Chapter21胺及其衍生物
•Télécharger en tant que PPT, PDF•
0 j'aime•620 vues
Signaler
Partager
Signaler
Partager
Recommandé
Contenu connexe
Tendances
Tendances (16)
24 - Amino Acids, Peptides, and Proteins - Wade 7th

24 - Amino Acids, Peptides, and Proteins - Wade 7th
Reactions and pharmaceutical applications of aniline

Reactions and pharmaceutical applications of aniline
General Chemistry 2 Assignment - Preparation of amine (Group 13 and 18)

General Chemistry 2 Assignment - Preparation of amine (Group 13 and 18)
Amines, Nomenclature, Physical properties and Chemical by Shabab

Amines, Nomenclature, Physical properties and Chemical by Shabab
Similaire à Chapter21胺及其衍生物
Similaire à Chapter21胺及其衍生物 (20)
Organic chemistry (Amines), Sharda Public School, Almora U.K.

Organic chemistry (Amines), Sharda Public School, Almora U.K.
class 12 chemistry amines formula and structure pdf

class 12 chemistry amines formula and structure pdf
Plus de superxuds
Plus de superxuds (20)
Dernier
Saudi Arabia [ Abortion pills) Jeddah/riaydh/dammam/++918133066128☎️] cytotec tablets uses abortion pills 💊💊 How effective is the abortion pill? 💊💊 +918133066128) "Abortion pills in Jeddah" how to get cytotec tablets in Riyadh " Abortion pills in dammam*💊💊 The abortion pill is very effective. If you’re taking mifepristone and misoprostol, it depends on how far along the pregnancy is, and how many doses of medicine you take:💊💊 +918133066128) how to buy cytotec pills
At 8 weeks pregnant or less, it works about 94-98% of the time. +918133066128[ 💊💊💊 At 8-9 weeks pregnant, it works about 94-96% of the time. +918133066128) At 9-10 weeks pregnant, it works about 91-93% of the time. +918133066128)💊💊 If you take an extra dose of misoprostol, it works about 99% of the time. At 10-11 weeks pregnant, it works about 87% of the time. +918133066128) If you take an extra dose of misoprostol, it works about 98% of the time. In general, taking both mifepristone and+918133066128 misoprostol works a bit better than taking misoprostol only. +918133066128 Taking misoprostol alone works to end the+918133066128 pregnancy about 85-95% of the time — depending on how far along the+918133066128 pregnancy is and how you take the medicine. +918133066128 The abortion pill usually works, but if it doesn’t, you can take more medicine or have an in-clinic abortion. +918133066128 When can I take the abortion pill?+918133066128 In general, you can have a medication abortion up to 77 days (11 weeks)+918133066128 after the first day of your last period. If it’s been 78 days or more since the first day of your last+918133066128 period, you can have an in-clinic abortion to end your pregnancy.+918133066128
Why do people choose the abortion pill? Which kind of abortion you choose all depends on your personal+918133066128 preference and situation. With+918133066128 medication+918133066128 abortion, some people like that you don’t need to have a procedure in a doctor’s office. You can have your medication abortion on your own+918133066128 schedule, at home or in another comfortable place that you choose.+918133066128 You get to decide who you want to be with during your abortion, or you can go it alone. Because+918133066128 medication abortion is similar to a miscarriage, many people feel like it’s more “natural” and less invasive. And some+918133066128 people may not have an in-clinic abortion provider close by, so abortion pills are more available to+918133066128 them. +918133066128 Your doctor, nurse, or health center staff can help you decide which kind of abortion is best for you. +918133066128 More questions from patients: Saudi Arabia+918133066128 CYTOTEC Misoprostol Tablets. Misoprostol is a medication that can prevent stomach ulcers if you also take NSAID medications. It reduces the amount of acid in your stomach, which protects your stomach lining. The brand name of this medication is Cytotec®.+918133066128) Unwanted Kit is a combination of two medicines, ounwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE AbudhabiAbortion pills in Kuwait Cytotec pills in Kuwait
Saudi Arabia [ Abortion pills) Jeddah/riaydh/dammam/++918133066128☎️] cytotec tablets uses abortion pills 💊💊 How effective is the abortion pill? 💊💊 +918133066128) "Abortion pills in Jeddah" how to get cytotec tablets in Riyadh " Abortion pills in dammam*💊💊 The abortion pill is very effective. If you’re taking mifepristone and misoprostol, it depends on how far along the pregnancy is, and how many doses of medicine you take:💊💊 +918133066128) how to buy cytotec pills
At 8 weeks pregnant or less, it works about 94-98% of the time. +918133066128[ 💊💊💊 At 8-9 weeks pregnant, it works about 94-96% of the time. +918133066128) At 9-10 weeks pregnant, it works about 91-93% of the time. +918133066128)💊💊 If you take an extra dose of misoprostol, it works about 99% of the time. At 10-11 weeks pregnant, it works about 87% of the time. +918133066128) If you take an extra dose of misoprostol, it works about 98% of the time. In general, taking both mifepristone and+918133066128 misoprostol works a bit better than taking misoprostol only. +918133066128 Taking misoprostol alone works to end the+918133066128 pregnancy about 85-95% of the time — depending on how far along the+918133066128 pregnancy is and how you take the medicine. +918133066128 The abortion pill usually works, but if it doesn’t, you can take more medicine or have an in-clinic abortion. +918133066128 When can I take the abortion pill?+918133066128 In general, you can have a medication abortion up to 77 days (11 weeks)+918133066128 after the first day of your last period. If it’s been 78 days or more since the first day of your last+918133066128 period, you can have an in-clinic abortion to end your pregnancy.+918133066128
Why do people choose the abortion pill? Which kind of abortion you choose all depends on your personal+918133066128 preference and situation. With+918133066128 medication+918133066128 abortion, some people like that you don’t need to have a procedure in a doctor’s office. You can have your medication abortion on your own+918133066128 schedule, at home or in another comfortable place that you choose.+918133066128 You get to decide who you want to be with during your abortion, or you can go it alone. Because+918133066128 medication abortion is similar to a miscarriage, many people feel like it’s more “natural” and less invasive. And some+918133066128 people may not have an in-clinic abortion provider close by, so abortion pills are more available to+918133066128 them. +918133066128 Your doctor, nurse, or health center staff can help you decide which kind of abortion is best for you. +918133066128 More questions from patients: Saudi Arabia+918133066128 CYTOTEC Misoprostol Tablets. Misoprostol is a medication that can prevent stomach ulcers if you also take NSAID medications. It reduces the amount of acid in your stomach, which protects your stomach lining. The brand name of this medication is Cytotec®.+918133066128) Unwanted Kit Mifty kit IN Salmiya (+918133066128) Abortion pills IN Salmiyah Cytotec pills

