Mitral Regurgitation: Causes, Symptoms and Treatment
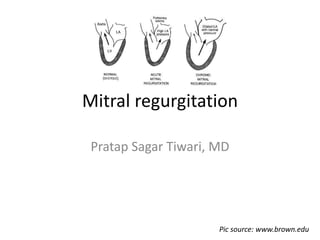
- 1. Mitral regurgitation Pratap Sagar Tiwari, MD Pic source: www.brown.edu
- 2. Overview • Mitral regurgitation (MR) is defined as an abnormal reversal of blood flow from the left ventricle (LV) to the left atrium (LA). • It is caused by disruption in any part of the mitral valve (MV) apparatus. • Mitral regurgitation can be divided into 3 stages: 1. acute 2. chronic compensated 3. chronic decompensated Pic source: www.abbottvascular.com
- 3. Anatomy: Mitral valve The mitral apparatus is composed of 1. the left atrial wall 2. the annulus 3. the leaflets 4. the chordae tendineae 5. the papillary muscles 6. the left ventricular wall • The mitral valve connects the left atrium (LA) and the left ventricle (LV). • The mitral valve opens during diastole to allow the blood flow from the LA to the LV. • During ventricular systole, the mitral valve closes and prevents backflow to the LA. Pic source: www.heart-valve-surgery.com
- 4. Anatomy: MV Annulus 1. Figure2: http://emedicine.medscape.com • The mitral annulus is a fibrous ring that connects with the leaflets. • The annulus functions as a sphincter that contracts and reduces the surface area of the valve during systole to ensure complete closure of the leaflets. • Annulus contraction during systole promotes valve competence • Thus, annular dilatation of the mitral valve causes poor leaflet apposition, which results in mitral regurgitation. • Annulus may dilate in ischemic or dilated cardiomyopathy.
- 5. Anatomy: Mitral valve leaflets 1. Picture source: http://emedicine.medscape.com 2. Harken DE, Ellis LB, Dexter L, Farrand RE, Dickson JF. The responsibility of the physician in the selection of patients with mitral stenosis for surgical treatment. Circulation. Mar 1952;5(3):349-62. Harken et al have described the mitral valve as a continuous veil inserted around the circumference of the mitral orifice.[2] The free edges of the leaflets have several indentations. Two of these indentations, the anterolateral and posteromedial commissures, divide the leaflets into anterior and posterior . These commissures can be accurately identified by the insertions of the commissural chordae tendineae into the leaflets. • Inflammatory, degenerative, or infectious diseases can result in perforation or poor coaptation of leaflets 1
- 6. Anatomy: Chordae tendineae 1. Pic source: http://emedicine.medscape.com • These are small fibrous strings that originate either from the apical portion of the papillary muscles or directly from the ventricular wall and insert into the valve leaflets or the muscle. • Chordal rupture or foreshortening prevents leaflet coaptation 1
- 7. Anatomy: Papillary muscles and left ventricular wall 1. http://emedicine.medscape.com 2. Harken DE, Ellis LB, Dexter L, Farrand RE, Dickson JF. The responsibility of the physician in the selection of patients with mitral stenosis for surgical treatment. Circulation. Mar 1952;5(3):349-62. • These 2 structures represent the muscular components of the mitral apparatus. • PM normally arise from apex and mid third of the LV wall. • The anterolateral PM is normally larger than the posteromedial PM & is supplied by LAD artery or LCX artery. • The posteromedial PM is supplied by the RCA. • Rupture of a PM, usually the complication of AMI, will result in acute MR. • Infarction or LV wall motion abnormalities may lead to valve incompetence. • Infarction of PM itself more likely to cause acute MR. 1
- 8. Anatomy: Mitral valve 1. Figure2: http://emedicine.medscape.com 2. Perloff JK, Roberts WC. The mitral apparatus. Functional anatomy of mitral regurgitation. Circulation. Aug 1972;46(2):227-39. Left atrial wall • The left atrial myocardium extends over the proximal portion of the posterior leaflet. • Thus, left atrial enlargement can result in MR by affecting the posterior leaflet. The anterior leaflet is not affected, because of its attachment to the root of the aorta.2
- 9. Mitral regurgitation: etiology Acute Chronic Primary MR Endocarditis Papillary muscle rupture (post-MI) Trauma Chordal rupture/leaflet flail (MVP, IE) Myxomatous (MVP) Rheumatic fever Endocarditis (healed) Mitral annular calcification Congenital (cleft, AV canal) HOCM with SAM Radiation Chronic secondary MR Ischemic (LV remodeling) Dilated cardiomyopathy The abnormal and dilated left ventricle causes papillary muscle displacement, which in turn results in leaflet tethering with associated annular dilation that prevents coaptation.
