...

Effects of Extreme Temperatures From Climate Change on the Medicare Populatio...Congressional Budget Office
Dernier (20)
Russian anarchist and anti-war movement in the third year of full-scale war

Russian anarchist and anti-war movement in the third year of full-scale war
PNRR MADRID GREENTECH FOR BROWN NETWORKS NETWORKS MUR_MUSA_TEBALDI.pdf

PNRR MADRID GREENTECH FOR BROWN NETWORKS NETWORKS MUR_MUSA_TEBALDI.pdf
Canadian Immigration Tracker March 2024 - Key Slides

Canadian Immigration Tracker March 2024 - Key Slides
PPT Item # 9 - 2024 Street Maintenance Program(SMP) Amendment

PPT Item # 9 - 2024 Street Maintenance Program(SMP) Amendment
2024: The FAR - Federal Acquisition Regulations, Part 36

2024: The FAR - Federal Acquisition Regulations, Part 36
Effects of Extreme Temperatures From Climate Change on the Medicare Populatio...

Effects of Extreme Temperatures From Climate Change on the Medicare Populatio...
What is the point of small housing associations.pptx

What is the point of small housing associations.pptx
ZGB - The Role of Generative AI in Government transformation.pdf

ZGB - The Role of Generative AI in Government transformation.pdf
Sae reporting timelines and compensation as per new drugs and clinical trials rules 2019
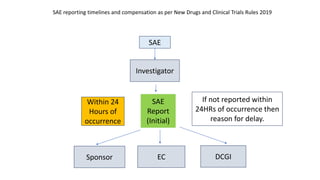
- 1. SAE Within 24 Hours of occurrence Investigator Sponsor EC DCGI If not reported within 24HRs of occurrence then reason for delay. SAE Report (Initial) SAE reporting timelines and compensation as per New Drugs and Clinical Trials Rules 2019
- 2. Analyzed report Within 14 Days of knowledge of occurrence (death cases) Investigator Sponsor DCGI Within 14 Days of reporting of SAE (non- death cases)
- 3. Within 30 days of receiving SAE report from Investigator Due analysis and opinion on compensation DCGI EC
- 4. DCGI Expert Committee chairperson (death cases) Reports from Sponsor, EC and Investigator 60 days from the receipt of SAE report Expert committee recommendations
- 5. DCGI ( with recommendation of expert committee) Order for compensation Sponsor Within 30days of receipt of order Pay compensation (if related to trial) Within 90days of receipt of SAE report