Mifty kit IN Salmiya (+918133066128) Abortion pills IN Salmiyah Cytotec pillsAbortion pills in Kuwait Cytotec pills in Kuwait
Dernier (20)
Pre Engineered Building Manufacturers Hyderabad.pptx

Pre Engineered Building Manufacturers Hyderabad.pptx
Lundin Gold - Q1 2024 Conference Call Presentation (Revised)

Lundin Gold - Q1 2024 Conference Call Presentation (Revised)
Falcon Invoice Discounting: Tailored Financial Wings

Falcon Invoice Discounting: Tailored Financial Wings
Falcon Invoice Discounting: Empowering Your Business Growth

Falcon Invoice Discounting: Empowering Your Business Growth
How to Get Started in Social Media for Art League City

How to Get Started in Social Media for Art League City
Falcon Invoice Discounting: Aviate Your Cash Flow Challenges

Falcon Invoice Discounting: Aviate Your Cash Flow Challenges
Call 7737669865 Vadodara Call Girls Service at your Door Step Available All Time

Call 7737669865 Vadodara Call Girls Service at your Door Step Available All Time
HomeRoots Pitch Deck | Investor Insights | April 2024

HomeRoots Pitch Deck | Investor Insights | April 2024
Horngren’s Cost Accounting A Managerial Emphasis, Canadian 9th edition soluti...

Horngren’s Cost Accounting A Managerial Emphasis, Canadian 9th edition soluti...
unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi
Mifty kit IN Salmiya (+918133066128) Abortion pills IN Salmiyah Cytotec pills

Mifty kit IN Salmiya (+918133066128) Abortion pills IN Salmiyah Cytotec pills
Rice Manufacturers in India | Shree Krishna Exports

Rice Manufacturers in India | Shree Krishna Exports
Uneak White's Personal Brand Exploration Presentation