- 12. Laplace Law • The hemodynamic changes in acute MR are more severe than those in chronic MR due in part to the lack of time for the left atrium and left ventricle to adapt to the MR. • This is in contrast to chronic MR where these adaptations have time to develop and typically preserve hemodynamic stability. As stated by Laplace law: LV wall stress is directly proportional to the cavity pressure and inversely proportional to wall thickness. Due to the added regurgitant vol there is ↑ in LV volume that leads to an ↑ in LV cavity pressure and it causes an ↑in wall stress. If a hypertrophic response occurs, ↑↑ thickness can return wall stress to N.
- 13. Chronic MR: Pathophysiology Vol load imposed on LA & LV (usually it gradually ↑ over time) Large total SV (supra normal EF) and normal forward SV MR begets MR (viscious cycle in which further LV/annular dilatation ↑ MR ↑ Preload, LV hypertrophy, & reduced or normal afterload (low resistance LA provides unloading of LV) ↑ LVEDP &↑ LAP Compensatory dilatation of LA & LV to accommodate vol load at lower pressure; this helps relieve pul congestion LV hypertrophy (eccentric) stimulated by LV dilatation (↑ wall stress- Laplace Law)
- 14. Chronic MR: Pathophysiology..continue Reduced forward SV/CO MR begets MR (viscious cycle in which further LV/annular dilatation ↑ MR Pul congestion & pHTN Contractile dysfunction ↓ EF, ↑ end-systolic volume ↑ LVEDP/ vol, ↑ LAP
- 15. Basic mechanisms of acute native valve acute MR 1. Flail leaflet due to myxomatous disease (MVP), IE, or trauma. 2. Chordae tendineae rupture due to trauma, IE, or acute rheumatic fever. 3. Papillary muscle rupture or displacement due to AMI or severe ischemia or trauma.
- 16. Pathophysiology: AMR • Flail leaflet • Chordae tendineae rupture • Papillary muscle rupture MR Abnormal reversal of blood flow from :LV LA. Sudden large Vol load imposed on LA & LV of normal size & compliance ↑ LA volume /LAP Pulmonary congestion ↓ Forward SV Neurohumoral Response : ↑Vascular resistance ↓ CO Compensatory ↑ HR Shock
- 17. Neurohumoral Responses Activation of RAAS Low output state Contribute to maintenance of perfusion of vital organs : 1. Maintenance of systemic pressure by vasoconstriction, resulting in redistribution of blood flow to vital organs 2. Restoration of CO by ↑ myocardial contractility and HR and by expansion of the ECF volume • Catecholamine-stimulated contractility and ↑ HR. • Volume expansion ↑ venous return ↑ EDV ↑SV(via FSM) Activation of SNS
- 18. Neurohumoral Maladaptive Responses Activation of RAAS Low output state • Catecholamine-stimulated contractility and ↑ HR. • Volume expansion ↑ venous return ↑ EDV ↑SV(via FSM) Activation of SNS Maladaptive responses as below • The ↑ in diastolic pressures transmitted to the atria and to the pulmonary circulations↑ in capillary pressures pulmonary congestion & peripheral edema • The ↑ in LV afterload induced by the ↑ in PVR can both directly depress cardiac fn and enhance the rate of deterioration of myocardial function . • Catecholamine-stimulated contractility & ↑HR worsen coronary ischemia (also ↑HR ↓EDFilling ↓EDV ↓ SV/CO • Catecholamines and ATII promote the loss of myocytes by apoptosis, and hypertrophy
- 19. Eccentric vs concentric hypertrophy EH :where the walls and chamber of a hollow organ undergo growth in which the overall size and volume are enlarged. It is applied especially to the LV.Sarcomeres are added in series, as for example in DCM (in contrast to HOCM, a type of CH, where sarcomeres are added in parallel). Pic source: www.revespcardiol.org
