255Ch5OverheadsSu14.ppt
•Télécharger en tant que PPT, PDF•
0 j'aime•3 vues
This document discusses types of isomers including enantiomers, which are nonsuperimposable mirror images. It provides examples of enantiomers including ibuprofen's R and S forms which have different biological activities. It also discusses other isomer types like diastereomers and conformational isomers. Key concepts covered include chirality, stereocenters, Fischer projections, optical activity, and methods for separating enantiomers including forming diastereomeric salts.
Signaler
Partager
Signaler
Partager
Recommandé
Recommandé
Contenu connexe
Similaire à 255Ch5OverheadsSu14.ppt
Similaire à 255Ch5OverheadsSu14.ppt (20)
Structural isomers and stereoisomers configuration | types of isomers | isome...

Structural isomers and stereoisomers configuration | types of isomers | isome...
Plus de ssusera6a60c1
Plus de ssusera6a60c1 (8)
EXP-PR-SM070-EN-RO%20-%20Transmission%20Systems-1.pptx

EXP-PR-SM070-EN-RO%20-%20Transmission%20Systems-1.pptx
Dernier
Booking open Available Pune Call Girls Koregaon Park 6297143586 Call Hot Indian Girls Waiting For You To Fuck
Booking Contact Details
WhatsApp Chat: +91-6297143586
pune Escort Service includes providing maximum physical satisfaction to their clients as well as engaging conversation that keeps your time enjoyable and entertaining. Plus they look fabulously elegant; making an impressionable.
Independent Escorts pune understands the value of confidentiality and discretion - they will go the extra mile to meet your needs. Simply contact them via text messaging or through their online profiles; they'd be more than delighted to accommodate any request or arrange a romantic date or fun-filled night together.
We provide -
01-may-2024(v.n)
Booking open Available Pune Call Girls Koregaon Park 6297143586 Call Hot Ind...

Booking open Available Pune Call Girls Koregaon Park 6297143586 Call Hot Ind...Call Girls in Nagpur High Profile
Call Girls Service Nagpur Tanvi Call 7001035870 Meet With Nagpur Escorts
Booking Contact Details
WhatsApp Chat: +91-7001035870
Nagpur Escort Service includes providing maximum physical satisfaction to their clients as well as engaging conversation that keeps your time enjoyable and entertaining. Plus they look fabulously elegant; making an impressionable.
Independent Escorts Nagpur understands the value of confidentiality and discretion - they will go the extra mile to meet your needs. Simply contact them via text messaging or through their online profiles; they'd be more than delighted to accommodate any request or arrange a romantic date or fun-filled night together.
We provide -
27-april-2024(v.n)
Call Girls Service Nagpur Tanvi Call 7001035870 Meet With Nagpur Escorts

Call Girls Service Nagpur Tanvi Call 7001035870 Meet With Nagpur EscortsCall Girls in Nagpur High Profile
High Profile Call Girls Nagpur Meera Call 7001035870 Meet With Nagpur Escorts
Booking Contact Details
WhatsApp Chat: +91-7001035870
Nagpur Escort Service includes providing maximum physical satisfaction to their clients as well as engaging conversation that keeps your time enjoyable and entertaining. Plus they look fabulously elegant; making an impressionable.
Independent Escorts Nagpur understands the value of confidentiality and discretion - they will go the extra mile to meet your needs. Simply contact them via text messaging or through their online profiles; they'd be more than delighted to accommodate any request or arrange a romantic date or fun-filled night together.
We provide -
27-april-2024(v.n)
High Profile Call Girls Nagpur Meera Call 7001035870 Meet With Nagpur Escorts

High Profile Call Girls Nagpur Meera Call 7001035870 Meet With Nagpur EscortsCall Girls in Nagpur High Profile
Top Rated Pune Call Girls Budhwar Peth ⟟ 6297143586 ⟟ Call Me For Genuine Sex Service At Affordable Rate
Booking Contact Details
WhatsApp Chat: +91-6297143586
pune Escort Service includes providing maximum physical satisfaction to their clients as well as engaging conversation that keeps your time enjoyable and entertaining. Plus they look fabulously elegant; making an impressionable.
Independent Escorts pune understands the value of confidentiality and discretion - they will go the extra mile to meet your needs. Simply contact them via text messaging or through their online profiles; they'd be more than delighted to accommodate any request or arrange a romantic date or fun-filled night together.
We provide -
01-may-2024(v.n)
Top Rated Pune Call Girls Budhwar Peth ⟟ 6297143586 ⟟ Call Me For Genuine Se...

