Chemistry- Chp 18 - Reaction Rates and Equilibrium - Study Guide
•Télécharger en tant que DOC, PDF•
4 j'aime•1,777 vues
Signaler
Partager
Signaler
Partager
Recommandé
Contenu connexe
Tendances
Tendances (20)
Chem 2 - Chemical Equilibrium X: Le Chatelier's Principle and Temperature Cha...

Chem 2 - Chemical Equilibrium X: Le Chatelier's Principle and Temperature Cha...
Chem 2 - Chemical Equilibrium VI: Heterogeneous Equilibria

Chem 2 - Chemical Equilibrium VI: Heterogeneous Equilibria
Similaire à Chemistry- Chp 18 - Reaction Rates and Equilibrium - Study Guide
Similaire à Chemistry- Chp 18 - Reaction Rates and Equilibrium - Study Guide (20)
UNIT 8 CHEMICAL KINETICS.pptxUNIT 8 CHEMICAL KINETICS.pptx

UNIT 8 CHEMICAL KINETICS.pptxUNIT 8 CHEMICAL KINETICS.pptx
ctre 3&4-1.pdf chemical reaction engineering batch reactor

ctre 3&4-1.pdf chemical reaction engineering batch reactor
Chemical-Equilibrium-Class-10-important-questions.pdf

Chemical-Equilibrium-Class-10-important-questions.pdf
Plus de Mr. Walajtys
Plus de Mr. Walajtys (20)
Chemistry - Chp 20 - Oxidation Reduction Reactions - PowerPoint

Chemistry - Chp 20 - Oxidation Reduction Reactions - PowerPoint
Chemistry - Chp 19 - Acids, Bases, and Salt - PowerPoints

Chemistry - Chp 19 - Acids, Bases, and Salt - PowerPoints
Chemistry - Chp 11 - Chemical Reactions - PowerPoint

Chemistry - Chp 11 - Chemical Reactions - PowerPoint
Chemistry - Chp 10 - Chemical Quantities - PowerPoint

Chemistry - Chp 10 - Chemical Quantities - PowerPoint
Chemistry - Chp 7 - Ionic and Metallic Bonding - PowerPoint

Chemistry - Chp 7 - Ionic and Metallic Bonding - PowerPoint
Chemistry - Chp 8 - Covalent Bonding - Study Guide

Chemistry - Chp 8 - Covalent Bonding - Study Guide
Chemistry - Chp 13 - States of Matter - Study Guide

Chemistry - Chp 13 - States of Matter - Study Guide
Chemistry - Chp 3 - Scientific Measurement - Study Guide 

Chemistry - Chp 3 - Scientific Measurement - Study Guide
Chemistry - Chp 14 - The Behavior of Gases - Study Guide

Chemistry - Chp 14 - The Behavior of Gases - Study Guide
Chemistry - Chp 20 - Oxidation Reduction - Study Guide

Chemistry - Chp 20 - Oxidation Reduction - Study Guide
Dernier
Dernier (20)
UGC NET Paper 1 Mathematical Reasoning & Aptitude.pdf

UGC NET Paper 1 Mathematical Reasoning & Aptitude.pdf
Basic Civil Engineering first year Notes- Chapter 4 Building.pptx

Basic Civil Engineering first year Notes- Chapter 4 Building.pptx
Unit-IV; Professional Sales Representative (PSR).pptx

Unit-IV; Professional Sales Representative (PSR).pptx
Micro-Scholarship, What it is, How can it help me.pdf

Micro-Scholarship, What it is, How can it help me.pdf
Mixin Classes in Odoo 17 How to Extend Models Using Mixin Classes

Mixin Classes in Odoo 17 How to Extend Models Using Mixin Classes
Python Notes for mca i year students osmania university.docx

Python Notes for mca i year students osmania university.docx
This PowerPoint helps students to consider the concept of infinity.

This PowerPoint helps students to consider the concept of infinity.
HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx

HMCS Max Bernays Pre-Deployment Brief (May 2024).pptx
Vishram Singh - Textbook of Anatomy Upper Limb and Thorax.. Volume 1 (1).pdf

Vishram Singh - Textbook of Anatomy Upper Limb and Thorax.. Volume 1 (1).pdf
Salient Features of India constitution especially power and functions

Salient Features of India constitution especially power and functions
Chemistry- Chp 18 - Reaction Rates and Equilibrium - Study Guide
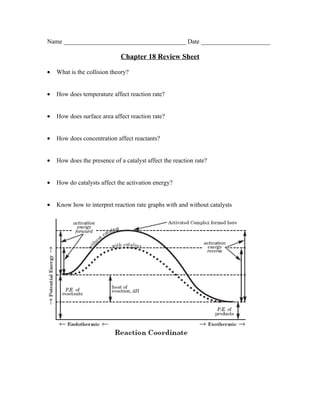
- 1. Name _______________________________________ Date ______________________ Chapter 18 Review Sheet • What is the collision theory? • How does temperature affect reaction rate? • How does surface area affect reaction rate? • How does concentration affect reactants? • How does the presence of a catalyst affect the reaction rate? • How do catalysts affect the activation energy? • Know how to interpret reaction rate graphs with and without catalysts
- 2. • What is equilibrium? How does the rates of the forward reaction compare to the rates of the reverse reaction? • What is LeChatlier’s principle? • What kind of shifts in equilibrium are expected with changes in: Concentration Temperature Pressure • Know that the amount of energy released or absorbed during a chemical reaction is the heat of reaction Heat of reaction = PE of products – PE of reactants • What is an endothermic reaction? Is their heat of reaction + or -? • What is an exothermic reaction? Is their heat of reaction + or -? • How do you calculate the Keq (equilibrium constant) for an equation Ex.) aA + bB cC dD • How does Keq determine whether the products or reactants are favored at equilibrium? • What is entropy? What is the entropy trend for systems? • What is enthalpy? What is the enthalpy trend for systems? • Spontaneous vs. non spontaneous reactions