- 20. Functional mitral regurgitation In patients with functional or secondary mitral regurgitation (MR), the papillary muscles, chordae, and valve leaflets are normal. • There are two major causes of this problem: 1. ischemia 2. any cause of dilated left ventricle. In these settings, MR may result from one or both of the following [1]: • Annular enlargement secondary to left ventricular dilatation • Papillary muscle displacement due to left ventricular remodeling, which results in tethering and excess tenting of the leaflet .2 1. Trichon BH, O'Connor CM. Secondary mitral and tricuspid regurgitation accompanying left ventricular systolic dysfunction: is it important, and how is it treated? Am Heart J 2002; 144:373. 2. Yiu SF, Enriquez-Sarano M, Tribouilloy C, et al. Determinants of the degree of functional mitral regurgitation in patients with systolic left ventricular dysfunction: A quantitative clinical study. Circulation 2000; 102:1400.
- 21. PREVALENCE of FMR • Some degree of functional MR is almost always present in patients with severe dilated cardiomyopathy, regardless of etiology [1,2,3]. • A review of 50 patients with DCM(36 idiopathic, 14 ischemic) referred for heart transplantation found that all patients had at least moderate MR by Doppler echo [2]. • Functional MR is also a common problem in patients with ESRD on maintenance HD. In this setting, volume expansion rather than intrinsic cardiac disease is primarily responsible for the cardiac enlargement. 1. Trichon BH, O'Connor CM. Secondary mitral and tricuspid regurgitation accompanying left ventricular systolic dysfunction: is it important, and how is it treated? Am Heart J 2002; 144:373. 2. Strauss RH, Stevenson LW, Dadourian BA, Child JS. Predictability of mitral regurgitation detected by Doppler echocardiography in patients referred for cardiac transplantation. Am J Cardiol 1987; 59:892. 3. Koelling TM, Aaronson KD, Cody RJ, et al. Prognostic significance of mitral regurgitation and tricuspid regurgitation in patients with left ventricular systolic dysfunction. Am Heart J 2002; 144:524.
- 22. Ischemic mitral regurgitation • Ischemic MR is a complication of coronary heart disease. • It primarily occurs in patients with a prior myocardial infarction (MI). • MR may also occur with acute ischemia, a setting in which the MR typically resolves after the ischemia resolves. • Following an MI, the MR is usually due to infarction with permanent damage to the papillary muscle or adjacent myocardium.
- 23. IMR:classified by the mechanism of the valve dysfunction 1. IMR secondary to papillary muscle rupture is a life- threatening complication of acute MI that accounts for 5 % of deaths in these patients . This complication usually occurs 2-7days after the infarct. 2. The papillary muscle may be infarcted with acute MI, but not ruptured. 3. The vast majority of patients have "functional" ischemic MR. In these individuals, the papillary muscles, chordae, and valve leaflets are normal . However, the leaflets do not coapt and restricted leaflet motion is frequently noted on echo.
- 24. IMR: papillary muscle dysfunction • The term "papillary muscle dysfunction" has been used in the literature to describe patients with functional ischemic MR, but this is a misnomer since papillary muscle dysfunction does not explain the mechanism of this disorder [1,2,3]. • The mechanism for MR in this setting is better described as "papillary muscle displacement" due to dysfunction of underlying left ventricular wall and associated alteration of ventricular geometry. 1. Levine RA, Schwammenthal E. Ischemic mitral regurgitation on the threshold of a solution: from paradoxes to unifying concepts. Circulation 2005; 112:745. 2. Kaul S, Spotnitz WD, Glasheen WP, Touchstone DA. Mechanism of ischemic mitral regurgitation. An experimental evaluation. Circulation 1991; 84:2167. 3. Uemura T, Otsuji Y, Nakashiki K, et al. Papillary muscle dysfunction attenuates ischemic mitral regurgitation in patients with localized basal inferior left ventricular remodeling: insights from tissue Doppler strain imaging. J Am Coll Cardiol 2005; 46:113.