Top Rated Pune Call Girls Budhwar Peth ⟟ 6297143586 ⟟ Call Me For Genuine Se...Call Girls in Nagpur High Profile
Booking open Available Pune Call Girls Pargaon 6297143586 Call Hot Indian Girls Waiting For You To Fuck
Booking Contact Details
WhatsApp Chat: +91-6297143586
pune Escort Service includes providing maximum physical satisfaction to their clients as well as engaging conversation that keeps your time enjoyable and entertaining. Plus they look fabulously elegant; making an impressionable.
Independent Escorts pune understands the value of confidentiality and discretion - they will go the extra mile to meet your needs. Simply contact them via text messaging or through their online profiles; they'd be more than delighted to accommodate any request or arrange a romantic date or fun-filled night together.
We provide -
01-may-2024(v.n)
Booking open Available Pune Call Girls Pargaon 6297143586 Call Hot Indian Gi...

Booking open Available Pune Call Girls Pargaon 6297143586 Call Hot Indian Gi...Call Girls in Nagpur High Profile
Dernier (20)
Call Girls Pimpri Chinchwad Call Me 7737669865 Budget Friendly No Advance Boo...

Call Girls Pimpri Chinchwad Call Me 7737669865 Budget Friendly No Advance Boo...
Booking open Available Pune Call Girls Koregaon Park 6297143586 Call Hot Ind...

Booking open Available Pune Call Girls Koregaon Park 6297143586 Call Hot Ind...
Call Girls Service Nagpur Tanvi Call 7001035870 Meet With Nagpur Escorts

Call Girls Service Nagpur Tanvi Call 7001035870 Meet With Nagpur Escorts
Roadmap to Membership of RICS - Pathways and Routes

Roadmap to Membership of RICS - Pathways and Routes
result management system report for college project

result management system report for college project
High Profile Call Girls Nagpur Meera Call 7001035870 Meet With Nagpur Escorts

High Profile Call Girls Nagpur Meera Call 7001035870 Meet With Nagpur Escorts
Call for Papers - African Journal of Biological Sciences, E-ISSN: 2663-2187, ...

Call for Papers - African Journal of Biological Sciences, E-ISSN: 2663-2187, ...
Top Rated Pune Call Girls Budhwar Peth ⟟ 6297143586 ⟟ Call Me For Genuine Se...

Top Rated Pune Call Girls Budhwar Peth ⟟ 6297143586 ⟟ Call Me For Genuine Se...
UNIT-V FMM.HYDRAULIC TURBINE - Construction and working

UNIT-V FMM.HYDRAULIC TURBINE - Construction and working
Structural Analysis and Design of Foundations: A Comprehensive Handbook for S...

Structural Analysis and Design of Foundations: A Comprehensive Handbook for S...
MANUFACTURING PROCESS-II UNIT-1 THEORY OF METAL CUTTING

MANUFACTURING PROCESS-II UNIT-1 THEORY OF METAL CUTTING
(PRIYA) Rajgurunagar Call Girls Just Call 7001035870 [ Cash on Delivery ] Pun...![(PRIYA) Rajgurunagar Call Girls Just Call 7001035870 [ Cash on Delivery ] Pun...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![(PRIYA) Rajgurunagar Call Girls Just Call 7001035870 [ Cash on Delivery ] Pun...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
(PRIYA) Rajgurunagar Call Girls Just Call 7001035870 [ Cash on Delivery ] Pun...
Booking open Available Pune Call Girls Pargaon 6297143586 Call Hot Indian Gi...

