Chapter #3 Lectures Part Ii
•Télécharger en tant que PPT, PDF•
0 j'aime•529 vues
Signaler
Partager
Signaler
Partager
Recommandé
Role of biocatalysts in green synthesis , green chemistry

Role of biocatalysts in green synthesis , green chemistryMinal Saini , student of Chaudhary bansilal University , Bhiwani, Haryana
Recommandé
Role of biocatalysts in green synthesis , green chemistry

Role of biocatalysts in green synthesis , green chemistryMinal Saini , student of Chaudhary bansilal University , Bhiwani, Haryana
Synthesis, characterisation and antibacterial activity of copolymer (n vinylp...

Synthesis, characterisation and antibacterial activity of copolymer (n vinylp...eSAT Publishing House
Contenu connexe
Tendances
Synthesis, characterisation and antibacterial activity of copolymer (n vinylp...

Synthesis, characterisation and antibacterial activity of copolymer (n vinylp...eSAT Publishing House
Tendances (6)
Synthesis, characterisation and antibacterial activity of copolymer (n vinylp...

Synthesis, characterisation and antibacterial activity of copolymer (n vinylp...
Crosslinked Microgels as Platform for Hydrolytic Catalysts Article pubs.acs.o...

Crosslinked Microgels as Platform for Hydrolytic Catalysts Article pubs.acs.o...
SYNTHESIS OF BIOACTIVE HETEROCYCLES BY USING SONOCHEMISTRY

SYNTHESIS OF BIOACTIVE HETEROCYCLES BY USING SONOCHEMISTRY
En vedette
En vedette (18)
2011 topic 01 lecture 3 - limiting reactant and percent yield

2011 topic 01 lecture 3 - limiting reactant and percent yield
Rate of reaction =measure rate and intro and collision theory

Rate of reaction =measure rate and intro and collision theory
K to 12 Mathematics Curriculum Guide for Grades 1 to 10

K to 12 Mathematics Curriculum Guide for Grades 1 to 10
Similaire à Chapter #3 Lectures Part Ii
Similaire à Chapter #3 Lectures Part Ii (20)
Dernier
Dernier (20)
Apidays New York 2024 - Passkeys: Developing APIs to enable passwordless auth...

Apidays New York 2024 - Passkeys: Developing APIs to enable passwordless auth...
Why Teams call analytics are critical to your entire business

Why Teams call analytics are critical to your entire business
EMPOWERMENT TECHNOLOGY GRADE 11 QUARTER 2 REVIEWER

EMPOWERMENT TECHNOLOGY GRADE 11 QUARTER 2 REVIEWER
Biography Of Angeliki Cooney | Senior Vice President Life Sciences | Albany, ...

Biography Of Angeliki Cooney | Senior Vice President Life Sciences | Albany, ...
Web Form Automation for Bonterra Impact Management (fka Social Solutions Apri...

Web Form Automation for Bonterra Impact Management (fka Social Solutions Apri...
Introduction to Multilingual Retrieval Augmented Generation (RAG)

Introduction to Multilingual Retrieval Augmented Generation (RAG)
Cloud Frontiers: A Deep Dive into Serverless Spatial Data and FME

Cloud Frontiers: A Deep Dive into Serverless Spatial Data and FME
Finding Java's Hidden Performance Traps @ DevoxxUK 2024

Finding Java's Hidden Performance Traps @ DevoxxUK 2024
Vector Search -An Introduction in Oracle Database 23ai.pptx

Vector Search -An Introduction in Oracle Database 23ai.pptx
Apidays New York 2024 - The value of a flexible API Management solution for O...

Apidays New York 2024 - The value of a flexible API Management solution for O...
Boost Fertility New Invention Ups Success Rates.pdf

Boost Fertility New Invention Ups Success Rates.pdf
Cloud Frontiers: A Deep Dive into Serverless Spatial Data and FME

Cloud Frontiers: A Deep Dive into Serverless Spatial Data and FME
Strategies for Landing an Oracle DBA Job as a Fresher

Strategies for Landing an Oracle DBA Job as a Fresher
TrustArc Webinar - Unlock the Power of AI-Driven Data Discovery

TrustArc Webinar - Unlock the Power of AI-Driven Data Discovery
Six Myths about Ontologies: The Basics of Formal Ontology

Six Myths about Ontologies: The Basics of Formal Ontology
Chapter #3 Lectures Part Ii
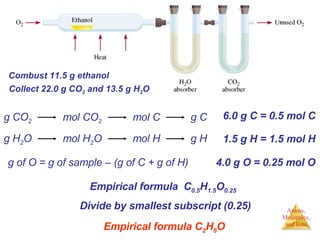
- 1. g of O = g of sample – (g of C + g of H) Combust 11.5 g ethanol Collect 22.0 g CO 2 and 13.5 g H 2 O 6.0 g C = 0.5 mol C 1.5 g H = 1.5 mol H 4.0 g O = 0.25 mol O Empirical formula C 0.5 H 1.5 O 0.25 Divide by smallest subscript (0.25) Empirical formula C 2 H 6 O g CO 2 mol CO 2 mol C g C g H 2 O mol H 2 O mol H g H
- 2. A process in which one or more substances is changed into one or more new substances is a chemical reaction A chemical equation uses chemical symbols to show what happens during a chemical reaction
- 3. How to “Read” Chemical Equations 2 Mg + O 2 2 MgO
- 10. Methanol burns in air according to the equation If 209 g of methanol are used up in the combustion, what mass of water is produced? 2CH 3 OH + 3O 2 2CO 2 + 4H 2 O
- 14. In one process, 124 g of Al are reacted with 601 g of Fe 2 O 3 Calculate the mass of Al 2 O 3 formed. 2Al + Fe 2 O 3 Al 2 O 3 + 2Fe
- 15. Use limiting reagent (Al) to calculate amount of product that can be formed.
- 16. Theoretical Yield is the amount of product that would result if all the limiting reagent reacted. Actual Yield is the amount of product actually obtained from a reaction.