- 25. Auscultation • S1 is generally absent, soft, or buried in the holosystolic murmur of chronic MR. • In patients with severe MR, the AV may close prematurely, resulting in wide but physiologic splitting of S2. • A low-pitched S3 occurring 0.12–0.17 s after the AV closure sound, i.e., at the completion of the rapid-filling phase of the LV, : DUE TO sudden tensing of the papillary muscles, chordae tendineae, and valve leaflets. It may be followed by a short, rumbling, mid-diastolic murmur, even in the absence of structural MS. • S4 is often audible in ps with acute severe MR who are in sinus rhythm.
- 26. Auscultation • Chronic severe MR: A systolic murmur of at least grade III/VI intensity and is usually holosystolic. • The systolic murmur of chronic MR is usually most prominent at the apex and radiates to the axilla. • Acute severe MR: is decrescendo and ceases in mid- to late systole .
- 27. Auscultation • Ruptured chordae tendineae or primary involvement of the posterior mitral leaflet with prolapse or flail : regurgitant jet is eccentric, directed anteriorly, and strikes the LA wall the systolic murmur is transmitted to the base of the heart (therefore, may be confused with the murmur of AS). • ruptured chordae tendineae, the systolic murmur may have a cooing or "sea gull" quality. • A flail leaflet: murmur with a musical quality. • The systolic murmur of chronic MR not due to MVP is intensified by isometric exercise (handgrip:↑LVV) but is reduced during the strain phase of the Valsalva maneuver because of the associated decrease in LV preload.
- 28. Mitral valve prolapse • The most important finding is the mid- or late (nonejection) systolic click, which occurs 0.14 s or more after S1 and is thought to be generated by the sudden tensing of slack, elongated chordae tendineae or by the prolapsing mitral leaflet when it reaches its maximum excursion. • Systolic clicks may be multiple and may be followed by a high-pitched, late systolic crescendo-decrescendo murmur, which occasionally is "whooping" or "honking" and is heard best at the apex. • The click and murmur occur earlier with standing, during the strain phase of the Valsalva maneuver, and with any intervention that ↓ LV volume, exaggerating the propensity of mitral leaflet prolapse. • Conversely, squatting and isometric exercises, which ↑LV volume, diminish MVP; the click-murmur complex is delayed, moves away from S1, and may even disappear. • Some patients have a mid-systolic click without a murmur; others have a murmur without a click. Still others have both sounds at different times.
- 29. Acute vs Chronic MR
Notes de l'éditeur
- Mitral valve regurgitation occurs when the mitral valve fails to close completely during ventricular systole, which causes blood to flow back (regurgitate) into the left atrium (LA) as the left ventricle (LV) contracts (see figure at right). This causes the left atrium to be become engorged with blood because blood is entering the LA from the LV during ventricular systole as well as from the pulmonary veins. This causes LA pressure to increase (25 mmHg in this example). During LV filling, the higher pressure and volume of the LA leads to an increase in LV end-diastolic pressure (25 mmHg in this example) and LV end-diastolic volume. This increase in LV preloadcauses the LV to contract more forcefully (Frank-Starling mechanism), which enables it to increase its stroke volume. Although the LV stroke volume (end-diastolic minus end-systolic volume) is increased, the net amount of blood ejected into the aorta is reduced because part of the LV stroke volume (regurgitant fraction) is also ejected into the LA. If the volume of blood ejected into the aorta is sufficiently reduced, then aortic pressure may fall (110/75 mmHg in this example). In acute mitral regurgitation (e.g., after sudden rupture of the chordae tendineae), the atrial pressure can become very elevated. In long-standing or chronic mitral regurgitation, the left atrium adapts to the larger volume by dilating, which increases its compliance. The LV also undergoes anatomic dilation. This remodeling helps to limit the increases in LA and LV pressures. The backward flow of blood into the LA during ventricular systole results in a holosystolic murmur.