Booking open Available Pune Call Girls Pargaon 6297143586 Call Hot Indian Gi...
Introduction to IEEE STANDARDS and its different types.pptx

Introduction to IEEE STANDARDS and its different types.pptx
255Ch5OverheadsSu14.ppt
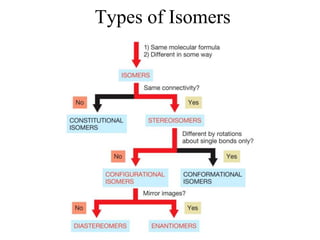
- 2. Enantiomers • Enantiomers are nonsuperimposable mirror images. • Molecules are nonsuperimposable if there is no orientation in which all atoms of both molecules can be superimposed.
- 3. Ibuprofen R form non-active side effects S form active anti-inflammatory
- 4. Thalidomide is now being used to treat plasma cell cancer, leprosy, and has shown anti-HIV activity
- 5. Chirality • A molecule is chiral if it has an enantiomer. – Does NOT have mirror symmetry • A molecule is achiral if it does not have an enantiomer. – DOES have mirror symmetry
- 6. Terminology in Stereochemistry • Stereocenter – An atom about which exchange of 2 groups produces a stereoisomer • Chiral Center – Type of Stereocenter – A tetrahedral atom that is bonded to four different groups
- 8. Fischer Projections • The Fischer projection is a convenient way to depict complex molecules having more than one stereocenter. • The intersection of a horizontal line and a vertical line indicates a carbon stereocenter. • The substituents on the horizontal bonds are understood to point toward you (like a bowtie), whereas the substituents on the vertical bonds are understood to point away from you.
- 9. Chirality and Conformational Isomers • Rotation about single bonds can determine whether a molecule is chiral or achiral. • If a molecule and its mirror image are rapidly interconverting conformational isomers, then the molecule is effectively achiral. • The mirror image of 1,2-dibromoethane in one of its gauche conformations is its second gauche conformation.
- 10. Haworth Projections • Haworth projections are helpful in determining the chirality of all cyclics. • Taking the chair conformation of cyclohexane to a flat hexagon allows us to quickly and accurately determine whether it is chiral or achiral.
- 11. biphenyls has been resolved half-life for racemization is 78 min at 118oC “Frozen” Conformational Isomers
- 12. Physical and Chemical Properties of Isomers Constitutional Isomers Due to different connectivities, these isomers must have different physical and chemical properties. Enantiomers Have the same connectivities and precisely the same polarities. Diastereomers Have the same connectivities but different 3-D arrangement, so they have different properties.
- 13. Optical Activity • Chiral molecules interact with plane-polarized light. • When all photons from a light source have their electric fields oscillating in the same plane, then the light is plane polarized.
- 14. The Polarizer • Most light sources emit light that is unpolarized. • A polarizer generates plane-polarized light by filtering out light whose electric field oscillates in any other plane. • If plane-polarized light passes through a sample of a compound, the plane in which the light is polarized can change, depending upon whether the compound is chiral or achiral.
- 15. Enantiomers rotate Plane Polarized Light • One enantiomer rotates polarized light in one direction while the other enantiomer rotates it in the opposite direction. • Enantiomers have identical physical and chemical properties except the direction at which they rotate polarized light.
- 17. Louis Pasteur • Louis Pasteur was the first to isolate a pair of enantiomers from each other. • Pasteur noted that the crystals appeared to grow in one of two varieties—left- handed crystals and right-handed crystals—that are mirror images of each other • Pasteur physically separated the two types of crystals using tweezers.
- 18. Option for Separating Enantiomers 1.Pasteur Method 2.Conversion to Diastereomers 1. Temporarily convert the enantiomers into a pair of diastereomers (will now have different physical properties). 2. Separate those diastereomers from each other by exploiting their different physical and chemical properties. 3. Regenerate the enantiomers from the separated diastereomers. 3.Enzymes (version of #2) 4.Chiral Chromatography (version of #2) Separating (Resolving) Enantiomers
- 19. enantiomers to be separated one enantiomer of a chiral amine diastereomeric salts one diastereomer one pure enantiomer