Uneak White's Personal Brand Exploration Presentation
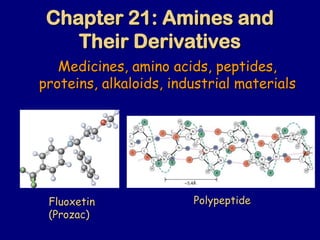
Chapter21胺及其衍生物
- 1. Chapter 21: Amines and Their Derivatives Medicines, amino acids, peptides, proteins, alkaloids, industrial materials Fluoxetin (Prozac) O F F F Polypeptide
- 2. Amphetamine MescalineEpinephrine (Adrenaline) Alkaloids (natural amines) HO OH HO H NHCH3 H2C H2N H CH3 H3CO OCH3 OCH3 NH2
- 3. Amines in Industry: Nylon Nylon 6,6 Hosiery, gears, textile fiber,.......
- 4. Primary amines Secondary amines Tertiary amines Naming: Alkanamines N-alkyl, as in amides
- 5. Substituent name: Amino- Common names: Alkylamine 4-(2-Aminoethyl)benzoic acid HOOC NH2
- 6. Physical Properties 1. Tetrahedral structure Basic Ea = 5-7 kcal mol-1 Inversion
- 7. 3. Basicity-Nucleophilicity RNH2 : HCl RNH3 + Cl -+ Primary ammonium chloride :: : : RNH2 : CH3I RNH2CH3 + I + :: : : - Secondary ammonium chloride 2. Chiral amines racemize
- 8. Ammonium salts are configurationally stable N H3C C6H5 CH2CH3 N H3C C6H5 CH2CH3 CH2Cl N H3C C6H5 CH2CH3 H2C N H3C C6H5 CH2CH3 H2C + Cl: : :: - Cl: : :: - + + + + Can be resolved, e.g, by fractional crystallization of a salt with optically active anion
- 9. 4. Weaker hydrogen bonds than OH NR H H NH H R : : Only primary and secondary amines can function as proton donors Less electronegative than oxygen
- 11. Spectroscopy Infrared spectrum of cyclohexanamine N-H IR stretch: 3300–3500 cm-1
- 12. 1H NMR spectrum of azacyclohexane NH usually broad, variable, like OH
- 13. 13C Chemical Shifts (ppm) N is less deshielding than O
- 14. Mass spectrum of N,N -diethylethanamine Odd molecular weight
- 15. •C : Mass 12 and tetravalent, e.g. CH4, results in even M+ • N: Mass 14 and trivalent, e.g. NH3, results in odd M+ for N = 1,3,5... • Preferred fragmentation α–β bond cleavage (even fragments)
- 17. • Preparation of Sodium Amide: • Preparation of LDA: Needs strong base 2Na + 2NH3 2NaNH2 + H2 Catalytic Fe3+
- 18. Basicity: Kb = [RNH3] [HO ] [RNH2] + - ~ 10-4 Ammonium ion pKb ~ 4 Recall: pKa + pKb = 14 Thus, pKa of protonated amine ~ 10
- 19. Better (to compare with other pKa values): O H H R + pKa = -3
- 20. Synthesis 1. Alkylation of NH3, primary, and secondary amines Problem: • Overalkylation: Even though amine gets more hindered, it also gets more nucleophilic One solution is to have excess of RNH2 and use RX as limiting reagent
- 21. 2. Via nitriles 3. Arenamine via nitroarene 1,3-Benzenediamine HNO3,H2SO4 NO2 HNO3,H2SO4 NO2 NO2 H2,Ni CF3CO3H NH2 NH2
- 22. 4. Gabriel Synthesis ( RX RNH2) Solves problem of overalkylation
- 23. 5. Reductive Amination Primary amine Secondary amine Tertiary amine Goes through iminium ion Goes through iminium ion
- 24. Formaldehyde and a secondary amine: Use of formaldehyde effects monomethylation of an amine. RedAmin
- 25. 6. Reduction of amides RCNHR’ LiAlH4 O RCH2NHR’ Recall: 7. Hofmann Rearrangement RCCl O Amide and RCNHR’ O RCNR’’R’ O August Wilhelm von Hofmann 1818-1892
- 26. Reactions of Amines 1. Hofmann Elimination Recall: :: Oxonium ion Analogous (but attenuated) behavior in quaternary ammonium ions. Works for E2 ROH H+ R OH2 +
- 27. Hofmann eliminations are often used to break down larger amines, e.g., alkaloids, into smaller fragments. Procedure: First, methylate exhaustively to the quaternary ammonium salt; second, treat with base.
- 28. Degradation of a simple alkaloid H H
- 29. 2. Mannich Reaction Alkylation of Enols by Iminium Ions The enol is derived from an enolizable ketone/aldehyde and the iminium ion is formed in situ by condensation of a second, more reactive carbonyl component with an amine. The outcome is a β- aminocarbonyl product. Form iminium ion Form iminium ion Carl Ulrich Franz Mannich 1877-1947
- 30. Net: α–Amino- methylation of enolizable alde- hyde Mannich
- 31. 3. Reaction with nitrous acid • Nitrosyl cation: like C O , but more electrophilic (charged) and gets attacked by amines :: Tertiary amine (stable) Secondary amine N –Nitrosamines are implicated in the carcinogenicity of cured meats
- 32. Primary N -nitrosamines decompose R = Alkyl mess, R = Arene stable (Chapter 22)
- 33. 4. Diazomethane CH2 N N Amides are also subject to N -Nitrosation N-Methyl-N-nitrosourea is a precursor to diazomethane ――――
- 34. Step 1: Hydroxide Addition C O NH2H3CN N O OH C O NH2H3CN N O OH : :: :: : :: : : : : : - : : : - Mechanism: Step 2: Elimination C O NH2H3CN N O OH CH3N N O C HO NH2 O HOH CH3N NOH OH CO2 HNH2 - : : :: : : : : : : : : : : : : : : : :: : :: : :: - + + +
- 35. CH2N NOH OH -H2O H2C N N OH H2C N N H2C N NH Step 3: Dehydration : : : : : : :: :: : : : : :: : : - OH - - - + - : Diazomethane has two applications as a reagent: In esterifications and as a source of carbene CH2.
- 36. H2C N NC O OR H C O OR H3C N N N N C O OCH3R • Formation of Esters • Generation of Carbene • Cyclopropanation H2C N N + - : H2C N N C H H :: + - hν or Cu or : + : : - +: : : + :: : : -