- To understand the pathophysiology of condition like mitral regurgitation ,it is important to understand the law of laplace. As stated by Laplace law: left ventricular (LV) wall stress is directly proportional to the cavity pressure and inversely proportional to wall thickness. Due to the added regurgitant vol there is increase in LV volume that leads to an elevation in LV cavity pressure and it causes an increase in wall stress. If a hypertrophic response occurs, increasing thickness can return wall stress to normal. All forms of MR produce a similar hemodynamic abnormality. The inability of the MV leaflets to remain closed or coapted during systole results in a portion of the left ventricular stroke volume leaking back from the L Vinto the LA. The added volume of regurgitant blood produces an increase in LVEDV and that results in an elevation of wall stress. The heart responds with compensatory increase in wall thickness ,so that increasing thickness can return wall stress to normal. THIS compensatory myocardial hypertrophy occurs in chornic MR. The combination of hypertrophy and chamber enlargement raises the total stroke volume. AND also as we remember the starlings law ie the increase in LDEDV ie the preload results in increased stretching of the myocytes leading to increased contractility. The net effect is that forward stroke volume and hence CO are initially maintained or the EF can even be supra normal despite the regurgitant lesion. Thus, the heart initially adapts well to chronic MR, functioning as a very efficient and compliant high output pump.
- The hemodynamic changes in acute MR are more severe than those in chronic MR due in part to the lack of time for the left atrium and left ventricle to adapt to the mitral regurgitation. This is in contrast to chronic MR where these adaptations have time to develop and typically preserve hemodynamic stability. The degree of hemodynamic deterioration in acute MR depends upon the etiology and degree of MR, which is often dramatic and rapid in onset. An important factor is left atrial compliance, which is usually normal unless the acute regurgitation is superimposed upon chronic MR. Since the normal left atrium is not compliant, the sudden and marked increase in left atrial volume in acute MR results in an abrupt elevation in pressure within the left atrium. This is immediately reflected back into the pulmonary circulation, often leading to pulmonary edema. In addition, because the left ventricle is not dilated and most of the blood ejected by the ventricle goes backward across the mitral valve, effective forward flow is limited. Despite a compensatory increase in heart rate, cardiac output falls, possibly precipitating cardiogenic shock. The neurohumoral response to the reduction in cardiac output is an increase in vascular resistance, which exacerbates the regurgitation.
- However, in patients with ruptured chordae tendineae or primary involvement of the posterior mitral leaflet with prolapse or flail, the regurgitant jet is eccentric, directed anteriorly, and strikes the LA wall adjacent to the aortic root. In this situation, the systolic murmur is transmitted to the base of the heart and, therefore, may be confused with the murmur of AS. In patients with ruptured chordae tendineae, the systolic murmur may have a cooing or "sea gull" quality, while a flail leaflet may produce a murmur with a musical quality. The systolic murmur of chronic MR not due to MVP is intensified by isometric exercise (handgrip) but is reduced during the strain phase of the Valsalva maneuver because of the associated decrease in LV preload.
- The click moves closer to the 1st heart sound (S1) with maneuvers that decrease left ventricle (LV) size (eg, sitting, standing, Valsalva maneuver); the same maneuvers cause an MR murmur to appear or become louder and last longer. These effects occur because decreasing LV size causes papillary muscles and chordae tendineae to pull together more centrally beneath the valve, resulting in quicker, more forceful prolapse with earlier, more severe regurgitation. Conversely, squatting or isometric handgrip delays the S1 click and shortens the MR murmur. The systolic click may be confused with the click of congenital aortic stenosis; the latter may be distinguished because it occurs very early in systole and does not move with postural or LV volume changes. Other findings include a systolic honk or whoop, thought to be caused by valvular leaflet vibration; these findings are usually transient and may vary with respiratory phase. An early diastolic opening snap caused by return of the prolapsed valve to its normal position is rarely heard. In some patients, especially children, the findings of MVP may be more noticeable after exertion.
